Substituted tryptamine
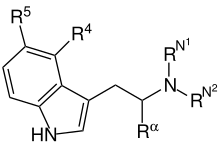
Substituted tryptamines, or serotonin analogues, are organic compounds which may be thought of as being derived from tryptamine itself. The molecular structures of all tryptamines contain an indole ring, joined to an amino (NH2) group via an ethyl (−CH2–CH2−) sidechain. In substituted tryptamines, the indole ring, sidechain, and/or amino group are modified by substituting another group for one of the hydrogen (H) atoms.
Well-known tryptamines include serotonin, an important neurotransmitter, and melatonin, a hormone involved in regulating the sleep-wake cycle. Tryptamine alkaloids are found in fungi, plants and animals; and sometimes used by humans for the neurological or psychotropic effects of the substance. Prominent examples of tryptamine alkaloids include psilocybin (from "Psilocybin mushrooms") and DMT. In South America, dimethyltryptamine is obtained from numerous plant sources, like chacruna, and it is often used in ayahuasca brews. Many synthetic tryptamines have also been made, including the migraine drug sumatriptan, and psychedelic drugs.
The tryptamine structure, in particular its indole ring, may be part of the structure of some more complex compounds, for example: LSD, ibogaine, mitragynine and yohimbine. A thorough investigation of dozens of tryptamine compounds was published by Ann and Alexander Shulgin under the title TiHKAL.
List of substituted tryptamines
Substituted alpha-alkyltryptamines also comprise a large class and are listed separately.
| Chemical structure | Short Name | Origin | Rα | R4 | R5 | RN1 | RN2 | Full Name |
|---|---|---|---|---|---|---|---|---|
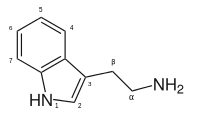 | Tryptamine | Natural | H | H | H | H | H | 3-(2-aminoethyl)indole / 2-(1H-indol-3-yl)ethanamine |
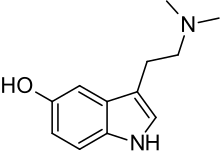 | Bufotenin | Natural | H | H | OH | CH3 | CH3 | 5-hydroxy-N,N-dimethyltryptamine |
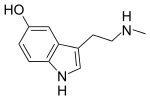 | Nω-Methylserotonin (norbufotenin) | Natural | H | H | OH | CH3 | H | 5-hydroxy-N-methyltryptamine |
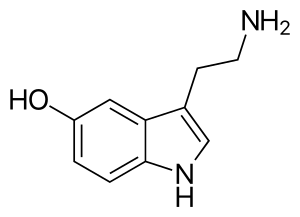 | Serotonin | Natural | H | H | OH | H | H | 5-hydroxytryptamine |
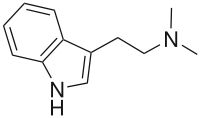 | DMT | Natural | H | H | H | CH3 | CH3 | N,N-dimethyltryptamine |
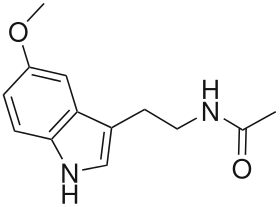 | Melatonin | Natural | H | H | OCH3 | O=C-CH3 | H | 5-methoxy-N-acetyltryptamine |
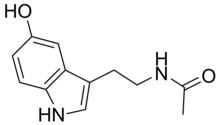 | N-Acetylserotonin | Natural | H | H | OH | O=C-CH3 | H | 5-hydroxy-N-acetyltryptamine |
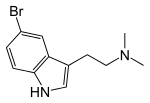 | 5-Bromo-DMT | Natural | H | H | Br | CH3 | CH3 | 5-bromo-N,N-dimethyltryptamine |
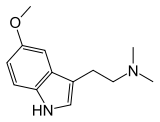 | 5-MeO-DMT | Natural | H | H | OCH3 | CH3 | CH3 | 5-methoxy-N,N-dimethyltryptamine |
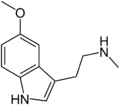 | 5-MeO-NMT | Natural | H | H | OCH3 | CH3 | H | 5-methoxy-N-methyltryptamine |
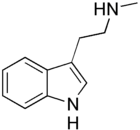 | NMT | Natural | H | H | H | H | CH3 | N-methyltryptamine |
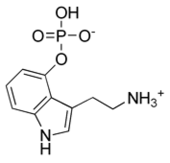 | Norbaeocystin | Natural | H | OPO3H2 | H | H | H | 4-phosphoryloxy-tryptamine |
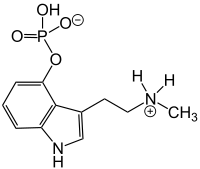 | Baeocystin | Natural | H | OPO3H2 | H | CH3 | H | 4-phosphoryloxy-N-methyl-tryptamine |
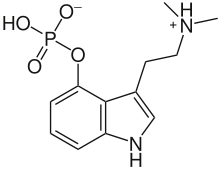 | Psilocybin | Natural | H | PO4 | H | CH3 | CH3 | 4-phosphoryloxy-N,N-dimethyltryptamine |
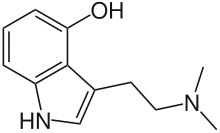 | Psilocin | Natural | H | OH | H | CH3 | CH3 | 4-hydroxy-N,N-dimethyltryptamine |
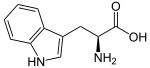 | Tryptophan | Natural | COOH | H | H | H | H | α-carboxyltryptamine |
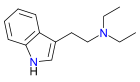 | DET | artificial | H | H | H | CH2CH3 | CH2CH3 | N,N-diethyltryptamine |
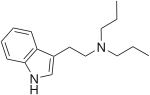 | DPT | artificial | H | H | H | CH2CH2CH3 | CH2CH2CH3 | N,N-dipropyltryptamine |
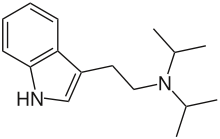 | DiPT | artificial | H | H | H | CH(CH3)2 | CH(CH3)2 | N,N-diisopropyltryptamine |
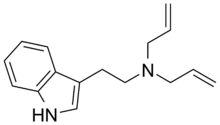 | DALT | artificial | H | H | H | H2C=CH-CH2 | H2C=CH-CH2 | N,N-diallyltryptamine |
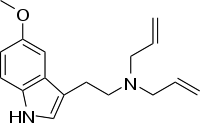 | 5-MeO-DALT | artificial | H | H | OCH3 | H2C=CH-CH2 | H2C=CH-CH2 | 5-methoxy-N,N-diallyltryptamine |
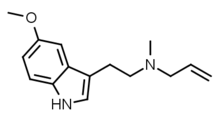 | 5-MeO-MALT | artificial | H | H | OCH3 | H2C=CH-CH2 | CH3 | 5-methoxy-N-Methyl-N-allyltryptamine |
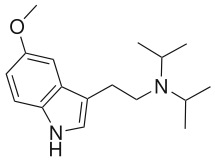 | 5-MeO-DIPT | artificial | H | H | OCH3 | CH(CH3)2 | CH(CH3)2 | 5-methoxy-N,N-diisopropyltryptamine |
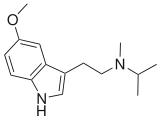 | 5-MeO-MiPT | artificial | H | H | OCH3 | CH3 | CH(CH3)2 | 5-methoxy-N,N-methylisopropyltryptamine |
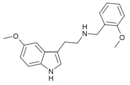 | 5-MT-NBOMe | artificial | H | H | OCH3 | H | CH2C6H4(o-OCH3) | 5-methoxy-N-(ortho-methoxybenzyl)tryptamine |
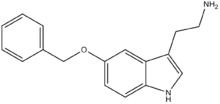 | 5-BT | artificial | H | H | OCH2C6H5 | H | H | 5-Benzyloxytryptamine |
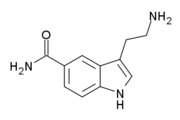 | 5-CT | artificial | H | H | CONH2 | H | H | 5-Carboxamidotryptamine |
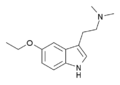 | 5-Ethoxy-DMT | artificial | H | H | OCH2CH3 | CH3 | CH3 | 5-ethoxy-N,N-dimethyltryptamine |
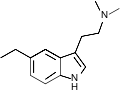 | 5-Ethyl-DMT | artificial | H | H | CH2CH3 | CH3 | CH3 | 5-ethyl-N,N-dimethyltryptamine |
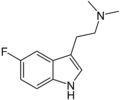 | 5-Fluoro-DMT | artificial | H | H | F | CH3 | CH3 | 5-fluoro-N,N-dimethyltryptamine |
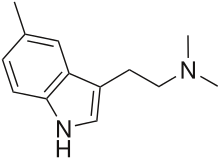 | 5-Methyl-DMT (5,N,N-TMT) | artificial | H | H | CH3 | CH3 | CH3 | 5,N,N-trimethyltryptamine |
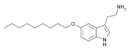 | 5-(Nonyloxy)tryptamine | artificial | H | H | O(CH2)8CH3 | H | H | 5-nonyloxytryptamine |
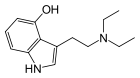 | 4-HO-DET | artificial | H | OH | H | CH2CH3 | CH2CH3 | 4-hydroxy-N,N-diethyltryptamine |
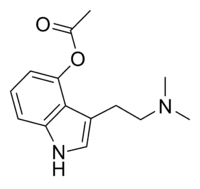 | 4-AcO-DMT | artificial | H | OCOCH3 | H | CH3 | CH3 | 4-acetoxy-N,N-dimethyltryptamine |
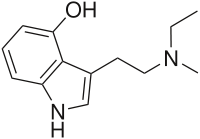 | 4-HO-MET | artificial | H | OH | H | CH3 | CH2CH3 | 4-hydroxy-N-methyl-N-ethyltryptamine |
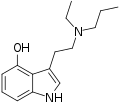 | 4-HO-EPT | artificial | H | OH | H | CH2CH3 | CH2CH2CH3 | 4-hydroxy-N-ethyl-N-propyltryptamine |
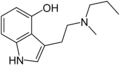 | 4-HO-MPT | artificial | H | OH | H | CH3 | CH2CH2CH3 | 4-hydroxy-N-methyl-N-propyltryptamine |
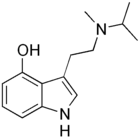 | 4-HO-MiPT | artificial | H | OH | H | CH(CH3)2 | CH3 | 4-hydroxy-N-isopropyl-N-methyltryptamine |
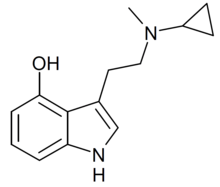 | 4-HO-McPT | artificial | H | OH | H | C3H5 | CH3 | 4-hydroxy-N-cyclopropyl-N-methyltryptamine |
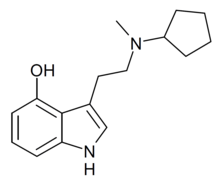 | 4-HO-McPeT | artificial | H | OH | H | C5H9 | CH3 | 4-hydroxy-N-cyclopentyl-N-methyltryptamine |
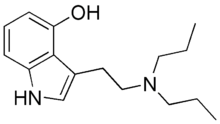 | 4-HO-DPT | artificial | H | OH | H | CH2CH2CH3 | CH2CH2CH3 | 4-hydroxy-N,N-dipropyltryptamine |
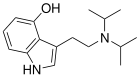 | 4-HO-DIPT | artificial | H | OH | H | CH(CH3)2 | CH(CH3)2 | 4-hydroxy-N,N-diisopropyltryptamine |
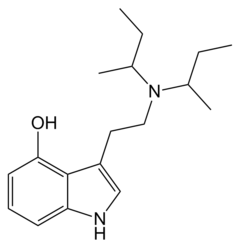 | 4-HO-DSBT | artificial | H | OH | H | CH(CH3)CH2CH3 | CH(CH3)CH2CH3 | 4-hydroxy-N,N-disecbutyltryptamine |
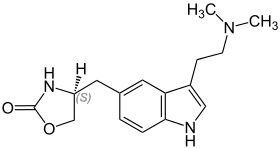 | Zolmitriptan | artificial | H | H | -(CHNHC=OOCH2) | CH3 | CH3 | 5-( 4-(S)-1,3-oxazolidin-2-one)-N,N-dimethyltryptamine |
| Chemical Structure | Short Name | Origin | Rα | R4 | R5 | RN1 | RN2 | Full Name |