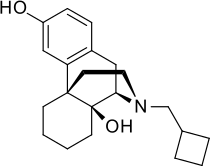
Butorphanol
Butorphanol is a morphinan-type synthetic agonist–antagonist opioid analgesic developed by Bristol-Myers.[1][2][3][4][5] Butorphanol is most closely structurally related to levorphanol. Butorphanol is available as the tartrate salt in injectable, tablet, and intranasal spray formulations. The tablet form is only used in dogs, cats and horses due to low bioavailability in humans.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Stadol, others |
| Other names | BC 2627 |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a682667 |
| Pregnancy category |
|
| Routes of administration | IV, intranasal, oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Nasal: 60-70% |
| Metabolism | Liver hydroxylated & glucuronidated |
| Elimination half-life | 4-7 hours |
| Excretion | Kidney, 75% Biliary, 11-14% Fecal, 15% |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.050.717 |
| Chemical and physical data | |
| Formula | C21H29NO2 |
| Molar mass | 327.473 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
It was patented in 1971 and approved for medical use in 1979.[6]
Medical uses
The most common indication for butorphanol is management of migraine using the intranasal spray formulation. It may also be used parenterally for management of moderate-to-severe pain, as a supplement for balanced general anesthesia, and management of pain during labor. Butorphanol is also quite effective at reducing post-operative shivering (owing to its Kappa agonist activity). Butorphanol is more effective in reducing pain in women than in men.[7]
Pharmacology
Butorphanol exhibits partial agonist and antagonist activity at the μ-opioid receptor, as well as partial agonist activity at the κ-opioid receptor (Ki = 2.5 nM; EC50 = 57 nM; Emax = 57%).[7][8] Stimulation of these receptors on central nervous system neurons causes an intracellular inhibition of adenylate cyclase, closing of influx membrane calcium channels, and opening of membrane potassium channels. This leads to hyperpolarization of the cell membrane potential and suppression of action potential transmission of ascending pain pathways. Because of its κ-agonist activity, at analgesic doses butorphanol increases pulmonary arterial pressure and cardiac work. Additionally, κ-agonism can cause dysphoria at therapeutic or supertherapeutic doses; this gives butorphanol a lower potential for abuse than other opioid drugs.[9]
Side effects
As with other opioid analgesics, central nervous system effects (such as sedation, confusion, and dizziness) are considerations with butorphanol. Nausea and vomiting are common. Less common are the gastrointestinal effects of other opioids (mostly constipation). Another side effect experienced by people taking the medication is increased perspiration.
Society and culture
Name
Butorphanol is the INN, USAN, BAN, and AAN, while torbugesic is the JAN.[3][4][5] As the tartrate salt, butorphanol is known as butorphanol tartrate (USAN, BAN).[3][4][5]
Brand name Stadol was recently discontinued by the manufacturer. It is now only available in its generic formulations, manufactured by Novex, Mylan, Apotex and Ben Venue Laboratories.
Availability
Butorphanol is available in the U.S. as a generic drug; it is available in various nations under one of any number of trade names, including Moradol and Beforal (Brand name Stadol no longer available in the US); veterinary trade names include Butorphic, Dolorex, Morphasol, Torbugesic, and Torbutrol.
Legality
Butorphanol is listed under the Single Convention on Narcotic Drugs 1961 and in the United States is a Schedule IV Narcotic controlled substance with a DEA ACSCN of 9720; being in Schedule IV it is not subject to annual aggregate manufacturing quotas. The free base conversion ratio of the hydrochloride is 0.69.[10] Butorphanol was originally in Schedule II and at one point it was decontrolled.
Veterinary use
In veterinary anesthesia, butorphanol (trade name: Torbugesic) is widely used as a sedative and analgesic in dogs, cats and horses. For sedation, it may be combined with tranquilizers such as alpha-2 agonists (medetomidine), benzodiazepines, or acepromazine in dogs, cats and exotic animals. It is frequently combined with xylazine or detomidine in horses.[11]
Butorphanol is frequently used for post-operative and accident-related pain in small mammals such as dogs, cats, ferrets, coatis, raccoons, mongooses, various marsupials, some rodents and perhaps some larger birds, both in the operating suite and as a regular prescription medication for home use, for management of moderate to severe pain.
Although butorphanol is commonly used for pain relief in reptiles, no studies (as of 2014) have conclusively shown that is an effective analgesic in reptiles.[12]
Use in horses
Butorphanol is a commonly used narcotic for pain relief in horses. It is administered either IM or IV, with its analgesic properties beginning to take effect about 15 minutes after injection and lasting 4 hours. It is also commonly paired with sedatives, such as xylazine and detomidine, to make the horse easier to handle during veterinary procedures.
Side effects specific to horses include sedation, CNS excitement (displayed by head pressing or tossing). Overdosing may result in seizures, falling, salivation, constipation, and muscle twitching. If an overdose occurs, a narcotic antagonist, such as naloxone, may be given. Caution should be used if butorphanol is administered in addition to other narcotics, sedatives, depressants, or antihistamines as it will cause an additive effect.
Butorphanol can cross the placenta, and it will be present in the milk of lactating mares who are given the drug.
The drug is also prohibited for use in competition by most equestrian organizations, including the FEI, which considers it a class A drug.
See also
Notes
- US Patent US Patent 3819635 14-hydroxymorphinan derivatives
- US 3775414 Process for the preparation of 14-hydroxymorphinan derivatives
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 200–. ISBN 978-1-4757-2085-3.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 154–. ISBN 978-3-88763-075-1.
- Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 58–. ISBN 978-94-011-4439-1.
- Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 529. ISBN 978-3-527-60749-5.
- Gear RW, Miaskowski C, Gordon NC, Paul SM, Heller PH, Levine JD (November 1999). "The kappa opioid nalbuphine produces gender- and dose-dependent analgesia and antianalgesia in patients with postoperative pain". Pain. 83 (2): 339–45. doi:10.1016/S0304-3959(99)00119-0. PMID 10534607.
- Gharagozlou P, Hashemi E, DeLorey TM, Clark JD, Lameh J (January 2006). "Pharmacological profiles of opioid ligands at kappa opioid receptors". BMC Pharmacology. 6 (1): 3. doi:10.1186/1471-2210-6-3. PMC 1403760. PMID 16433932.
- "Critical Review of Butorphanol" (PDF). 34th ECDD 2006/4.1. World Health Organization.
- "Conversion Factors for Controlled Substances". DEA Diversion Control Division. United States Drug Enforcement Administration.
- Compendium of Data sheets for Animal Medicines. 2005. National Office of Animal Health. October 2004. ISBN 978-0-9548037-0-4.
- Sladky KK (2014). "Chapter 18: Analgesia". In Mader DR, Divers SJ (eds.). Current therapy in reptile medicine & surgery. Elsevier Health Sciences. pp. 217–229. ISBN 978-0-323-24293-6.
References
- Katzung BG (2001). Basic & Clinical Pharmacology (8th ed.). New York: McGraw-Hill. ISBN 978-0-8385-0598-4.
- Talbert RL, Yee GC, Matzke GR, Wells BG, Posey LM, DiPiro JT (2005). Pharmacotherapy: A Pathophysiologic Approach (6th ed.). New York: McGraw-Hill. ISBN 0-07-141613-7.
- Forney BC (2007). Equine Medications (Revised ed.). Lexington, Kentucky: Blood Horse Publications.
- The Merck Manual of Veterinary Medicine. 2004.