Clofedanol
Clofedanol (INN) or chlophedianol (BAN) is a centrally acting cough suppressant used in the treatment of dry cough. Clofedanol has local anesthetic and antihistamine properties, and may have anticholinergic effects at high doses.[1] It is marketed in Canada under the trade name Ulone, but is not available currently in the United States. In August 2009, Centrix Pharmaceutical announced the launch of a cough syrup containing a mixture of clophedianol and pseudoephedrine, marketed under the brand name Clofera. It has a release to market date in the beginning of the fourth quarter 2009.[2] Chlophedianol was approved for OTC status in 1987 by the FDA OTC monograph process[3] and its safety and efficacy data are limited.
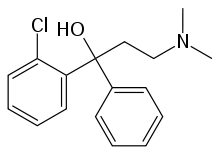 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Oral |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.011.219 |
| Chemical and physical data | |
| Formula | C17H20ClNO |
| Molar mass | 289.8 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Synthesis
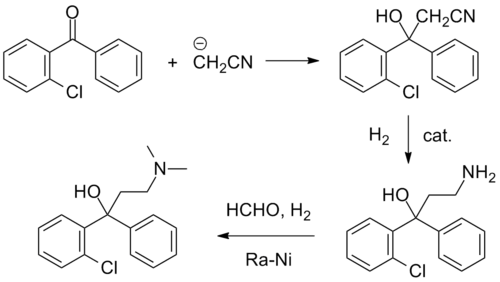
Clofedanol synthesis: R. Lorenz and H. Henecka, U.S. Patent 3,031,377 (1962).
See also
References
- "Clofedanol" (in French). BIAM. 1998-07-24. Retrieved 2007-04-15.
- "Centrix Pharmaceutical Announces Clofera(TM), a Unique Antitussive and Nasal Decongestant for the Temporary Relief of Cough and Nasal Congestion". Cenrx.com. Archived from the original on 2009-08-21. Retrieved 2009-09-27.
- "Department of Health and Human Services. Food and Drug Administration. 21 CFR Parts 310, 341, and 369. Docket No. 76N-052T. Cold, cough, allergy, bronchodilator, and antiasthmatic drug products for over-the0counter human use; final monograph for OTC antitussive drug products. Federal Register 1987;52(155):30042-57" (PDF). FDA.gov. 1987-08-12.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.