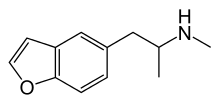
5-MAPB
5-MAPB (1-(benzofuran-5-yl)-N-methylpropan-2-amine) is an entactogenic designer drug similar to MDMA in its structure and effects.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral, Insufflated, Rectal |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C12H15NO |
| Molar mass | 189.25 g/mol (freebase) 225.7 g/mol (HCl salt) g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Legal Status
Canada
5-MAPB is not listed itself in the CDSA but since it is structurally related to MDMA it may be considered illegal in Canada, although this has not been tested in court.[2]
China
As of October 2015 5-MAPB is a controlled substance in China.[3]
Pharmacokinetics
Pharmacodynamics
5-MAPB binds to the dopamine transporter in rat brain cells with a lower potency than cocaine. In silico data suggests that the primary action on dopamine is through reversal of the transporter to release dopamine. This is consistent with the effects and it is likely that it exerts a similar action on serotonin and norepinephrine transporters.[10]
References
- "Temporary class drug order report on 5-6APB and NBOMe compounds". UK Home Office. 4 Jun 2013. Retrieved 2013-07-10.
- "Schedule I". Government Of Canada. 2014-12-12. Archived from the original on 2013-11-22. Retrieved 2014-12-13.
- "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Retrieved 1 October 2015.
- "'NBOMe' and 'Benzofury' banned". UK Home Office. 2013-06-04. Retrieved 10 April 2014.
- UK Home Office (2014-03-05). "The Misuse of Drugs Act 1971 (Ketamine etc.) (Amendment) Order 2014". UK Government. Retrieved 2014-03-11.
- McCann UD; Ricaurte GA (1991). "Major metabolites of (±)3,4-methylenedioxyamphetamine (MDA) do not mediate its toxic effects on brain serotonin neurons". Brain Research. 545 (1–2): 279–282. doi:10.1016/0006-8993(91)91297-E. PMID 1860050.
- Miller RT; Lau SS; Monks TJ (1997). "2,5-Bis-(glutathion-S-yl)-alpha-methyldopamine, a putative metabolite of (+/-)-3,4-methylenedioxyamphetamine, decreases brain serotonin concentrations". Eur J Pharmacol. 323 (2–3): 173–80. doi:10.1016/S0014-2999(97)00044-7. PMID 9128836.
- Conway EL; Louis WJ; Jarrott B (1978). "Acute and chronic administration of alpha-methyldopa: regional levels of endogenous and alpha-methylated catecholamines in rat brain". Eur J Pharmacol. 52 (3–4): 271–80. doi:10.1016/0014-2999(78)90279-0. PMID 729639.
- Welter, Jessica; Kavanagh, Pierce; Meyer, Markus R.; Maurer, Hans H. (2014). "Benzofuran analogues of amphetamine and methamphetamine: Studies on the metabolism and toxicological analysis of 5-APB and 5-MAPB in urine and plasma using GC-MS and LC-(HR)-MSn techniques". Analytical and Bioanalytical Chemistry. 407 (5): 1371–88. doi:10.1007/s00216-014-8360-0. PMID 25471293.
- Sahai, Michelle A; Davidson, Colin; Khelashvili, George (2016). "Combined in vitro and in silico approaches to the assessment of stimulant properties of novel psychoactive substances – The case of the benzofuran 5-MAPB" (PDF). Progress in Neuro-Psychopharmacology and Biological Psychiatry. 75: 1–9. doi:10.1016/j.pnpbp.2016.11.004. PMID 27890676.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.