Jimscaline
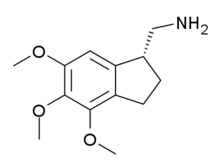
Jimscaline (C-(4,5,6-trimethoxyindan-1-yl)methanamine) is a conformationally-restricted derivative of the cactus-derived hallucinogen mescaline, which was discovered in 2006 by a team at Purdue University led by David E. Nichols. It acts as a potent agonist for the 5-HT2A and 5-HT2C receptors with the more active (R)-enantiomer having a Ki of 69 nM at the human 5-HT2A receptor, and around three times the potency of mescaline in drug-substitution experiments in animals.[1] This discovery that the side chain of the phenethylamine hallucinogens could be constrained to give chiral ligands with increased activity then led to the later development of the super-potent benzocyclobutene derivative TCB-2.[2][3]
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C13H19NO3 |
| Molar mass | 237.294 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
References
- McLean TH, Chambers JJ, Parrish JC, Braden MR, Marona-Lewicka D, Kurrasch-Orbaugh D, Nichols DE (13 July 2006), "C-(4,5,6-trimethoxyindan-1-yl)methanamine: a mescaline analogue designed using a homology model of the 5-HT2A receptor.", Journal of Medicinal Chemistry, 49 (14): 4269–74, CiteSeerX 10.1.1.690.1860, doi:10.1021/jm060272y, PMID 16821786CS1 maint: uses authors parameter (link)
- McLean TH, Parrish JC, Braden MR, Marona-Lewicka D, Gallardo-Godoy A, Nichols DE (21 September 2006), "1-Aminomethylbenzocycloalkanes: conformationally restricted hallucinogenic phenethylamine analogues as functionally selective 5-HT2A receptor agonists.", Journal of Medicinal Chemistry, 49 (19): 5794–803, CiteSeerX 10.1.1.688.9849, doi:10.1021/jm060656o, PMID 16970404CS1 maint: uses authors parameter (link)
- Michael Robert Braden PhD. Towards a biophysical understanding of hallucinogen action. Purdue University 2007.
Serotonin receptor modulators | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.