CP 47,497
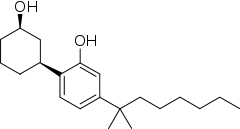
CP 47,497 or (C7)-CP 47,497 is a cannabinoid receptor agonist drug, developed by Pfizer in the 1980s.[1] It has analgesic effects and is used in scientific research. It is a potent CB1 agonist with a Kd of 2.1 nM.[2][3][4]
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| ChEMBL |
|
| Chemical and physical data | |
| Formula | C21H34O2 |
| Molar mass | 318.492 g/mol g·mol−1 |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
Homologue
On the 19th of January 2009, the University of Freiburg in Germany announced that an analog of CP 47,497 is the main active ingredient in the herbal "incense" product Spice, specifically the 1,1-dimethyloctyl homologue of CP 47,497. Both the dimethylheptyl and dimethyloctyl homologues were detected in different batches, with considerable variation in the concentration present in different samples that were analysed. The weaker dimethylhexyl and dimethylnonyl homologues were not found in any batches of smoking blends tested, but have been legally scheduled alongside the others in some jurisdictions, to forestall any potential use for this purpose.[5][6][7] The 1,1-dimethyloctyl homologue of CP 47,497 is in fact several times more potent than the parent compound,[8] which is somewhat unexpected as the 1,1-dimethylheptyl is the most potent substituent in classical cannabinoid compounds such as HU-210.[9] The unapproved use of these compounds in herbal smoking blends has led to a resurgence in legitimate scientific research into their use,[10] and consequently the C8 homologue of CP 47,497 has been assigned a proper name, cannabicyclohexanol.[11]
Legal status
Germany
On 22 January 2009, CP 47,497 was added to the German controlled drug schedules ("Betäubungsmittelgesetz"),[12] along with its dimethylhexyl, dimethyloctyl and dimethylnonyl homologues.[13]
France
CP 47,497 and its C6, C8, and C9 homologues were made illegal in France on 24 February 2009.[14]
Latvia
CP 47,497 and its C6, C8, and C9 homologues were made illegal in Latvia on 28 November 2009.[15]
Lithuania
CP 47,497 and its C6, C8, and C9 homologues were made illegal in Lithuania on 5 June 2009.[16]
Sweden
CP 47,497 and its C6, C7, C8, and C9 homologues were made illegal in Sweden on 15 September 2009.
Romania
CP 47,497 and its C6, C7, C8, and C9 homologues were made illegal in Romania on 15 February 2010.(Illegal Substances in Romania after 15.02.2010
See also
- (C6)-CP 47,497
- (C8)-CP 47,497
- (C9)-CP 47,497
- CP 50,556-1
- CP 55,244
- CP 55,940
- CP-945,598
- HHC
- O-1871
References
- Weissman A, Milne GM, Melvin LS Jr. Cannabimimetic activity from CP-47,497, a derivative of 3-phenylcyclohexanol. Journal of Pharmacology and Experimental Therapeutics. 1982 Nov;223(2):516-23. PMID 6290642
- Shim JY, Welsh WJ, Howlett AC. Homology model of the CB1 cannabinoid receptor: sites critical for nonclassical cannabinoid agonist interaction. Biopolymers. 2003;71(2):169-89. PMID 12767117
- Roger Pertwee. Cannabinoids. Handbook of Experimental Pharmacology Volume 168. Springer. ISBN 3-540-22565-X
- Little PJ, et al. Pharmacology and stereoselectivity of structurally novel cannabinoids in mice. Journal of Pharmacology and Experimental Therapeutics 1988; 247:1046–1051.
- Hauptwirkstoff von „Spice“ identifiziert, University of Freiburg http://www.pr.uni-freiburg.de/pm/2009/pm.2009-01-19.19/
- Spice - weitere Analyseresultate http://www.basg.at/servlet/sls/Tornado/web/ages/content/4E5A4B86295BF5C0C125753E006A5E3C%5B%5D
- Auwärter V, et al. 'Spice' and other herbal blends: harmless incense or cannabinoid designer drugs? Journal of Mass Spectrometry. 2009 Feb 2. PMID 19189348
- Compton DR, Johnson MR, Melvin LS, Martin BR. Pharmacological profile of a series of bicyclic cannabinoid analogs: classification as cannabimimetic agents. Journal of Pharmacology and Experimental Therapeutics. 1992 Jan;260(1):201-9. PMID 1309872
- Martin BR, et al. Behavioral, biochemical, and molecular modeling evaluations of cannabinoid analogs. Pharmacology Biochemistry and Behavior. 1991 Nov;40(3):471-8. PMID 1666911
- Uchiyama N, et al. Effects of synthetic cannabinoids on electroencephalogram power spectra in rats. Forensic Science International. 2011 Jun 1. PMID 21640532
- Uchiyama N, Kikura-Hanajiri R, Ogata J, Goda Y (May 2010). "Chemical analysis of synthetic cannabinoids as designer drugs in herbal products". Forensic Science International. 198 (1–3): 31–8. doi:10.1016/j.forsciint.2010.01.004. PMID 20117892.
- "404". Bundesgesundheitsministerium.
- BGBl I Nr. 3 vom 21.01.2009, 22. BtMÄndV vom 19. Januar 2009, S. 49–50.
- "Version électronique authentifiée publiée au JO n° 0049 du 27/02/2009 - Legifrance". www.legifrance.gouv.fr.
- "Grozījumi Ministru kabineta 2005.gada 8.novembra noteikumos Nr.847 "Noteikumi par Latvijā kontrolējamajām narkotiskajām vielām, psihotropajām vielām un prekursoriem"". LIKUMI.LV.
- http://www3.lrs.lt/pls/inter3/dokpaieska.showdoc_l?p_id=345197
- Cook, Morgan (2011-02-28). "Synthetic marijuana illegal as of Tuesday". North County Times. San Diego. Archived from the original on 2011-03-03. Retrieved 2011-02-28.
- "Schedules of Controlled Substances: Temporary Placement of Five Synthetic Cannabinoids Into Schedule I". Federal Register. 1 March 2011.