Homarylamine
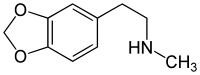
Homarylamine (INN;[1] also known as 3,4-methylenedioxy-N-methylphenethylamine and MDMPEA) is a substituted phenethylamine. It is the N-methylated analog of MDPEA. Homoarylamine was patented as an antitussive in 1956,[2] but has never been used medically as such.
 | |
| Clinical data | |
|---|---|
| Other names | 1,3-benzodioxolyl-N-methyl-5-ethanamine; 3,4-methylenedioxy-N-methyl-2-phenylethylamine; Norlobivine |
| Routes of administration | Various |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C10H13NO2 |
| Molar mass | 179.21572 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Reactions
Reaction of homoarylamine with formaldehyde gives hydrastinine.
See also
- Hydrastine, an alkaloid derivative of homarylamine
References
- "International Non-Proprietary Names for Pharmaceutical Preparations" (PDF). Chronicle of the World Health Organization. 12 (3). 1958.
- U.S. Patent 2,820,739
Empathogens/entactogens | |
|---|---|
| Phenylalkyl- amines (other than cathinones) |
|
| Cyclized phenyl- alkylamines | |
| Cathinones |
|
| Tryptamines | |
| Chemical classes | |
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.