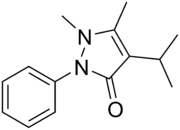
Propyphenazone
Propyphenazone is a derivative of phenazone[1] with similar analgesic and antipyretic effects. It is known as isopropylantipyrine in Japan.[2]
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.855 |
| Chemical and physical data | |
| Formula | C14H18N2O |
| Molar mass | 230.306 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
It was patented in 1931.[3]
Adverse effects
It is used in making of Saridon (by Piramal Enterprises in India and Bayer for Global market) There have been case reports describing risk of acute inferior myocardial infarction with low atrial rhythm due to propyphenazone (Kounis syndrome).[4]
Banned
Excerpt from WHO comments:
- “Propyphenazone, a pyrazolone derivative with anti-inflammatory, analgesic and antipyretic activity, was introduced in 1951 for the treatment of rheumatic disorders. As it is structurally related to aminophenazone it has been associated with severe blood dyscrasias. However, it cannot be transformed into potentially carcinogenic nitrosamines and has therefore been widely used as a replacement drug for aminophenazone. In certain countries, products containing propyphenazone have now been restricted in their indications, whereas in others they are still available, sometimes as over-the-counter preparations.”[5]
References
- Göres, E.; Kossowicz, J.; Schneider, H. G. (2004). "Propyphenazone. Pharmacology and use" [Propyphenazone. Pharmacology and use]. Medizinische Monatsschrift für Pharmazeuten (in German). 27 (3): 72–76. PMID 15032249.
- "Isopropylantipyrine". Drugs.com. Retrieved 8 March 2018.
- Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 530. ISBN 9783527607495.
- Akyel, A.; Alsancak, Y.; Yayla, Ç. R.; Şahinarslan, A.; Özdemir, M. (2011). "Acute inferior myocardial infarction with low atrial rhythm due to propyphenazone: Kounis syndrome". International Journal of Cardiology. 148 (3): 352–353. doi:10.1016/j.ijcard.2010.05.038. PMID 20541820.
- Consolidated List of Products whose Consumption and/or Sale have been Banned, Withdrawn, Severely Restricted or not Approved by Governments, Twelfth Issue (pdf). New York: Department of Economic and Social Affairs of the United Nations Secretariat. 2005. p. 232.
- "Multi-Country Survey On Banned And Restricted Pharmaceuticals". Health Action International Asia Pacific. August 2008. p. 7.
- "Govt bans Saridon, 327 other combination drugs - Times of India". The Times of India. Retrieved 2018-09-13.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.