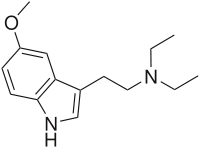
5-MeO-DET
5-MeO-DET or 5-methoxy-N,N-diethyltryptamine is a hallucinogenic tryptamine.
 | |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C15H22N2O |
| Molar mass | 246.36 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Pharmacology
5-MeO-DET inhibits serotonin reuptake with an IC50 value of 2.4 μM and activates 5-HT2A receptors with an EC50 value of 8.11 nM.[1][2][3][4][5][6][7]
Effects
Low dosages (0.5–1 mg) are reported to produce a relaxing body high and mild entheogenic effects. Shulgin reports in TiHKAL that higher dosages (1–3 mg) can produce very unpleasant reactions.
See also
- 5-MeO-DiPT
- 5-MeO-DMT
- 5-MeO-MiPT
- TiHKAL
References
- Meike Schulze-Alexandru; Karl-Artur Kovar; Angelo Vedani (December 1999). "Quasi-atomistic Receptor Surrogates for the 5-HT2A Receptor: A 3D-QSAR Study on Hallucinogenic Substances". Quantitative Structure-Activity Relationships. 18 (6): 548–560. CiteSeerX 10.1.1.669.1877. doi:10.1002/(SICI)1521-3838(199912)18:6<548::AID-QSAR548>3.0.CO;2-B.
- Michael B. Gatch; Michael J. Forster; Aaron Janowsky; Amy J. Eshleman (April 2011). "Abuse Liability Profile of Three Substituted Tryptamines". Journal of Pharmacology and Experimental Therapeutics. 338 (1): 280–289. doi:10.1124/jpet.111.179705. PMC 3126641. PMID 21474568.
- Richard A. Glennon; Peter K. Gessner (April 1979). "Serotonin receptor binding affinities of tryptamine analogs". Journal of Medicinal Chemistry. 22 (4): 428–432. doi:10.1021/jm00190a014. PMID 430481.
- Adam L. Halberstadt; Mark A. Geyer (September 2011). "Multiple receptors contribute to the behavioral effects of indoleamine hallucinogens". Neuropharmacology. 61 (3): 364–381. doi:10.1016/j.neuropharm.2011.01.017. PMC 3110631. PMID 21256140.
- P.K. Gessner; D.D. Godse; A.H. Krull; J.M. McMullan (March 1968). "Structure-activity relationships among 5-methoxy-N:N-dimethyltryptamine, 4-hydroxy-N:N-dimethyltryptamine (psilocin) and other substituted tryptamines". Life Sciences. 7 (5): 267–277. doi:10.1016/0024-3205(68)90200-2. PMID 5641719.
- Robert A. Lyon; Milt Titeler; Mark R. Seggel; Richard A. Glennon (January 1988). "Indolealkylamine analogs share 5-HT2 binding characteristics with phenylalkylamine hallucinogens". European Journal of Pharmacology. 145 (3): 291–297. doi:10.1016/0014-2999(88)90432-3. PMID 3350047.
- Bruce E. Blough; Antonio Landavazo; Ann M. Decker; John S. Partilla; Michael H. Baumann; Richard B. Rothman (October 2014). "Interaction of psychoactive tryptamines with biogenic amine transporters and serotonin receptor subtypes". Psychopharmacology. 231 (21): 4135–4144. doi:10.1007/s00213-014-3557-7. PMC 4194234. PMID 24800892.
External links
Drugs mentioned in TiHKAL | |
|---|---|
|
Serotonin receptor modulators | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.