Cocaine
Cocaine, also known as coke, is a strong stimulant most frequently used as a recreational drug.[10] It is commonly snorted, inhaled as smoke, or dissolved and injected into a vein.[9] Mental effects may include loss of contact with reality, an intense feeling of happiness, or agitation.[9] Physical symptoms may include a fast heart rate, sweating, and large pupils.[9] High doses can result in very high blood pressure or body temperature.[11] Effects begin within seconds to minutes of use and last between five and ninety minutes.[9] Cocaine has a small number of accepted medical uses such as numbing and decreasing bleeding during nasal surgery.[12]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | kəʊˈkeɪn |
| Trade names | Neurocaine,[1] other |
| Other names | Benzoylmethylecgonine, coke, blow, crack (in freebase form) |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Dependence liability | High[2] |
| Addiction liability | High[3] |
| Routes of administration | Topical, oral, insufflation, intravenous |
| Drug class | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability |
|
| Metabolism | liver CYP3A4 |
| Metabolites | Norcocaine, benzoylecgonine, cocaethylene |
| Onset of action | seconds to minutes[9] |
| Elimination half-life | 1 hour |
| Duration of action | 5 to 90 minutes[9] |
| Excretion | Kidney |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| ECHA InfoCard | 100.000.030 |
| Chemical and physical data | |
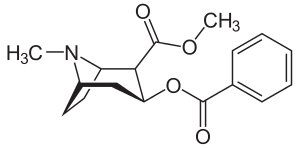
| Formula | C17H21NO4 |
| Molar mass | 303.353 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 98 °C (208 °F) |
| Boiling point | 187 °C (369 °F) |
| Solubility in water | ≈1.8 mg/mL (20 °C) |
SMILES
| |
InChI
| |
| See also: data page | |
| | |
Cocaine is addictive due to its effect on the reward pathway in the brain.[10] After a short period of use, there is a high risk that dependence will occur.[10] Its use also increases the risk of stroke, myocardial infarction, lung problems in those who smoke it, blood infections, and sudden cardiac death.[10][13] Cocaine sold on the street is commonly mixed with local anesthetics, cornstarch, quinine, or sugar, which can result in additional toxicity.[14] Following repeated doses a person may have decreased ability to feel pleasure and be very physically tired.[10]
Cocaine acts by inhibiting the reuptake of serotonin, norepinephrine, and dopamine.[10] This results in greater concentrations of these three neurotransmitters in the brain.[10] It can easily cross the blood–brain barrier and may lead to the breakdown of the barrier.[15][16] In 2013, 419 kilograms were produced legally.[17] It is estimated that the illegal market for cocaine is 100 to US$500 billion each year.[10] With further processing, crack cocaine can be produced from cocaine.[10]
Cocaine is the second most frequently used illegal drug globally, after cannabis.[18] Between 14 and 21 million people use the drug each year.[10] Use is highest in North America followed by Europe and South America.[10] Between one and three percent of people in the developed world have used cocaine at some point in their life.[10] In 2013, cocaine use directly resulted in 4,300 deaths, up from 2,400 in 1990.[19] It is named after the coca plant from which it is isolated.[9] The plant's leaves have been used by Peruvians since ancient times.[14] Cocaine was first isolated from the leaves in 1860.[10] Since 1961, the international Single Convention on Narcotic Drugs has required countries to make recreational use of cocaine a crime.[20]
Uses
Medical

Topical cocaine can be used as a local numbing agent to help with painful procedures in the mouth or nose.[21]
Cocaine is now predominantly used for nasal and lacrimal duct surgery. The major disadvantages of this use are cocaine's potential for cardiovascular toxicity, glaucoma, and pupil dilation.[21] Medicinal use of cocaine has decreased as other synthetic local anesthetics such as benzocaine, proparacaine, lidocaine, and tetracaine are now used more often.[21] If vasoconstriction is desired for a procedure (as it reduces bleeding), the anesthetic is combined with a vasoconstrictor such as phenylephrine or epinephrine. Some ENT specialists occasionally use cocaine within the practice when performing procedures such as nasal cauterization. In this scenario dissolved cocaine is soaked into a ball of cotton wool, which is placed in the nostril for the 10–15 minutes immediately before the procedure, thus performing the dual role of both numbing the area to be cauterized, and vasoconstriction. Even when used this way, some of the used cocaine may be absorbed through oral or nasal mucosa and give systemic effects. An alternative method of administration for ENT surgery is mixed with adrenaline and sodium bicarbonate, as Moffett's solution.
Recreational
Cocaine is a powerful nervous system stimulant.[22] Its effects can last from 15 or 30 minutes to an hour. The duration of cocaine's effects depends on the amount taken and the route of administration.[23] Cocaine can be in the form of fine white powder, bitter to the taste. When inhaled or injected, it causes a numbing effect. Crack cocaine is a smokeable form of cocaine made into small "rocks" by processing cocaine with sodium bicarbonate (baking soda) and water.[9][24] Crack cocaine is referred to as "crack" because of the crackling sounds it makes when heated.[9]
Cocaine use leads to increases in alertness, feelings of well-being and euphoria, increased energy and motor activity, and increased feelings of competence and sexuality.[25]
Coca leaves
Coca leaves are typically mixed with an alkaline substance (such as lime) and chewed into a wad that is retained in the mouth between gum and cheek (much the same as chewing tobacco is chewed) and sucked of its juices. The juices are absorbed slowly by the mucous membrane of the inner cheek and by the gastrointestinal tract when swallowed. Alternatively, coca leaves can be infused in liquid and consumed like tea. Ingesting coca leaves generally is an inefficient means of administering cocaine.
Because cocaine is hydrolyzed and rendered inactive in the acidic stomach, it is not readily absorbed when ingested alone. Only when mixed with a highly alkaline substance (such as lime) can it be absorbed into the bloodstream through the stomach. The efficiency of absorption of orally administered cocaine is limited by two additional factors. First, the drug is partly catabolized by the liver. Second, capillaries in the mouth and esophagus constrict after contact with the drug, reducing the surface area over which the drug can be absorbed. Nevertheless, cocaine metabolites can be detected in the urine of subjects that have sipped even one cup of coca leaf infusion.
Orally administered cocaine takes approximately 30 minutes to enter the bloodstream. Typically, only a third of an oral dose is absorbed, although absorption has been shown to reach 60% in controlled settings. Given the slow rate of absorption, maximum physiological and psychotropic effects are attained approximately 60 minutes after cocaine is administered by ingestion. While the onset of these effects is slow, the effects are sustained for approximately 60 minutes after their peak is attained.
Contrary to popular belief, both ingestion and insufflation result in approximately the same proportion of the drug being absorbed: 30 to 60%. Compared to ingestion, the faster absorption of insufflated cocaine results in quicker attainment of maximum drug effects. Snorting cocaine produces maximum physiological effects within 40 minutes and maximum psychotropic effects within 20 minutes, however, a more realistic activation period is closer to 5 to 10 minutes. Physiological and psychotropic effects from nasally insufflated cocaine are sustained for approximately 40–60 minutes after the peak effects are attained.[26]
Coca tea, an infusion of coca leaves, is also a traditional method of consumption. The tea has often been recommended for travelers in the Andes to prevent altitude sickness.[27] However, its actual effectiveness has never been systematically studied.[27]
In 1986 an article in the Journal of the American Medical Association revealed that U.S. health food stores were selling dried coca leaves to be prepared as an infusion as "Health Inca Tea."[28] While the packaging claimed it had been "decocainized," no such process had actually taken place. The article stated that drinking two cups of the tea per day gave a mild stimulation, increased heart rate, and mood elevation, and the tea was essentially harmless. Despite this, the DEA seized several shipments in Hawaii, Chicago, Georgia, and several locations on the East Coast of the United States, and the product was removed from the shelves.
Insufflation

Nasal insufflation (known colloquially as "snorting", "sniffing", or "blowing") is a common method of ingestion of recreational powdered cocaine.[29] The drug coats and is absorbed through the mucous membranes lining the nasal passages. Cocaine's desired euphoric effects are delayed when snorted through the nose by about five minutes. This occurs because cocaine's absorption is slowed by its constricting effect on the blood vessels of the nose.[9] Insufflation of cocaine also leads to the longest duration of its effects (60–90 minutes).[9] When insufflating cocaine, absorption through the nasal membranes is approximately 30–60%, with higher doses leading to increased absorption efficiency. Any material not directly absorbed through the mucous membranes is collected in mucus and swallowed (this "drip" is considered pleasant by some and unpleasant by others).
In a study of cocaine users, the average time taken to reach peak subjective effects was 14.6 minutes.[30] Any damage to the inside of the nose is because cocaine highly constricts blood vessels – and therefore blood and oxygen/nutrient flow – to that area. Nosebleeds after cocaine insufflation are due to irritation and damage of mucus membranes by foreign particles and adulterants and not the cocaine itself; as a vasoconstrictor, cocaine acts to reduce bleeding.
Rolled up banknotes, hollowed-out pens, cut straws, pointed ends of keys, specialized spoons, long fingernails, and (clean) tampon applicators are often used to insufflate cocaine. Such devices are often called "tooters" by users. The cocaine typically is poured onto a flat, hard surface (such as a mirror, CD case or book) and divided into "bumps," "lines" or "rails," and then insufflated.[31] The amount of cocaine in a line varies widely from person to person and occasion to occasion (the purity of the cocaine is also a factor), but one line is generally considered to be a single dose and is typically 35 mg (a "bump") to 100 mg (a "rail"). As tolerance builds rapidly in the short-term (hours), many lines are often snorted to produce greater effects. A 2001 study reported that the sharing of straws used to "snort" cocaine can spread blood diseases such as hepatitis C.[32]
Injection
Drug injection by turning the drug into a solution provides the highest blood levels of drug in the shortest amount of time. Subjective effects not commonly shared with other methods of administration include a ringing in the ears moments after injection (usually when in excess of 120 milligrams) lasting two to 5 minutes including tinnitus and audio distortion. This is colloquially referred to as a "bell ringer". In a study of cocaine users, the average time taken to reach peak subjective effects was 3.1 minutes.[30] The euphoria passes quickly. Aside from the toxic effects of cocaine, there is also danger of circulatory emboli from the insoluble substances that may be used to cut the drug. As with all injected illicit substances, there is a risk of the user contracting blood-borne infections if sterile injecting equipment is not available or used. Additionally, because cocaine is a vasoconstrictor, and usage often entails multiple injections within several hours or less, subsequent injections are progressively more difficult to administer, which in turn may lead to more injection attempts and more consequences from improperly performed injection.
An injected mixture of cocaine and heroin, known as "speedball" is a particularly dangerous combination, as the converse effects of the drugs actually complement each other, but may also mask the symptoms of an overdose. It has been responsible for numerous deaths, including celebrities such as comedians/actors John Belushi and Chris Farley, Mitch Hedberg, River Phoenix, grunge singer Layne Staley and actor Philip Seymour Hoffman. Experimentally, cocaine injections can be delivered to animals such as fruit flies to study the mechanisms of cocaine addiction.[33]
Inhalation
Inhalation by smoking cocaine is one of the several ways the drug is consumed. The onset of cocaine's desired euphoric effects is fastest with inhaling cocaine and begins after 3–5 seconds.[9] In contrast, inhalation of cocaine leads to the shortest duration of its effects (5–15 minutes).[9] The two main ways cocaine is smoked are freebasing and by using cocaine which has been converted to smokable "crack cocaine". Cocaine is smoked by inhaling the vapor produced when solid cocaine is heated to the point that it sublimates.[34] In a 2000 Brookhaven National Laboratory medical department study, based on self reports of 32 abusers who participated in the study,"peak high" was found at mean of 1.4min +/- 0.5 minutes.[30] Pyrolysis products of cocaine that occur only when heated/smoked have been shown to change the effect profile, i.e. anhydroecgonine methyl ester when co-administered with cocaine increases the dopamine in CPu and NAc brain regions, and has M1- and M3- receptor affinity.[35]
Smoking freebase or crack cocaine is most often accomplished using a pipe made from a small glass tube, often taken from "love roses", small glass tubes with a paper rose that are promoted as romantic gifts.[36] These are sometimes called "stems", "horns", "blasters" and "straight shooters". A small piece of clean heavy copper or occasionally stainless steel scouring pad – often called a "brillo" (actual Brillo Pads contain soap, and are not used) or "chore" (named for Chore Boy brand copper scouring pads) – serves as a reduction base and flow modulator in which the "rock" can be melted and boiled to vapor. Crack smokers also sometimes smoke through a soda can with small holes on the side or bottom. Crack is smoked by placing it at the end of the pipe; a flame held close to it produces vapor, which is then inhaled by the smoker. The effects, felt almost immediately after smoking, are very intense and do not last long – usually 2 to 10 minutes.[37] When smoked, cocaine is sometimes combined with other drugs, such as cannabis, often rolled into a joint or blunt. Powdered cocaine is also sometimes smoked, though heat destroys much of the chemical; smokers often sprinkle it on cannabis. The language referring to paraphernalia and practices of smoking cocaine vary, as do the packaging methods in the street level sale.
Suppository
Another way users consume cocaine is by making it into a suppository which they then insert into the anus or vagina. The drug is then absorbed by the membranes of these body parts. Little research has been focused on the suppository (anal or vaginal insertion) method of administration, also known as "plugging". This method of administration is commonly administered using an oral syringe. Cocaine can be dissolved in water and withdrawn into an oral syringe which may then be lubricated and inserted into the anus or vagina before the plunger is pushed. Anecdotal evidence of its effects is infrequently discussed, possibly due to social taboos in many cultures. The rectum and the vaginal canal is where the majority of the drug would be taken up through the membranes lining its walls.
Adverse effects
Acute
With excessive or prolonged use, the drug can cause itching, fast heart rate, hallucinations, and paranoid delusions or sensations of insects crawling on the skin.[38] Overdoses may cause abnormally high body temperature and a marked elevation of blood pressure, which can be life-threatening,[38] abnormal heart rhythms, and death.
Anxiety, paranoia, and restlessness can also occur, especially during the comedown. With excessive dosage, tremors, convulsions and increased body temperature are observed.[22] Severe cardiac adverse events, particularly sudden cardiac death, become a serious risk at high doses due to cocaine's blocking effect on cardiac sodium channels.
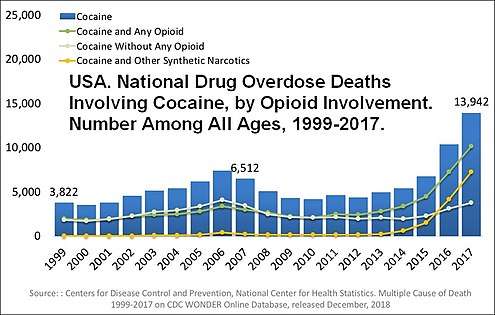 Opioid involvement in cocaine overdose deaths. Green line is cocaine and any opioid (top line in 2017). Gray line is cocaine without any opioids (bottom line in 2017). Yellow line is cocaine and other synthetic opioids (middle line in 2017).[40]
Opioid involvement in cocaine overdose deaths. Green line is cocaine and any opioid (top line in 2017). Gray line is cocaine without any opioids (bottom line in 2017). Yellow line is cocaine and other synthetic opioids (middle line in 2017).[40]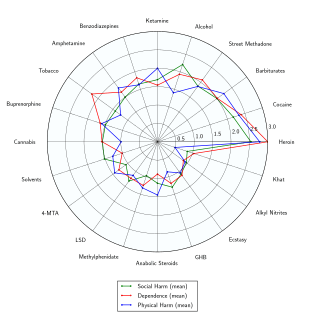 Delphic analysis regarding 20 popular recreational drugs based on expert opinion. Cocaine was ranked the 2nd in dependence and physical harm and 3rd in social harm.[41]
Delphic analysis regarding 20 popular recreational drugs based on expert opinion. Cocaine was ranked the 2nd in dependence and physical harm and 3rd in social harm.[41]
Chronic
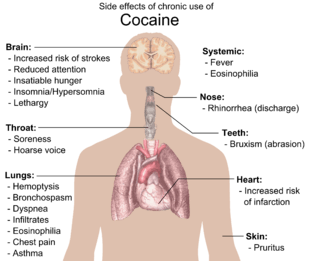
Chronic cocaine intake causes strong imbalances of transmitter levels in order to compensate extremes. Thus, receptors disappear from the cell surface or reappear on it, resulting more or less in an "off" or "working mode" respectively, or they change their susceptibility for binding partners (ligands) – mechanisms called downregulation and upregulation. However, studies suggest cocaine abusers do not show normal age-related loss of striatal dopamine transporter (DAT) sites, suggesting cocaine has neuroprotective properties for dopamine neurons.[42] Possible side effects include insatiable hunger, aches, insomnia/oversleeping, lethargy, and persistent runny nose. Depression with suicidal ideation may develop in very heavy users. Finally, a loss of vesicular monoamine transporters, neurofilament proteins, and other morphological changes appear to indicate a long term damage of dopamine neurons. All these effects contribute a rise in tolerance thus requiring a larger dosage to achieve the same effect.[43] The lack of normal amounts of serotonin and dopamine in the brain is the cause of the dysphoria and depression felt after the initial high. Physical withdrawal is not dangerous. Physiological changes caused by cocaine withdrawal include vivid and unpleasant dreams, insomnia or hypersomnia, increased appetite and psychomotor retardation or agitation.[43]
Physical side effects from chronic smoking of cocaine include coughing up blood, bronchospasm, itching, fever, diffuse alveolar infiltrates without effusions, pulmonary and systemic eosinophilia, chest pain, lung trauma, sore throat, asthma, hoarse voice, dyspnea (shortness of breath), and an aching, flu-like syndrome. Cocaine constricts blood vessels, dilates pupils, and increases body temperature, heart rate, and blood pressure. It can also cause headaches and gastrointestinal complications such as abdominal pain and nausea. A common but untrue belief is that the smoking of cocaine chemically breaks down tooth enamel and causes tooth decay. However, cocaine does often cause involuntary tooth grinding, known as bruxism, which can deteriorate tooth enamel and lead to gingivitis.[44] Additionally, stimulants like cocaine, methamphetamine, and even caffeine cause dehydration and dry mouth. Since saliva is an important mechanism in maintaining one's oral pH level, chronic stimulant abusers who do not hydrate sufficiently may experience demineralization of their teeth due to the pH of the tooth surface dropping too low (below 5.5). Cocaine use also promotes the formation of blood clots.[9] This increase in blood clot formation is attributed to cocaine-associated increases in the activity of plasminogen activator inhibitor, and an increase in the number, activation, and aggregation of platelets.[9]
Chronic intranasal usage can degrade the cartilage separating the nostrils (the septum nasi), leading eventually to its complete disappearance. Due to the absorption of the cocaine from cocaine hydrochloride, the remaining hydrochloride forms a dilute hydrochloric acid.[45]
Cocaine may also greatly increase the risk of developing rare autoimmune or connective tissue diseases such as lupus, Goodpasture syndrome, vasculitis, glomerulonephritis, Stevens–Johnson syndrome, and other diseases.[46][47][48][49] It can also cause a wide array of kidney diseases and kidney failure.[50][51]
Cocaine use leads to an increased risk of hemorrhagic and ischemic strokes.[24] Cocaine use also increases the risk of having a heart attack.[52]
Addiction
Cocaine addiction occurs through ΔFosB overexpression in the nucleus accumbens, which results in altered transcriptional regulation in neurons within the nucleus accumbens.
ΔFosB levels have been found to increase upon the use of cocaine.[53] Each subsequent dose of cocaine continues to increase ΔFosB levels with no ceiling of tolerance. Elevated levels of ΔFosB leads to increases in brain-derived neurotrophic factor (BDNF) levels, which in turn increases the number of dendritic branches and spines present on neurons involved with the nucleus accumbens and prefrontal cortex areas of the brain. This change can be identified rather quickly, and may be sustained weeks after the last dose of the drug.
Transgenic mice exhibiting inducible expression of ΔFosB primarily in the nucleus accumbens and dorsal striatum exhibit sensitized behavioural responses to cocaine.[54] They self-administer cocaine at lower doses than control,[55] but have a greater likelihood of relapse when the drug is withheld.[55][56] ΔFosB increases the expression of AMPA receptor subunit GluR2[54] and also decreases expression of dynorphin, thereby enhancing sensitivity to reward.[56]
Dependence and withdrawal
Cocaine dependence is a form of psychological dependence that develops from regular cocaine use and produces a withdrawal state with emotional-motivational deficits upon cessation of cocaine use.
During pregnancy
Cocaine is known to have a number of deleterious effects during pregnancy. Pregnant people who use cocaine have an elevated risk of placental abruption, a condition where the placenta detaches from the uterus and causes bleeding.[57] Due to its vasoconstrictive and hypertensive effects, they are also at risk for hemorrhagic stroke and myocardial infarction. Cocaine is also teratogenic, meaning that it can cause birth defects and fetal malformations. In-utero exposure to cocaine is associated with behavioral abnormalities, cognitive impairment, cardiovascular malformations, intrauterine growth restriction, preterm birth, urinary tract malformations, and cleft lip and palate.[58]
Pharmacology
Pharmacodynamics
The pharmacodynamics of cocaine involve the complex relationships of neurotransmitters (inhibiting monoamine uptake in rats with ratios of about: serotonin:dopamine = 2:3, serotonin:norepinephrine = 2:5).[59][10] The most extensively studied effect of cocaine on the central nervous system is the blockade of the dopamine transporter protein. Dopamine transmitter released during neural signaling is normally recycled via the transporter; i.e., the transporter binds the transmitter and pumps it out of the synaptic cleft back into the presynaptic neuron, where it is taken up into storage vesicles. Cocaine binds tightly at the dopamine transporter forming a complex that blocks the transporter's function. The dopamine transporter can no longer perform its reuptake function, and thus dopamine accumulates in the synaptic cleft. The increased concentration of dopamine in the synapse activates post-synaptic dopamine receptors, which makes the drug rewarding and promotes the compulsive use of cocaine.[60]
Cocaine affects certain serotonin (5-HT) receptors; in particular, it has been shown to antagonize the 5-HT3 receptor, which is a ligand-gated ion channel. The overabundance of 5-HT3 receptors in cocaine conditioned rats display this trait, however the exact effect of 5-HT3 in this process is unclear.[61] The 5-HT2 receptor (particularly the subtypes 5-HT2AR, 5-HT2BR and 5-HT2CR) are involved in the locomotor-activating effects of cocaine.[62]
Cocaine has been demonstrated to bind as to directly stabilize the DAT transporter on the open outward-facing conformation. Further, cocaine binds in such a way as to inhibit a hydrogen bond innate to DAT. Cocaine's binding properties are such that it attaches so this hydrogen bond will not form and is blocked from formation due to the tightly locked orientation of the cocaine molecule. Research studies have suggested that the affinity for the transporter is not what is involved in habituation of the substance so much as the conformation and binding properties to where and how on the transporter the molecule binds.[63]
Sigma receptors are affected by cocaine, as cocaine functions as a sigma ligand agonist.[64] Further specific receptors it has been demonstrated to function on are NMDA and the D1 dopamine receptor.[65]
Cocaine also blocks sodium channels, thereby interfering with the propagation of action potentials;[66] thus, like lignocaine and novocaine, it acts as a local anesthetic. It also functions on the binding sites to the dopamine and serotonin sodium dependent transport area as targets as separate mechanisms from its reuptake of those transporters; unique to its local anesthetic value which makes it in a class of functionality different from both its own derived phenyltropanes analogues which have that removed. In addition to this cocaine has some target binding to the site of the Kappa-opioid receptor as well.[67] Cocaine also causes vasoconstriction, thus reducing bleeding during minor surgical procedures. The locomotor enhancing properties of cocaine may be attributable to its enhancement of dopaminergic transmission from the substantia nigra. Recent research points to an important role of circadian mechanisms[68] and clock genes[69] in behavioral actions of cocaine.
Cocaine can often cause reduced food intake, many chronic users lose their appetite and can experience severe malnutrition and significant weight loss. Cocaine effects, further, are shown to be potentiated for the user when used in conjunction with new surroundings and stimuli, and otherwise novel environs.[70]
Pharmacokinetics
Cocaine has a short half life of 0.7-1.5 hours and is extensively metabolized by cholinesterase enzymes (primarily in the liver and plasma), with only about 1% excreted unchanged in the urine.[9] The metabolism is dominated by hydrolytic ester cleavage, so the eliminated metabolites consist mostly of benzoylecgonine (BE), the major metabolite, and other significant metabolites in lesser amounts such as ecgonine methyl ester (EME) and ecgonine.[9] Further minor metabolites of cocaine include norcocaine, p-hydroxycocaine, m-hydroxycocaine, p-hydroxybenzoylecgonine (pOHBE), and m-hydroxybenzoylecgonine.[71] If consumed with alcohol, cocaine combines with alcohol in the liver to form cocaethylene.[9] Studies have suggested cocaethylene is both more euphoric, and has a higher cardiovascular toxicity than cocaine by itself.[9]
Depending on liver and kidney function, cocaine metabolites are detectable in urine. Benzoylecgonine can be detected in urine within four hours after cocaine intake and remains detectable in concentrations greater than 150 ng/mL typically for up to eight days after cocaine is used. Detection of cocaine metabolites in hair is possible in regular users until the sections of hair grown during use are cut or fall out.[72]
Chemistry
Appearance


Cocaine in its purest form is a white, pearly product. Cocaine appearing in powder form is a salt, typically cocaine hydrochloride. Street cocaine is often adulterated or "cut" with talc, lactose, sucrose, glucose, mannitol, inositol, caffeine, procaine, phencyclidine, phenytoin, lignocaine, strychnine, amphetamine, or heroin.[73]
The color of "crack" cocaine depends upon several factors including the origin of the cocaine used, the method of preparation – with ammonia or baking soda – and the presence of impurities. It will generally range from white to a yellowish cream to a light brown. Its texture will also depend on the adulterants, origin and processing of the powdered cocaine, and the method of converting the base. It ranges from a crumbly texture, sometimes extremely oily, to a hard, almost crystalline nature.
Forms
Salts
Cocaine – a tropane alkaloid – is a weakly alkaline compound, and can therefore combine with acidic compounds to form salts. The hydrochloride (HCl) salt of cocaine is by far the most commonly encountered, although the sulfate (SO42-) and the nitrate (NO3−) salts are occasionally seen. Different salts dissolve to a greater or lesser extent in various solvents – the hydrochloride salt is polar in character and is quite soluble in water.
Base
As the name implies, "freebase" is the base form of cocaine, as opposed to the salt form. It is practically insoluble in water whereas hydrochloride salt is water-soluble.
Smoking freebase cocaine has the additional effect of releasing methylecgonidine into the user's system due to the pyrolysis of the substance (a side effect which insufflating or injecting powder cocaine does not create). Some research suggests that smoking freebase cocaine can be even more cardiotoxic than other routes of administration[74] because of methylecgonidine's effects on lung tissue[75] and liver tissue.[76]
Pure cocaine is prepared by neutralizing its compounding salt with an alkaline solution, which will precipitate to non-polar basic cocaine. It is further refined through aqueous-solvent liquid–liquid extraction.
Crack cocaine


Crack is a lower purity form of free-base cocaine that is usually produced by neutralization of cocaine hydrochloride with a solution of baking soda (sodium bicarbonate, NaHCO3) and water, producing a very hard/brittle, off-white-to-brown colored, amorphous material that contains sodium carbonate, entrapped water, and other by-products as the main impurities.
The "freebase" and "crack" forms of cocaine are usually administered by vaporization of the powdered substance into smoke, which is then inhaled.[77]
The origin of the name "crack" comes from the "crackling" sound (and hence the onomatopoeic moniker "crack") that is produced when the cocaine and its impurities (i.e. water, sodium bicarbonate) are heated past the point of vaporization.[78]
Pure cocaine base/crack can be smoked because it vaporizes smoothly, with little or no decomposition at 98 °C (208 °F),[79] which is below the boiling point of water.
In contrast, cocaine hydrochloride does not vaporize until heated to a much higher temperature (about 197 °C), and considerable decomposition/burning occurs at these high temperatures. This effectively destroys some of the cocaine and yields a sharp, acrid, and foul-tasting smoke.
Smoking or vaporizing cocaine and inhaling it into the lungs produces an almost immediate "high" that can be very powerful (and addicting) quite rapidly – this initial crescendo of stimulation is known as a "rush". While the stimulating effects may last for hours, the euphoric sensation is very brief, prompting the user to smoke more immediately.
Coca leaf infusions
Coca herbal infusion (also referred to as coca tea) is used in coca-leaf producing countries much as any herbal medicinal infusion would elsewhere in the world. The free and legal commercialization of dried coca leaves under the form of filtration bags to be used as "coca tea" has been actively promoted by the governments of Peru and Bolivia for many years as a drink having medicinal powers. In Peru, the National Coca Company, a state-run corporation, sells cocaine-infused teas and other medicinal products and also exports leaves to the U.S. for medicinal use.[80]
Visitors to the city of Cuzco in Peru, and La Paz in Bolivia are greeted with the offering of coca leaf infusions (prepared in teapots with whole coca leaves) purportedly to help the newly arrived traveler overcome the malaise of high altitude sickness. The effects of drinking coca tea are a mild stimulation and mood lift.[81] It does not produce any significant numbing of the mouth nor does it give a rush like snorting cocaine. In order to prevent the demonization of this product, its promoters publicize the unproven concept that much of the effect of the ingestion of coca leaf infusion would come from the secondary alkaloids, as being not only quantitatively different from pure cocaine but also qualitatively different.
It has been promoted as an adjuvant for the treatment of cocaine dependence. In one controversial study, coca leaf infusion was used—in addition to counseling—to treat 23 addicted coca-paste smokers in Lima, Peru. Relapses fell from an average of four times per month before treatment with coca tea to one during the treatment. The duration of abstinence increased from an average of 32 days prior to treatment to 217 days during treatment. These results suggest that the administration of coca leaf infusion plus counseling would be an effective method for preventing relapse during treatment for cocaine addiction.
Importantly, these results also suggest strongly that the primary pharmacologically active metabolite in coca leaf infusions is actually cocaine and not the secondary alkaloids. The cocaine metabolite benzoylecgonine can be detected in the urine of people a few hours after drinking one cup of coca leaf infusion.[82]
Biosynthesis
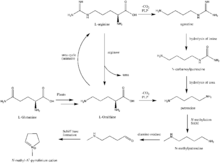
The first synthesis and elucidation of the cocaine molecule was by Richard Willstätter in 1898.[83] Willstätter's synthesis derived cocaine from tropinone. Since then, Robert Robinson and Edward Leete have made significant contributions to the mechanism of the synthesis. (-NO3)
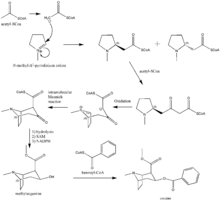
The additional carbon atoms required for the synthesis of cocaine are derived from acetyl-CoA, by addition of two acetyl-CoA units to the N-methyl-Δ1-pyrrolinium cation.[84] The first addition is a Mannich-like reaction with the enolate anion from acetyl-CoA acting as a nucleophile towards the pyrrolinium cation. The second addition occurs through a Claisen condensation. This produces a racemic mixture of the 2-substituted pyrrolidine, with the retention of the thioester from the Claisen condensation. In formation of tropinone from racemic ethyl [2,3-13C2]4(Nmethyl-2-pyrrolidinyl)-3-oxobutanoate there is no preference for either stereoisomer.[85] In the biosynthesis of cocaine, however, only the (S)-enantiomer can cyclize to form the tropane ring system of cocaine. The stereoselectivity of this reaction was further investigated through study of prochiral methylene hydrogen discrimination.[86] This is due to the extra chiral center at C-2.[87] This process occurs through an oxidation, which regenerates the pyrrolinium cation and formation of an enolate anion, and an intramolecular Mannich reaction. The tropane ring system undergoes hydrolysis, SAM-dependent methylation, and reduction via NADPH for the formation of methylecgonine. The benzoyl moiety required for the formation of the cocaine diester is synthesized from phenylalanine via cinnamic acid.[88] Benzoyl-CoA then combines the two units to form cocaine.
N-methyl-pyrrolinium cation
The biosynthesis begins with L-Glutamine, which is derived to L-ornithine in plants. The major contribution of L-ornithine and L-arginine as a precursor to the tropane ring was confirmed by Edward Leete.[89] Ornithine then undergoes a pyridoxal phosphate-dependent decarboxylation to form putrescine. In animals, however, the urea cycle derives putrescine from ornithine. L-ornithine is converted to L-arginine,[90] which is then decarboxylated via PLP to form agmatine. Hydrolysis of the imine derives N-carbamoylputrescine followed with hydrolysis of the urea to form putrescine. The separate pathways of converting ornithine to putrescine in plants and animals have converged. A SAM-dependent N-methylation of putrescine gives the N-methylputrescine product, which then undergoes oxidative deamination by the action of diamine oxidase to yield the aminoaldehyde. Schiff base formation confirms the biosynthesis of the N-methyl-Δ1-pyrrolinium cation.
Robert Robinson's acetonedicarboxylate
The biosynthesis of the tropane alkaloid, however, is still uncertain. Hemscheidt proposes that Robinson's acetonedicarboxylate emerges as a potential intermediate for this reaction.[91] Condensation of N-methylpyrrolinium and acetonedicarboxylate would generate the oxobutyrate. Decarboxylation leads to tropane alkaloid formation.
Reduction of tropinone
The reduction of tropinone is mediated by NADPH-dependent reductase enzymes, which have been characterized in multiple plant species.[92] These plant species all contain two types of the reductase enzymes, tropinone reductase I and tropinone reductase II. TRI produces tropine and TRII produces pseudotropine. Due to differing kinetic and pH/activity characteristics of the enzymes and by the 25-fold higher activity of TRI over TRII, the majority of the tropinone reduction is from TRI to form tropine.[93]
Detection in body fluids
Cocaine and its major metabolites may be quantified in blood, plasma, or urine to monitor for abuse, confirm a diagnosis of poisoning, or assist in the forensic investigation of a traffic or other criminal violation or a sudden death. Most commercial cocaine immunoassay screening tests cross-react appreciably with the major cocaine metabolites, but chromatographic techniques can easily distinguish and separately measure each of these substances. When interpreting the results of a test, it is important to consider the cocaine usage history of the individual, since a chronic user can develop tolerance to doses that would incapacitate a cocaine-naive individual, and the chronic user often has high baseline values of the metabolites in his system. Cautious interpretation of testing results may allow a distinction between passive or active usage, and between smoking versus other routes of administration.[94] In 2011, researchers at John Jay College of Criminal Justice reported that dietary zinc supplements can mask the presence of cocaine and other drugs in urine. Similar claims have been made in web forums on that topic.[95]
Usage
| Substance | Best estimate | Low estimate | High estimate |
|---|---|---|---|
| Amphetamine- type stimulants | 34.16 | 13.42 | 55.24 |
| Cannabis | 192.15 | 165.76 | 234.06 |
| Cocaine | 18.20 | 13.87 | 22.85 |
| Ecstasy | 20.57 | 8.99 | 32.34 |
| Opiates | 19.38 | 13.80 | 26.15 |
| Opioids | 34.26 | 27.01 | 44.54 |
According to a 2016 United Nations report, England and Wales are the countries with the highest rate of cocaine usage (2.4% of adults in the previous year).[97] Other countries where the usage rate meets or exceeds 1.5% are Spain and Scotland (2.2%), the United States (2.1%), Australia (2.1%), Uruguay (1.8%), Brazil (1.75%), Chile (1.73%), the Netherlands (1.5%) and Ireland (1.5%).[97]
Europe
Cocaine is the second most popular illegal recreational drug in Europe (behind cannabis). Since the mid-1990s, overall cocaine usage in Europe has been on the rise, but usage rates and attitudes tend to vary between countries. European countries with the highest usage rates are the United Kingdom, Spain, Italy, and the Republic of Ireland.
Approximately 17 million Europeans (5.1%) have used cocaine at least once and 3.5 million (1.1%) in the last year. About 1.9% (2.3 million) of young adults (15–34 years old) have used cocaine in the last year (latest data available as of 2018).[98]
Usage is particularly prevalent among this demographic: 4% to 7% of males have used cocaine in the last year in Spain, Denmark, Republic of Ireland, Italy, and the United Kingdom. The ratio of male to female users is approximately 3.8:1, but this statistic varies from 1:1 to 13:1 depending on country.[99]
In 2014 London had the highest amount of cocaine in its sewage out of 50 European cities.[100]
United States
Cocaine is the second most popular illegal recreational drug in the United States (behind cannabis)[101] and the U.S. is the world's largest consumer of cocaine.[102] Cocaine is commonly used in middle to upper-class communities and is known as a "rich man's drug". It is also popular amongst college students, as a party drug. A study throughout the entire United States has reported that around 48 percent of people who graduated from high school in 1979 have used cocaine recreationally during some point in their lifetime, compared to approximately 20 percent of students who graduated between the years of 1980 and 1995.[103] Its users span over different ages, races, and professions. In the 1970s and 1980s, the drug became particularly popular in the disco culture as cocaine usage was very common and popular in many discos such as Studio 54.
History
Discovery

For over a thousand years South American indigenous peoples have chewed the leaves of Erythroxylon coca, a plant that contains vital nutrients as well as numerous alkaloids, including cocaine. The coca leaf was, and still is, chewed almost universally by some indigenous communities. The remains of coca leaves have been found with ancient Peruvian mummies, and pottery from the time period depicts humans with bulged cheeks, indicating the presence of something on which they are chewing.[104] There is also evidence that these cultures used a mixture of coca leaves and saliva as an anesthetic for the performance of trepanation.[105]
When the Spanish arrived in South America, most at first ignored aboriginal claims that the leaf gave them strength and energy, and declared the practice of chewing it the work of the Devil.[106] But after discovering that these claims were true, they legalized and taxed the leaf, taking 10% off the value of each crop.[107] In 1569, Spanish botanist Nicolás Monardes described the indigenous peoples' practice of chewing a mixture of tobacco and coca leaves to induce "great contentment":
When they wished to make themselves drunk and out of judgment they chewed a mixture of tobacco and coca leaves which make them go as they were out of their wittes.[108]
In 1609, Padre Blas Valera wrote:
Coca protects the body from many ailments, and our doctors use it in powdered form to reduce the swelling of wounds, to strengthen broken bones, to expel cold from the body or prevent it from entering, and to cure rotten wounds or sores that are full of maggots. And if it does so much for outward ailments, will not its singular virtue have even greater effect in the entrails of those who eat it?[109]
Isolation and naming
Although the stimulant and hunger-suppressant properties of coca had been known for many centuries, the isolation of the cocaine alkaloid was not achieved until 1855. Various European scientists had attempted to isolate cocaine, but none had been successful for two reasons: the knowledge of chemistry required was insufficient at the time, and contemporary conditions of sea-shipping from South America could degrade the cocaine in the plant samples available to European chemists.
The cocaine alkaloid was first isolated by the German chemist Friedrich Gaedcke in 1855. Gaedcke named the alkaloid "erythroxyline", and published a description in the journal Archiv der Pharmazie.[110]
In 1856, Friedrich Wöhler asked Dr. Carl Scherzer, a scientist aboard the Novara (an Austrian frigate sent by Emperor Franz Joseph to circle the globe), to bring him a large amount of coca leaves from South America. In 1859, the ship finished its travels and Wöhler received a trunk full of coca. Wöhler passed on the leaves to Albert Niemann, a Ph.D. student at the University of Göttingen in Germany, who then developed an improved purification process.[111]
Niemann described every step he took to isolate cocaine in his dissertation titled Über eine neue organische Base in den Cocablättern (On a New Organic Base in the Coca Leaves), which was published in 1860—it earned him his Ph.D. and is now in the British Library. He wrote of the alkaloid's "colourless transparent prisms" and said that "Its solutions have an alkaline reaction, a bitter taste, promote the flow of saliva and leave a peculiar numbness, followed by a sense of cold when applied to the tongue." Niemann named the alkaloid "cocaine" from "coca" (from Quechua "cuca") + suffix "ine".[111][112] Because of its use as a local anesthetic, a suffix "-caine" was later extracted and used to form names of synthetic local anesthetics.
The first synthesis and elucidation of the structure of the cocaine molecule was by Richard Willstätter in 1898.[83] It was the first biomimetic synthesis of an organic structure recorded in academic chemical literature.[113][114] The synthesis started from tropinone, a related natural product and took five steps.
Medicalization

.jpg)
With the discovery of this new alkaloid, Western medicine was quick to exploit the possible uses of this plant.
In 1879, Vassili von Anrep, of the University of Würzburg, devised an experiment to demonstrate the analgesic properties of the newly discovered alkaloid. He prepared two separate jars, one containing a cocaine-salt solution, with the other containing merely salt water. He then submerged a frog's legs into the two jars, one leg in the treatment and one in the control solution, and proceeded to stimulate the legs in several different ways. The leg that had been immersed in the cocaine solution reacted very differently from the leg that had been immersed in salt water.[115]
Karl Koller (a close associate of Sigmund Freud, who would write about cocaine later) experimented with cocaine for ophthalmic usage. In an infamous experiment in 1884, he experimented upon himself by applying a cocaine solution to his own eye and then pricking it with pins. His findings were presented to the Heidelberg Ophthalmological Society. Also in 1884, Jellinek demonstrated the effects of cocaine as a respiratory system anesthetic. In 1885, William Halsted demonstrated nerve-block anesthesia,[116] and James Leonard Corning demonstrated peridural anesthesia.[117] 1898 saw Heinrich Quincke use cocaine for spinal anesthesia.
Popularization

In 1859, an Italian doctor, Paolo Mantegazza, returned from Peru, where he had witnessed first-hand the use of coca by the local indigenous peoples. He proceeded to experiment on himself and upon his return to Milan he wrote a paper in which he described the effects. In this paper he declared coca and cocaine (at the time they were assumed to be the same) as being useful medicinally, in the treatment of "a furred tongue in the morning, flatulence, and whitening of the teeth."
A chemist named Angelo Mariani who read Mantegazza's paper became immediately intrigued with coca and its economic potential. In 1863, Mariani started marketing a wine called Vin Mariani, which had been treated with coca leaves, to become cocawine. The ethanol in wine acted as a solvent and extracted the cocaine from the coca leaves, altering the drink's effect. It contained 6 mg cocaine per ounce of wine, but Vin Mariani which was to be exported contained 7.2 mg per ounce, to compete with the higher cocaine content of similar drinks in the United States. A "pinch of coca leaves" was included in John Styth Pemberton's original 1886 recipe for Coca-Cola, though the company began using decocainized leaves in 1906 when the Pure Food and Drug Act was passed.
In 1879 cocaine began to be used to treat morphine addiction. Cocaine was introduced into clinical use as a local anesthetic in Germany in 1884, about the same time as Sigmund Freud published his work Über Coca, in which he wrote that cocaine causes:[119]
Exhilaration and lasting euphoria, which in no way differs from the normal euphoria of the healthy person. You perceive an increase of self-control and possess more vitality and capacity for work. In other words, you are simply normal, and it is soon hard to believe you are under the influence of any drug. Long intensive physical work is performed without any fatigue. This result is enjoyed without any of the unpleasant after-effects that follow exhilaration brought about by alcoholic beverages. No craving for the further use of cocaine appears after the first, or even after repeated taking of the drug.[120]
In 1885 the U.S. manufacturer Parke-Davis sold cocaine in various forms, including cigarettes, powder, and even a cocaine mixture that could be injected directly into the user's veins with the included needle. The company promised that its cocaine products would "supply the place of food, make the coward brave, the silent eloquent and render the sufferer insensitive to pain."
.jpg)
By the late Victorian era, cocaine use had appeared as a vice in literature. For example, it was injected by Arthur Conan Doyle's fictional Sherlock Holmes, generally to offset the boredom he felt when he was not working on a case.
In early 20th-century Memphis, Tennessee, cocaine was sold in neighborhood drugstores on Beale Street, costing five or ten cents for a small boxful. Stevedores along the Mississippi River used the drug as a stimulant, and white employers encouraged its use by black laborers.[121]
In 1909, Ernest Shackleton took "Forced March" brand cocaine tablets to Antarctica, as did Captain Scott a year later on his ill-fated journey to the South Pole.[122]
During the mid-1940s, amidst World War II, cocaine was considered for inclusion as an ingredient of a future generation of 'pep pills' for the German military, code named D-IX.[123]
In modern popular culture references to the drug are prevalent, in it the drug has a glamorous image associated with the rich, famous and powerful with it also making users to "feel rich and beautiful".[124][125][126][127] In addition the pace of modern society − such as in finance − gives many the incentive to make use of the drug.[124]

Modern usage

In many countries, cocaine is a popular recreational drug. In the United States, the development of "crack" cocaine introduced the substance to a generally poorer inner-city market. Use of the powder form has stayed relatively constant, experiencing a new height of use during the late 1990s and early 2000s in the U.S., and has become much more popular in the last few years in the UK.
Cocaine use is prevalent across all socioeconomic strata, including age, demographics, economic, social, political, religious, and livelihood.
The estimated U.S. cocaine market exceeded US$70 billion in street value for the year 2005, exceeding revenues by corporations such as Starbucks.[128][129] There is a tremendous demand for cocaine in the U.S. market, particularly among those who are making incomes affording luxury spending, such as single adults and professionals with discretionary income. Cocaine's status as a club drug shows its immense popularity among the "party crowd".
In 1995 the World Health Organization (WHO) and the United Nations Interregional Crime and Justice Research Institute (UNICRI) announced in a press release the publication of the results of the largest global study on cocaine use ever undertaken. However, a decision by an American representative in the World Health Assembly banned the publication of the study, because it seemed to make a case for the positive uses of cocaine. An excerpt of the report strongly conflicted with accepted paradigms, for example "that occasional cocaine use does not typically lead to severe or even minor physical or social problems." In the sixth meeting of the B committee, the US representative threatened that "If World Health Organization activities relating to drugs failed to reinforce proven drug control approaches, funds for the relevant programs should be curtailed". This led to the decision to discontinue publication. A part of the study was recuperated and published in 2010, including profiles of cocaine use in 20 countries, but are unavailable as of 2015.[130]
In October 2010 it was reported that the use of cocaine in Australia has doubled since monitoring began in 2003.[131]
A problem with illegal cocaine use, especially in the higher volumes used to combat fatigue (rather than increase euphoria) by long-term users, is the risk of ill effects or damage caused by the compounds used in adulteration. Cutting or "stepping on" the drug is commonplace, using compounds which simulate ingestion effects, such as Novocain (procaine) producing temporary anesthaesia, as many users believe a strong numbing effect is the result of strong and/or pure cocaine, ephedrine or similar stimulants that are to produce an increased heart rate. The normal adulterants for profit are inactive sugars, usually mannitol, creatine or glucose, so introducing active adulterants gives the illusion of purity and to 'stretch' or make it so a dealer can sell more product than without the adulterants. The adulterant of sugars allows the dealer to sell the product for a higher price because of the illusion of purity and allows sale of more of the product at that higher price, enabling dealers to significantly increase revenue with little additional cost for the adulterants. A 2007 study by the European Monitoring Centre for Drugs and Drug Addiction showed that the purity levels for street purchased cocaine was often under 5% and on average under 50% pure.[132]
Society and culture
Legal status
The production, distribution, and sale of cocaine products is restricted (and illegal in most contexts) in most countries as regulated by the Single Convention on Narcotic Drugs, and the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances. In the United States the manufacture, importation, possession, and distribution of cocaine are additionally regulated by the 1970 Controlled Substances Act.
Some countries, such as Peru and Bolivia, permit the cultivation of coca leaf for traditional consumption by the local indigenous population, but nevertheless, prohibit the production, sale, and consumption of cocaine.[133] The provisions as to how much a coca farmer can yield annually is protected by laws such as the Bolivian Cato accord.[134] In addition, some parts of Europe, the United States, and Australia allow processed cocaine for medicinal uses only.
Australia
Cocaine is a Schedule 8 prohibited substance in Australia under the Poisons Standard (July 2016).[135] A schedule 8 substance is a controlled Drug – Substances which should be available for use but require restriction of manufacture, supply, distribution, possession and use to reduce abuse, misuse and physical or psychological dependence.[135]
In Western Australia under the Misuse of Drugs Act 1981 4.0g of cocaine is the amount of prohibited drugs determining a court of trial, 2.0g is the amount of cocaine required for the presumption of intention to sell or supply and 28.0g is the amount of cocaine required for purposes of drug trafficking.[136]
United States
The US federal government instituted a national labeling requirement for cocaine and cocaine-containing products through the Pure Food and Drug Act of 1906.[137] The next important federal regulation was the Harrison Narcotics Tax Act of 1914. While this act is often seen as the start of prohibition, the act itself was not actually a prohibition on cocaine, but instead set up a regulatory and licensing regime.[138] The Harrison Act did not recognize addiction as a treatable condition and therefore the therapeutic use of cocaine, heroin or morphine to such individuals was outlawed – leading a 1915 editorial in the journal American Medicine to remark that the addict "is denied the medical care he urgently needs, open, above-board sources from which he formerly obtained his drug supply are closed to him, and he is driven to the underworld where he can get his drug, but of course, surreptitiously and in violation of the law."[139] The Harrison Act left manufacturers of cocaine untouched so long as they met certain purity and labeling standards.[140] Despite that cocaine was typically illegal to sell and legal outlets were rarer, the quantities of legal cocaine produced declined very little.[140] Legal cocaine quantities did not decrease until the Jones–Miller Act of 1922 put serious restrictions on cocaine manufactures.[140]
Interdiction
.jpg)

In 2004, according to the United Nations, 589 tonnes of cocaine were seized globally by law enforcement authorities. Colombia seized 188 t, the United States 166 t, Europe 79 t, Peru 14 t, Bolivia 9 t, and the rest of the world 133 t.[141]
Economics
Because of the drug's potential for addiction and overdose, cocaine is generally treated as a "hard drug", with severe penalties for possession and trafficking. Demand remains high, and consequently, black market cocaine is quite expensive. Unprocessed cocaine, such as coca leaves, are occasionally purchased and sold, but this is exceedingly rare as it is much easier and more profitable to conceal and smuggle it in powdered form. The scale of the market is immense: 770 tonnes times $100 per gram retail = up to $77 billion.
Production
Colombia is as of 2019 the world's largest cocaine producer, with production more than tripling since 2013.[142][143] Three-quarters of the world's annual yield of cocaine has been produced in Colombia, both from cocaine base imported from Peru (primarily the Huallaga Valley) and Bolivia, and from locally grown coca. There was a 28% increase from the amount of potentially harvestable coca plants which were grown in Colombia in 1998. This, combined with crop reductions in Bolivia and Peru, made Colombia the nation with the largest area of coca under cultivation after the mid-1990s. Coca grown for traditional purposes by indigenous communities, a use which is still present and is permitted by Colombian laws, only makes up a small fragment of total coca production, most of which is used for the illegal drug trade.
An interview with a coca farmer published in 2003 described a mode of production by acid-base extraction that has changed little since 1905. Roughly 625 pounds (283 kg) of leaves were harvested per hectare, six times per year. The leaves were dried for half a day, then chopped into small pieces with a string trimmer and sprinkled with a small amount of powdered cement (replacing sodium carbonate from former times). Several hundred pounds of this mixture were soaked in 50 US gallons (190 L) of gasoline for a day, then the gasoline was removed and the leaves were pressed for remaining liquid, after which they could be discarded. Then battery acid (weak sulfuric acid) was used, one bucket per 55 lb (25 kg) of leaves, to create a phase separation in which the cocaine free base in the gasoline was acidified and extracted into a few buckets of "murky-looking smelly liquid". Once powdered caustic soda was added to this, the cocaine precipitated and could be removed by filtration through a cloth. The resulting material, when dried, was termed pasta and sold by the farmer. The 3750 pound yearly harvest of leaves from a hectare produced 6 lb (2.5 kg) of pasta, approximately 40–60% cocaine. Repeated recrystallization from solvents, producing pasta lavada and eventually crystalline cocaine were performed at specialized laboratories after the sale.[144]
Attempts to eradicate coca fields through the use of defoliants have devastated part of the farming economy in some coca growing regions of Colombia, and strains appear to have been developed that are more resistant or immune to their use. Whether these strains are natural mutations or the product of human tampering is unclear. These strains have also shown to be more potent than those previously grown, increasing profits for the drug cartels responsible for the exporting of cocaine. Although production fell temporarily, coca crops rebounded in numerous smaller fields in Colombia, rather than the larger plantations.
The cultivation of coca has become an attractive economic decision for many growers due to the combination of several factors, including the lack of other employment alternatives, the lower profitability of alternative crops in official crop substitution programs, the eradication-related damages to non-drug farms, the spread of new strains of the coca plant due to persistent worldwide demand.
| 2000 | 2001 | 2002 | 2003 | 2004 | |
|---|---|---|---|---|---|
| Net cultivation km2 (sq mi) | 1,875 (724) | 2,218 (856) | 2,007.5 (775.1) | 1,663 (642) | 1,662 (642) |
| Potential pure cocaine production (tonnes) | 770 | 925 | 830 | 680 | 645 |
The latest estimate provided by the U.S. authorities on the annual production of cocaine in Colombia refers to 290 metric tons. As of the end of 2011, the seizure operations of Colombian cocaine carried out in different countries have totaled 351.8 metric tons of cocaine, i.e. 121.3% of Colombia's annual production according to the U.S. Department of State's estimates. [146][147]
Synthesis
Synthetic cocaine would be highly desirable to the illegal drug industry as it would eliminate the high visibility and low reliability of offshore sources and international smuggling, replacing them with clandestine domestic laboratories, as are common for illicit methamphetamine. However, natural cocaine remains the lowest cost and highest quality supply of cocaine. Actual full synthesis of cocaine is rarely done. Formation of inactive stereoisomers (cocaine has 4 chiral centres – 1R, 2R, 3S, and 5S, 2 of them dependent, hence a total potential of 8 possible stereoisomers) plus synthetic by-products limits the yield and purity. Names like "synthetic cocaine" and "new cocaine" have been misapplied to phencyclidine (PCP) and various designer drugs.
Trafficking and distribution

Organized criminal gangs operating on a large scale dominate the cocaine trade. Most cocaine is grown and processed in South America, particularly in Colombia, Bolivia, Peru, and smuggled into the United States and Europe, the United States being the world's largest consumer of cocaine,[102] where it is sold at huge markups; usually in the US at $80–120 for 1 gram, and $250–300 for 3.5 grams (1/8 of an ounce, or an "eight ball").[148]
Caribbean and Mexican routes
As of 2005, cocaine shipments from South America transported through Mexico or Central America were generally moved over land or by air to staging sites in northern Mexico. The cocaine is then broken down into smaller loads for smuggling across the U.S.–Mexico border. The primary cocaine importation points in the United States have been in Arizona, southern California, southern Florida, and Texas. Typically, land vehicles are driven across the U.S.–Mexico border. Sixty-five percent of cocaine enters the United States through Mexico, and the vast majority of the rest enters through Florida.[149] As of 2015, the Sinaloa Cartel is the most active drug cartel involved in smuggling illicit drugs like cocaine into the United States and trafficking them throughout the United States.[150]
Cocaine traffickers from Colombia and Mexico have established a labyrinth of smuggling routes throughout the Caribbean, the Bahama Island chain, and South Florida. They often hire traffickers from Mexico or the Dominican Republic to transport the drug using a variety of smuggling techniques to U.S. markets. These include airdrops of 500 to 700 kg (1,100 to 1,500 lb) in the Bahama Islands or off the coast of Puerto Rico, mid-ocean boat-to-boat transfers of 500 to 2,000 kg (1,100 to 4,400 lb), and the commercial shipment of tonnes of cocaine through the port of Miami.
Chilean route
Another route of cocaine traffic goes through Chile, which is primarily used for cocaine produced in Bolivia since the nearest seaports lie in northern Chile. The arid Bolivia–Chile border is easily crossed by 4×4 vehicles that then head to the seaports of Iquique and Antofagasta. While the price of cocaine is higher in Chile than in Peru and Bolivia, the final destination is usually Europe, especially Spain where drug dealing networks exist among South American immigrants.
Techniques
Cocaine is also carried in small, concealed, kilogram quantities across the border by couriers known as "mules" (or "mulas"), who cross a border either legally, for example, through a port or airport, or illegally elsewhere. The drugs may be strapped to the waist or legs or hidden in bags, or hidden in the body. If the mule gets through without being caught, the gangs will reap most of the profits. If he or she is caught, however, gangs will sever all links and the mule will usually stand trial for trafficking alone.
Bulk cargo ships are also used to smuggle cocaine to staging sites in the western Caribbean–Gulf of Mexico area. These vessels are typically 150–250-foot (50–80 m) coastal freighters that carry an average cocaine load of approximately 2.5 tonnes. Commercial fishing vessels are also used for smuggling operations. In areas with a high volume of recreational traffic, smugglers use the same types of vessels, such as go-fast boats, as those used by the local populations.
Sophisticated drug subs are the latest tool drug runners are using to bring cocaine north from Colombia, it was reported on 20 March 2008. Although the vessels were once viewed as a quirky sideshow in the drug war, they are becoming faster, more seaworthy, and capable of carrying bigger loads of drugs than earlier models, according to those charged with catching them.[151]
Sales to consumers

Cocaine is readily available in all major countries' metropolitan areas. According to the Summer 1998 Pulse Check, published by the U.S. Office of National Drug Control Policy, cocaine use had stabilized across the country, with a few increases reported in San Diego, Bridgeport, Miami, and Boston. In the West, cocaine usage was lower, which was thought to be due to a switch to methamphetamine among some users; methamphetamine is cheaper, three and a half times more powerful, and lasts 12–24 times longer with each dose.[152][153] Nevertheless, the number of cocaine users remain high, with a large concentration among urban youth.
In addition to the amounts previously mentioned, cocaine can be sold in "bill sizes": As of 2007 for example, $10 might purchase a "dime bag", a very small amount (0.1–0.15 g) of cocaine. Twenty dollars might purchase 0.15–0.3 g. However, in lower Texas, it is sold cheaper due to it being easier to receive: a dime for $10 is 0.4 g, a 20 is 0.8–1.0 g and an 8-ball (3.5 g) is sold for $60 to $80, depending on the quality and dealer. These amounts and prices are very popular among young people because they are inexpensive and easily concealed on one's body. Quality and price can vary dramatically depending on supply and demand, and on geographic region.[154]
In 2008, the European Monitoring Centre for Drugs and Drug Addiction reports that the typical retail price of cocaine varied between €50 and €75 per gram in most European countries, although Cyprus, Romania, Sweden and Turkey reported much higher values.[155]
Consumption
World annual cocaine consumption, as of 2000, stood at around 600 tonnes, with the United States consuming around 300 t, 50% of the total, Europe about 150 t, 25% of the total, and the rest of the world the remaining 150 t or 25%.[156] It is estimated that 1.5 million people in the United States used cocaine in 2010 down from 2.4 million in 2006.[9] Conversely, cocaine use appears to be increasing in Europe with the highest prevalences in Spain, the United Kingdom, Italy, and Ireland.[9]
The 2010 UN World Drug Report concluded that "it appears that the North American cocaine market has declined in value from US$47 billion in 1998 to US$38 billion in 2008. Between 2006 and 2008, the value of the market remained basically stable".[157]
Research
In 2005, researchers proposed the use of cocaine in conjunction with phenylephrine administered in the form of an eye drop as a diagnostic test for Parkinson's disease.[158]
See also
- Black cocaine
- Coca alkaloids
- Coca eradication
- Cocaine and amphetamine regulated transcript
- Cocaine Anonymous
- Cocaine dependence
- Cocaine paste
- Crack cocaine § Crack lung
- Crack epidemic
- Illegal drug trade in Latin America
- Legal status of cocaine
- List of cocaine analogues
- Modafinil
- Pre-Columbian trans-oceanic evidence for cocaine in ancient Egypt
- Prenatal cocaine exposure
- Route 36, cocaine bar in Bolivia
- TA-CD
- Ypadu
References
- Nordegren, Thomas (2002). The A-Z Encyclopedia of Alcohol and Drug Abuse. Universal-Publishers. p. 461. ISBN 9781581124040.
- Ghodse H (2010). Ghodse's Drugs and Addictive Behaviour: A Guide to Treatment (4 ed.). Cambridge University Press. p. 91. ISBN 978-1-139-48567-8. Archived from the original on 10 September 2017.
- Introduction to Pharmacology Third Edition. Abingdon: CRC Press. 2007. pp. 222–223. ISBN 978-1-4200-4742-4. Archived from the original on 10 September 2017.
- "DEA / Drug Scheduling". www.dea.gov. Archived from the original on 9 August 2017. Retrieved 7 August 2017.
- Fattinger K, Benowitz NL, Jones RT, Verotta D (July 2000). "Nasal mucosal versus gastrointestinal absorption of nasally administered cocaine". European Journal of Clinical Pharmacology. 56 (4): 305–10. doi:10.1007/s002280000147. PMID 10954344.
- Barnett G, Hawks R, Resnick R (1981). "Cocaine pharmacokinetics in humans". Journal of Ethnopharmacology. 3 (2–3): 353–66. doi:10.1016/0378-8741(81)90063-5. PMID 7242115.
- Jeffcoat AR, Perez-Reyes M, Hill JM, Sadler BM, Cook CE (1989). "Cocaine disposition in humans after intravenous injection, nasal insufflation (snorting), or smoking". Drug Metabolism and Disposition. 17 (2): 153–9. PMID 2565204.
- Wilkinson P, Van Dyke C, Jatlow P, Barash P, Byck R (March 1980). "Intranasal and oral cocaine kinetics". Clinical Pharmacology and Therapeutics. 27 (3): 386–94. doi:10.1038/clpt.1980.52. PMID 7357795.
- Zimmerman JL (October 2012). "Cocaine intoxication". Critical Care Clinics. 28 (4): 517–26. doi:10.1016/j.ccc.2012.07.003. PMID 22998988.
- Pomara C, Cassano T, D'Errico S, Bello S, Romano AD, Riezzo I, Serviddio G (2012). "Data available on the extent of cocaine use and dependence: biochemistry, pharmacologic effects and global burden of disease of cocaine abusers". Current Medicinal Chemistry. 19 (33): 5647–57. doi:10.2174/092986712803988811. PMID 22856655.
- Connors NJ, Hoffman RS (November 2013). "Experimental treatments for cocaine toxicity: a difficult transition to the bedside". The Journal of Pharmacology and Experimental Therapeutics. 347 (2): 251–7. doi:10.1124/jpet.113.206383. PMID 23978563.
- Harper SJ, Jones NS (October 2006). "Cocaine: what role does it have in current ENT practice? A review of the current literature". The Journal of Laryngology and Otology. 120 (10): 808–11. doi:10.1017/s0022215106001459. PMID 16848922.
- Sordo L, Indave BI, Barrio G, Degenhardt L, de la Fuente L, Bravo MJ (September 2014). "Cocaine use and risk of stroke: a systematic review". Drug and Alcohol Dependence. 142: 1–13. doi:10.1016/j.drugalcdep.2014.06.041. PMID 25066468.
- Goldstein RA, DesLauriers C, Burda AM (January 2009). "Cocaine: history, social implications, and toxicity--a review". Disease-A-Month. 55 (1): 6–38. doi:10.1016/j.disamonth.2008.10.002. PMID 19081448.
- Sharma HS, Muresanu D, Sharma A, Patnaik R (2009). "Cocaine-induced breakdown of the blood-brain barrier and neurotoxicity". International Review of Neurobiology. 88: 297–334. doi:10.1016/S0074-7742(09)88011-2. ISBN 978-0-12-374504-0. PMID 19897082.
- Karch SB (2009). Karch's pathology of drug abuse (4 ed.). Boca Raton: CRC Press. p. 70. ISBN 978-0-8493-7881-2. Archived from the original on 10 September 2017.
- Narcotic Drugs 2014 (PDF). International Narcotics Control Board. 2015. p. 21. ISBN 9789210481571. Archived (PDF) from the original on 2 June 2015.
- Karila L, Zarmdini R, Petit A, Lafaye G, Lowenstein W, Reynaud M (January 2014). "[Cocaine addiction: current data for the clinician]". Presse Médicale. 43 (1): 9–17. doi:10.1016/j.lpm.2013.01.069. PMID 23727012.
- GBD 2013 Mortality Causes of Death Collaborators (January 2015). "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013". Lancet. 385 (9963): 117–71. doi:10.1016/S0140-6736(14)61682-2. PMC 4340604. PMID 25530442.
- Room R, Reuter P (January 2012). "How well do international drug conventions protect public health?". Lancet. 379 (9810): 84–91. doi:10.1016/s0140-6736(11)61423-2. PMID 22225673.
The international treaties have also constrained national policy experimentation because they require nation states to criminalise drug use
- Dwyer C, Sowerby L, Rotenberg BW (August 2016). "Is cocaine a safe topical agent for use during endoscopic sinus surgery?". The Laryngoscope (Review). 126 (8): 1721–3. doi:10.1002/lary.25836. PMID 27075241.
- World Health Organization (2004). Neuroscience of psychoactive substance use and dependence. p. 89. ISBN 9789241562355. Archived from the original on 30 April 2016.
- World Health Organization (2007). International medical guide for ships. p. 242. ISBN 9789241547208. Archived from the original on 30 April 2016.
- Sordo L, Indave BI, Barrio G, Degenhardt L, de la Fuente L, Bravo MJ (September 2014). "Cocaine use and risk of stroke: a systematic review". Drug and Alcohol Dependence (Systematic Review). 142: 1–13. doi:10.1016/j.drugalcdep.2014.06.041. PMID 25066468.
- Donroe JH, Tetrault JM (July 2017). "Substance Use, Intoxication, and Withdrawal in the Critical Care Setting". Critical Care Clinics (Review). 33 (3): 543–558. doi:10.1016/j.ccc.2017.03.003. PMID 28601134.
- Barnett G, Hawks R, Resnick R (1981). "Cocaine pharmacokinetics in humans". Journal of Ethnopharmacology. 3 (2–3): 353–66. doi:10.1016/0378-8741(81)90063-5. PMID 7242115.; Jones, supra note 19; Wilkinson et al., Van Dyke et al.
- Luks AM, McIntosh SE, Grissom CK, Auerbach PS, Rodway GW, Schoene RB, Zafren K, Hackett PH (June 2010). "Wilderness Medical Society consensus guidelines for the prevention and treatment of acute altitude illness" (PDF). Wilderness & Environmental Medicine. 21 (2): 146–55. doi:10.1016/j.wem.2010.03.002. PMID 20591379. Archived from the original (PDF) on 15 October 2012. (mirror: )
- Siegel RK, Elsohly MA, Plowman T, Rury PM, Jones RT (January 1986). "Cocaine in herbal tea". JAMA. 255 (1): 40. doi:10.1001/jama.255.1.40. PMID 3940302.
- "DrugFacts: Cocaine". National Institute on Drug Abuse. April 2013. Archived from the original on 11 July 2015. Retrieved 11 July 2015.
- Volkow ND, Wang GJ, Fischman MW, Foltin R, Fowler JS, Franceschi D, Franceschi M, Logan J, Gatley SJ, Wong C, Ding YS, Hitzemann R, Pappas N (August 2000). "Effects of route of administration on cocaine induced dopamine transporter blockade in the human brain". Life Sciences. 67 (12): 1507–15. doi:10.1016/S0024-3205(00)00731-1. PMID 10983846.
- "Cocaine terminology". Archived from the original on 9 July 2007.
- Bonkovsky HL, Mehta S (February 2001). "Hepatitis C: a review and update". Journal of the American Academy of Dermatology. 44 (2): 159–82. doi:10.1067/mjd.2001.109311. PMID 11174373.
- Dimitrijevic N, Dzitoyeva S, Manev H (August 2004). "An automated assay of the behavioral effects of cocaine injections in adult Drosophila". Journal of Neuroscience Methods. 137 (2): 181–4. doi:10.1016/j.jneumeth.2004.02.023. PMID 15262059.
- "Appendix B: Production of Cocaine Hydrochloride and Cocaine Base". US Justice Dep. Archived from the original on 30 November 2009.
- Garcia RC, Torres LH, Balestrin NT, Andrioli TC, Flório JC, de Oliveira CD, da Costa JL, Yonamine M, Sandoval MR, Camarini R, Marcourakis T (February 2017). "Anhydroecgonine methyl ester, a cocaine pyrolysis product, may contribute to cocaine behavioral sensitization". Toxicology. 376: 44–50. doi:10.1016/j.tox.2016.04.009. PMID 27129946.
- Reist M (16 January 2005). "A rose by another name: crack pipe". Lincoln Journal Star. Archived from the original on 26 July 2010. Retrieved 21 August 2009.
- "Cocaine | FRANK". www.talktofrank.com. Archived from the original on 2 February 2017. Retrieved 23 January 2017.
- Zhao W (2008). Mechanisms Mediating Sex Differences in the Effects of Cocaine. ProQuest. p. 3. ISBN 978-0-549-99458-9. Archived from the original on 4 April 2014. Retrieved 25 September 2012.
- Overdose Death Rates Archived 28 November 2015 at the Wayback Machine. By National Institute on Drug Abuse (NIDA).
- Nutt D, King LA, Saulsbury W, Blakemore C (March 2007). "Development of a rational scale to assess the harm of drugs of potential misuse". Lancet. 369 (9566): 1047–53. doi:10.1016/S0140-6736(07)60464-4. PMID 17382831.
- D'haenen H, den Boer JA, Willner P, eds. (2002). Biological Psychiatry. 2 (2 ed.). Wiley. p. 528. ISBN 978-0-471-49198-9.
- Lowinson JH, Ruiz P, Millman RB (2004). Substance abuse: a comprehensive textbook (4th ed.). Lippincott Williams & Wilkins. p. 204. ISBN 978-0-7817-3474-5. Archived from the original on 4 April 2014. Retrieved 5 January 2014.
- Baigent M (2003). "Physical complications of substance abuse: what the psychiatrist needs to know". Curr Opin Psychiatry. 16 (3): 291–296. doi:10.1097/00001504-200305000-00004.
- Pagliaro L, Pagliaro AM (2004). Pagliaros' Comprehensive Guide to Drugs and Substances of Abuse. Washington, D.C.: American Pharmacists Association. ISBN 978-1-58212-066-9.
- "More bad news for cocaine users: Drug can triple risk of aneurysm". Scienceblog.com. 1999. Archived from the original on 11 October 2007. Retrieved 10 July 2007.
- Trozak DJ, Gould WM (March 1984). "Cocaine abuse and connective tissue disease". Journal of the American Academy of Dermatology. 10 (3): 525. doi:10.1016/S0190-9622(84)80112-7. PMID 6725666.
- Peces R, Navascués RA, Baltar J, Seco M, Alvarez J (1999). "Antiglomerular basement membrane antibody-mediated glomerulonephritis after intranasal cocaine use". Nephron. 81 (4): 434–8. doi:10.1159/000045328. PMID 10095180.
- Moore PM, Richardson B (July 1998). "Neurology of the vasculitides and connective tissue diseases". Journal of Neurology, Neurosurgery, and Psychiatry. 65 (1): 10–22. doi:10.1136/jnnp.65.1.10. PMC 2170162. PMID 9667555.
- Jaffe JA, Kimmel PL (July 2006). "Chronic nephropathies of cocaine and heroin abuse: a critical review". Clinical Journal of the American Society of Nephrology. American Society of Nephrology. 1 (4): 655–67. doi:10.2215/CJN.00300106. PMID 17699270.
- van der Woude FJ (March 2000). "Cocaine use and kidney damage". Nephrology, Dialysis, Transplantation. Oxford University Press. 15 (3): 299–301. doi:10.1093/ndt/15.3.299. PMID 10692510. Archived from the original on 20 April 2011.
- Havakuk O, Rezkalla SH, Kloner RA (July 2017). "The Cardiovascular Effects of Cocaine". Journal of the American College of Cardiology (Review). 70 (1): 101–113. doi:10.1016/j.jacc.2017.05.014. PMID 28662796.
- Hope BT (May 1998). "Cocaine and the AP-1 transcription factor complex". Annals of the New York Academy of Sciences. 844 (1): 1–6. Bibcode:1998NYASA.844....1H. doi:10.1111/j.1749-6632.1998.tb08216.x. PMID 9668659.
- Kelz MB, Chen J, Carlezon WA, Whisler K, Gilden L, Beckmann AM, Steffen C, Zhang YJ, Marotti L, Self DW, Tkatch T, Baranauskas G, Surmeier DJ, Neve RL, Duman RS, Picciotto MR, Nestler EJ (September 1999). "Expression of the transcription factor deltaFosB in the brain controls sensitivity to cocaine". Nature. 401 (6750): 272–6. doi:10.1038/45790. PMID 10499584.
- Colby CR, Whisler K, Steffen C, Nestler EJ, Self DW (March 2003). "Striatal cell type-specific overexpression of DeltaFosB enhances incentive for cocaine". The Journal of Neuroscience. 23 (6): 2488–93. doi:10.1523/JNEUROSCI.23-06-02488.2003. PMID 12657709.
- Nestler EJ, Barrot M, Self DW (September 2001). "DeltaFosB: a sustained molecular switch for addiction". Proceedings of the National Academy of Sciences of the United States of America. 98 (20): 11042–6. doi:10.1073/pnas.191352698. PMC 58680. PMID 11572966.
- Hoffman BL, Schorge JO, Bradshaw KD, Halvorson LM, Schaffer JI, Corton MM (22 April 2016). Williams Gynecology, Third Edition. McGraw Hill Professional. ISBN 9780071849098.
- Leveno KJ, Spong CY, Dashe JS, Casey BM, Hoffman BL, Cunningham FG, Bloom SL (12 April 2018). Williams Obstetrics (25th ed.). McGraw-Hill Education. ISBN 9781259644320.
- Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI, Partilla JS (January 2001). "Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin". Synapse. 39 (1): 32–41. doi:10.1002/1098-2396(20010101)39:1<32::AID-SYN5>3.0.CO;2-3. PMID 11071707. (Table V. on page 37)
- Hummel, M; Unterwald, EM (April 2002). "D1 dopamine receptor: a putative neurochemical and behavioral link to cocaine action". Journal of Cellular Physiology. 191 (1): 17–27. doi:10.1002/jcp.10078. PMID 11920678.
- Carta M, Allan AM, Partridge LD, Valenzuela CF (January 2003). "Cocaine inhibits 5-HT3 receptor function in neurons from transgenic mice overexpressing the receptor". European Journal of Pharmacology. 459 (2–3): 167–9. doi:10.1016/S0014-2999(02)02867-4. PMID 12524142.
- Filip M, Bubar MJ, Cunningham KA (September 2004). "Contribution of serotonin (5-hydroxytryptamine; 5-HT) 5-HT2 receptor subtypes to the hyperlocomotor effects of cocaine: acute and chronic pharmacological analyses". The Journal of Pharmacology and Experimental Therapeutics. 310 (3): 1246–54. doi:10.1124/jpet.104.068841. PMID 15131246.
- Beuming T, Kniazeff J, Bergmann ML, Shi L, Gracia L, Raniszewska K, Newman AH, Javitch JA, Weinstein H, Gether U, Loland CJ (July 2008). "The binding sites for cocaine and dopamine in the dopamine transporter overlap". Nature Neuroscience. 11 (7): 780–9. doi:10.1038/nn.2146. PMC 2692229. PMID 18568020.
- "Sigma Receptors Play Role In Cocaine-induced Suppression Of Immune System". Sciencedaily.com. 6 May 2003. Archived from the original on 12 January 2011. Retrieved 9 March 2010.
- Lluch J, Rodríguez-Arias M, Aguilar MA, Miñarro J (November 2005). "Role of dopamine and glutamate receptors in cocaine-induced social effects in isolated and grouped male OF1 mice". Pharmacology Biochemistry and Behavior. 82 (3): 478–87. doi:10.1016/j.pbb.2005.10.003. PMID 16313950.
- Knuepfer, MM (March 2003). "Cardiovascular disorders associated with cocaine use: myths and truths". Pharmacology & Therapeutics. 97 (3): 181–222. doi:10.1016/S0163-7258(02)00329-7. PMID 12576134.
- "Drugbank website "drug card", "(DB00907)" for Cocaine: Giving ten targets of the molecule in vivo, including dopamine/serotonin sodium channel affinity & K-opioid affinity". Drugbank.ca. Archived from the original on 20 February 2010. Retrieved 9 March 2010.
- Uz T, Akhisaroglu M, Ahmed R, Manev H (December 2003). "The pineal gland is critical for circadian Period1 expression in the striatum and for circadian cocaine sensitization in mice". Neuropsychopharmacology. 28 (12): 2117–23. doi:10.1038/sj.npp.1300254. PMID 12865893.
- McClung CA, Sidiropoulou K, Vitaterna M, Takahashi JS, White FJ, Cooper DC, Nestler EJ (June 2005). "Regulation of dopaminergic transmission and cocaine reward by the Clock gene". Proceedings of the National Academy of Sciences of the United States of America. 102 (26): 9377–81. Bibcode:2005PNAS..102.9377M. doi:10.1073/pnas.0503584102. PMC 1166621. PMID 15967985.
- Carey RJ, Damianopoulos EN, Shanahan AB (January 2008). "Cocaine effects on behavioral responding to a novel object placed in a familiar environment". Pharmacology Biochemistry and Behavior. 88 (3): 265–71. doi:10.1016/j.pbb.2007.08.010. PMID 17897705.
- Kolbrich EA, Barnes AJ, Gorelick DA, Boyd SJ, Cone EJ, Huestis MA (October 2006). "Major and minor metabolites of cocaine in human plasma following controlled subcutaneous cocaine administration". Journal of Analytical Toxicology. 30 (8): 501–10. doi:10.1093/jat/30.8.501. PMID 17132243. Archived from the original on 18 July 2012.
- Czykanski, Marek (30 December 2015). "Cocaine Metabolites in Hair". Chemistry Views. Retrieved 22 July 2019.
- Pillay VV (2013), Modern Medical Toxicology (4th ed.), Jaypee, pp. 553–554, ISBN 978-93-5025-965-8
- Scheidweiler KB, Plessinger MA, Shojaie J, Wood RW, Kwong TC (December 2003). "Pharmacokinetics and pharmacodynamics of methylecgonidine, a crack cocaine pyrolyzate". The Journal of Pharmacology and Experimental Therapeutics. 307 (3): 1179–87. doi:10.1124/jpet.103.055434. PMID 14561847.
- Yang Y, Ke Q, Cai J, Xiao YF, Morgan JP (January 2001). "Evidence for cocaine and methylecgonidine stimulation of M(2) muscarinic receptors in cultured human embryonic lung cells". British Journal of Pharmacology. 132 (2): 451–60. doi:10.1038/sj.bjp.0703819. PMC 1572570. PMID 11159694.
- Fandiño AS, Toennes SW, Kauert GF (December 2002). "Studies on hydrolytic and oxidative metabolic pathways of anhydroecgonine methyl ester (methylecgonidine) using microsomal preparations from rat organs". Chemical Research in Toxicology. 15 (12): 1543–8. doi:10.1021/tx0255828. PMID 12482236.
- "Substances – Cocaine". The Steinhardt School of Culture, Education, and Human Development. Archived from the original on 2 May 2009. Retrieved 1 August 2009.
- George N (1998). Hip Hop America. Viking Penguin. p. 40.
- Ries RK, Miller SC, Fiellin DA (2009). Principles of addiction medicine. Lippincott Williams & Wilkins. p. 137. ISBN 978-0-7817-7477-2. Archived from the original on 4 April 2014. Retrieved 5 January 2014.
- https://www.businessinsider.com/britain-is-the-worlds-biggest-exporter-of-legal-cocaine-and-heroin-2018-4
- https://www.drugs.com/illicit/cocaine.html
- "Coca tea or mate de coca – the holy coca leaf infusion". inkanat.com. Archived from the original on 20 December 2015. Retrieved 17 December 2015.
- Humphrey AJ, O'Hagan D (October 2001). "Tropane alkaloid biosynthesis. A century old problem unresolved". Natural Product Reports. 18 (5): 494–502. doi:10.1039/b001713m. PMID 11699882.
- Dewick PM (2009). Medicinal Natural Products. Chicester: Wiley-Blackwell. ISBN 978-0-470-74276-1.
- Robins RJ, Abraham TE, Parr AJ, Eagles J, Walton NJ (1997). "The Biosynthesis of Tropane Alkaloids in Datura stramonium: The Identity of the Intermediates between N-Methylpyrrolinium Salt and Tropinone". J. Am. Chem. Soc. 119 (45): 10929–10934. doi:10.1021/ja964461p.
- Hoye TR, Bjorklund JA, Koltun DO, Renner MK (January 2000). "N-methylputrescine oxidation during cocaine biosynthesis: study of prochiral methylene hydrogen discrimination using the remote isotope method". Organic Letters. 2 (1): 3–5. doi:10.1021/ol990940s. PMID 10814231.
- Leete E, Bjorklund JA, Couladis MM, Kim SH (1991). "Late intermediates in the biosynthesis of cocaine: 4-(1-methyl-2-pyrrolidinyl)-3-oxobutanoate and methyl ecgonine". J. Am. Chem. Soc. 113 (24): 9286–9292. doi:10.1021/ja00024a039.
- Leete E, Bjorklund JA, Kim SH (1988). "The biosynthesis of the benzoyl moiety of cocaine". Phytochemistry. 27 (8): 2553–2556. doi:10.1016/0031-9422(88)87026-2.
- Leete E, Marion L, Spenser ID (October 1954). "Biogenesis of hyoscyamine". Nature. 174 (4431): 650–1. Bibcode:1954Natur.174..650L. doi:10.1038/174650a0. PMID 13203600.
- Robins RJ, Waltons NJ, Hamill JD, Parr AJ, Rhodes MJ (October 1991). "Strategies for the genetic manipulation of alkaloid-producing pathways in plants". Planta Medica. 57 (7 Suppl): S27–35. doi:10.1055/s-2006-960226. PMID 17226220.
- Hemscheidt T, Vederas JC (2000). Leeper FJ, Vederas JC (eds.). "Tropane and Related Alkaloids". Top. Curr. Chem. Topics in Current Chemistry. 209: 175. doi:10.1007/3-540-48146-X. ISBN 978-3-540-66573-1.
- Portsteffen A, Draeger B, Nahrstedt A (1992). "Two tropinone reducing enzymes from Datura stramonium transformed root cultures". Phytochemistry. 31 (4): 1135–1138. doi:10.1016/0031-9422(92)80247-C.
- Boswell HD, Dräger B, McLauchlan WR, Portsteffen A, Robins DJ, Robins RJ, Walton NJ (November 1999). "Specificities of the enzymes of N-alkyltropane biosynthesis in Brugmansia and Datura". Phytochemistry. 52 (5): 871–8. doi:10.1016/S0031-9422(99)00293-9. PMID 10626376.
- R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 9th edition, Biomedical Publications, Seal Beach, California, 2011, pp. 390–394.
- Venkatratnam A, Lents NH (July 2011). "Zinc reduces the detection of cocaine, methamphetamine, and THC by ELISA urine testing". Journal of Analytical Toxicology. 35 (6): 333–40. doi:10.1093/anatox/35.6.333. PMID 21740689. Archived from the original on 18 May 2012.
- "Annual prevalence of use of drugs, by region and globally, 2016". World Drug Report 2018. United Nations Office on Drugs and Crime. 2018. Retrieved 7 July 2018.
- "World Drug Report 2016 (interactive map)". United Nations Office on Drugs and Crime. 2016. Archived from the original on 9 March 2018.
- "Statistical Bulletin 2018 — prevalence of drug use | www.emcdda.europa.eu". www.emcdda.europa.eu. Retrieved 5 February 2019.
- The State of the Drugs Problem in Europe 2008 (PDF). Luxembourg: European Monitoring Centre for Drugs and Drug Addiction. 2008. pp. 58–62. Archived (PDF) from the original on 25 April 2013. Retrieved 31 December 2013.
- Casciani D (4 June 2015). "Cocaine in sewage: London tops league table". BBC news. Archived from the original on 4 June 2015. Retrieved 4 June 2015.
- "Cocaine & Crack". Erowid.org. Archived from the original on 6 October 2007. Retrieved 10 July 2007.
- "Field Listing – Illicit drugs (by country)". Cia.gov. Archived from the original on 29 December 2010. Retrieved 15 January 2011.
- "McGraw-Hill Connect". connect.mcgraw-hill.com.
- Altman AJ, Albert DM, Fournier GA (1985). "Cocaine's use in ophthalmology: our 100-year heritage". Survey of Ophthalmology. 29 (4): 300–6. doi:10.1016/0039-6257(85)90153-5. PMID 3885453.
- Gay GR, Inaba DS, Sheppard CW, Newmeyer JA (1975). "Cocaine: history, epidemiology, human pharmacology, and treatment. a perspective on a new debut for an old girl". Clinical Toxicology. 8 (2): 149–78. doi:10.3109/15563657508988061. PMID 1097168.
- Baselmans J (6 December 2016). Drugs. Lulu.com. p. 55. ISBN 978-1-326-84325-0. Archived from the original on 10 September 2017.
- "Drug that spans the ages: The history of cocaine". London: The Independent (UK). 2006. Archived from the original on 28 February 2010. Retrieved 30 April 2010.
- Monardes N, Frampton J (1925). Joyfull Newes out of the Newe Founde Worlde. New York: Alfred Knopf.
- "InterAndean Institute of Coca Sciences". www.cienciadelacoca.org. Archived from the original on 30 December 2016.
- Gaedcke F (1855). "Ueber das Erythroxylin, dargestellt aus den Blättern des in Südamerika cultivirten Strauches Erythroxylon Coca". Archiv der Pharmazie. 132 (2): 141–150. doi:10.1002/ardp.18551320208.
- Niemann A (1860). "Ueber eine neue organische Base in den Cocablättern". Archiv der Pharmazie. 153 (2): 129–256. doi:10.1002/ardp.18601530202.
- Harper, Douglas. "Cocaine". Online Etymology Dictionary.
- Singh S (March 2000). "Chemistry, design, and structure-activity relationship of cocaine antagonists" (PDF). Chemical Reviews. 100 (3): 925–1024. doi:10.1021/cr9700538. PMID 11749256. Archived (PDF) from the original on 4 March 2016.
Page 970 (46th page of article) first, ninth, and tenth lines
- (a) Willstatter R (1903). "Synthese der Ecgoninsäure" [Synthesis of Tropine]. Liebigs Ann. (in German). 326 (1–2): 23. doi:10.1002/jlac.19033260105. (b) Robinson RJ (1917). "LXIII. A synthesis of tropinone". J. Chem. Soc., Trans. 111: 762–768. doi:10.1039/CT9171100762. (c) Schopf C, Lehman G (1935). "Die Synthese des Tropinons, Pseudopelletierins, Lobelanins und verwandter Alkaloide unter physiologischen Bedingungen" [The synthesis of tropinone, pseudopelletierin, lobelanin and related alkaloids under physiological conditions]. Liebigs Ann. (in German). 518: 1–37. doi:10.1002/jlac.19355180102.
- Yentis SM, Vlassakov KV (March 1999). "Vassily von Anrep, forgotten pioneer of regional anesthesia". Anesthesiology. 90 (3): 890–5. doi:10.1097/00000542-199903000-00033. PMID 10078692.
- Halsted W (1885). "Practical comments on the use and abuse of cocaine". New York Medical Journal. 42: 294–295.
- Corning JL (1885). "An experimental study". New York Medical Journal. 42: 483.
- "Experience Vin Mariani today | Grupo Mariani S.A". Cocanaturally.com. Archived from the original on 8 February 2011. Retrieved 15 January 2011.
- "How a Young Sigmund Freud Researched & Got Addicted to Cocaine, the New "Miracle Drug," in 1894". Open Culture. Archived from the original on 7 March 2017.
- "Sigmund Freud and Cocaine". cocaine.org. Archived from the original on 19 January 2017.
- Barlow W (1989). Looking Up At Down: The Emergence of Blues Culture. Philadelphia: Temple University Press. p. 207. ISBN 978-0-87722-583-6.
- Streatfeild D (2003). Cocaine: An Unauthorized Biography. Picador. ISBN 978-0-312-42226-4.
- "Jeevan Vasagar: cocaine-based "wonder drug" tested on concentration camp inmates". Amphetamines.com. 19 November 2002. Archived from the original on 27 February 2011. Retrieved 15 January 2011.
- Ryzik, Melena (10 June 2007). "Cocaine: Hidden in Plain Sight". The New York Times. Archived from the original on 11 August 2017. Retrieved 18 May 2017.
- "The Buyers – A Social History Of America's Most Popular Drugs | Drug Wars | FRONTLINE | PBS". PBS. Archived from the original on 14 May 2017. Retrieved 18 May 2017.
- Brisbane FL, Womble M (1985). Treatment of Black Alcoholics. Psychology Press. ISBN 978-0-86656-403-8. Archived from the original on 10 September 2017. Retrieved 18 May 2017.
- Waldorf D, Reinarman C, Murphy S (June 1992). Cocaine Changes: The Experience of Using and Quitting. Temple University Press. ISBN 978-1-56639-013-2. Archived from the original on 10 September 2017. Retrieved 18 May 2017.
- "Apple Sanity – Fetish – Blow: War on Drugs VS. Cocaine". Applesanity.com. 17 June 2008. Archived from the original on 17 June 2008. Retrieved 13 November 2011.
- "Cocaine Market". Havocscope.com. Archived from the original on 11 November 2012. Retrieved 9 March 2010.
- WHO/UNICRI (4 February 2010). "The WHO Cocaine Project". Transnational Institute. Archived from the original on 9 August 2012. Retrieved 8 June 2012.
- "Cocaine use doubles in a decade". Sydney Morning Herald. 15 October 2010. Archived from the original on 18 October 2010. Retrieved 19 October 2010.
- EMCDDA (2007). "EMCDDA Retail Cocaine Purity Study". Archived from the original on 1 January 2014. Retrieved 31 December 2013.
- Franklin J (18 August 2009). "The world's first cocaine bar". The Guardian. ISSN 0261-3077. Archived from the original on 12 January 2017. Retrieved 23 December 2016.
- Grisaffi, Thomas. "The Cato Accord: Bolivia's Humane and Effective Approach to Controlling Coca Cultivation" (PDF). ain-bolivia.org. Archived (PDF) from the original on 3 May 2016. Retrieved 12 January 2017.
- Poisons Standard July 2016 Comlaw.gov.au Archived 19 January 2016 at the Wayback Machine
- Misuse of Drugs Act 1981 (2015) Slp.wa.gov.au Archived 22 December 2015 at the Wayback Machine
- (Gootenberg 1999, p. 37)
- (Madge 2001, p. 106)
- "Narcotic drug addiction". American Medicine. American-Medicine Publishing Company: 799. November 1915. Archived from the original on 9 May 2018. Retrieved 29 April 2018.
- (Gootenberg 1999, p. 40)
- "Cocaine: Seizures, 1998–2003" (PDF). World Drug Report 2006. 2. New York: United Nations. 2006. Archived (PDF) from the original on 14 June 2007.
- Colombia Archived 13 May 2009 at the Wayback Machine. CIA World Factbook
- Peru Overtakes Colombia as Top Cocaine Producer. NBC News (31 July 2012) Archived 4 March 2016 at the Wayback Machine
- Streatfeild D (2003). Cocaine: An Unauthorized Biography. Macmillan. ISBN 978-0-312-42226-4. Archived from the original on 15 January 2014. Retrieved 5 January 2014.
- NDIC (2006). "National Drug Threat Assessment 2006". Archived from the original on 11 November 2010. Cite journal requires
|journal=(help) - "Cocaine Seized Worldwide Highest Ever in 2011". Flare Network (Flarenetwork.org). 18 January 2012. Archived from the original on 6 January 2014. Retrieved 5 January 2014.
- "Colombia". State.gov. Retrieved 26 March 2013.
- "How Much Is a Gram of Coke?". New Health Advisor. 3 September 2015. Archived from the original on 7 March 2017.
- Jacobson R (2006). Illegal drugs: America's anguish (2005 ed.). Farmington Hills, Michigan: Thomson Gale. ISBN 978-1-4144-0419-6.
- "2015 National Drug Threat Assessment Summary" (PDF). Drug Enforcement Administration. United States Department of Justice: Drug Enforcement Administration. October 2015. pp. 1–2. Archived from the original (PDF) on 10 April 2016. Retrieved 10 April 2016.
Mexican TCOs pose the greatest criminal drug threat to the United States; no other group is currently positioned to challenge them. These Mexican poly-drug organizations traffic heroin, methamphetamine, cocaine, and marijuana throughout the United States, using established transportation routes and distribution networks. ... While all of these Mexican TCOs transport wholesale quantities of illicit drugs into the United States, the Sinaloa Cartel appears to be the most active supplier. The Sinaloa Cartel leverages its expansive resources and dominance in Mexico to facilitate the smuggling and transportation of drugs throughout the United States.
- "Coast Guard hunts drug-running semi-subs". CNN. 20 March 2008. Archived from the original on 21 March 2008. Retrieved 20 March 2008.
- "Meth Info". Methproject.org. Archived from the original on 27 March 2010.
- "Drugs of Abuse". City of Denison Iowa. Archived from the original on 6 November 2011. Retrieved 13 November 2011.
- "Drugs: Pricing Power". The Economist. 28 June 2007. Archived from the original on 6 January 2014.
Prices: USA around $110/g, Israel/Germany/Britain around $46/g, Colombia $2/g, New Zealand recordbreaking $714.30/g.
- European Monitoring Centre for Drugs and Drug Addiction (2008). Annual report: the state of the drugs problem in Europe (PDF). Luxembourg: Office for Official Publications of the European Communities. p. 59. ISBN 978-92-9168-324-6. Archived (PDF) from the original on 25 April 2013. Retrieved 31 December 2013.
- The Cocaine Threat: A Hemispheric Perspective (PDF). United States Department of Defense. Archived from the original (PDF) on 11 September 2008.
- United Nations (June 2010). World Drug Report 2010. United Nations Publications. p. 77. ISBN 978-92-1-148256-0. Archived from the original on 26 April 2016.
- Sawada H, Yamakawa K, Yamakado H, Hosokawa R, Ohba M, Miyamoto K, Kawamura T, Shimohama S (February 2005). "Cocaine and phenylephrine eye drop test for Parkinson disease". JAMA. Journal of the American Medical Association. 293 (8): 932–4. doi:10.1001/jama.293.8.932-c. PMID 15728162.
Bibliography
- Gootenberg P, ed. (1999). Cocaine: Global Histories. London: Routledge. ISBN 978-0-203-02646-5.
- Madge T (2001). White Mischief: A Cultural History of Cocaine. Edinburgh: Mainstream Publishing Company. ISBN 978-1-84018-405-1.
- Spillane JF (2000). Cocaine: From Medical Marvel to Modern Menace in the United States, 1884–1920. Baltimore and London: The Johns Hopkins University Press. ISBN 978-0-8018-6230-4.
Further reading
- Feiling T (2009). The Candy Machine: How Cocaine Took Over the World. London: Penguin. ISBN 978-0-14-103446-1.
External links
| Wikiquote has quotations related to: Cocaine |
| Wikimedia Commons has media related to Cocaine. |
| Look up cocaine in Wiktionary, the free dictionary. |
- U.S. National Library of Medicine: Drug Information Portal – Cocaine
- European Monitoring Centre for Drugs (Cocaine)
- Erowid – Cocaine Information — A collection of data about cocaine including dose, effects, chemistry, legal status, images and more.
- Slang Dictionary for Cocaine.
- Cocaine Market Data and Value-Havocscope Black Markets at the Wayback Machine (archived 23 March 2011) Data on cocaine trafficking worldwide.