Metamfepramone
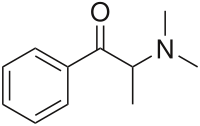
Metamfepramone (INN, also known as dimethylcathinone, dimethylpropion, and dimepropion (BAN)) is a stimulant drug of the phenethylamine, and cathinone chemical classes. Dimethylcathinone was evaluated as an appetite suppressant and for the treatment of hypotension, but was never widely marketed.[2]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.035.788 |
| Chemical and physical data | |
| Formula | C11H15NO |
| Molar mass | 177.243 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
It was used as a recreational drug in Israel under the name rakefet, but was made illegal in 2006.[3]
Metamfepramone is metabolized to produce N-methylpseudoephedrine and methcathinone.[4]
See also
References
- "Ustawa z dnia 15 kwietnia 2011 r. o zmianie ustawy o przeciwdziałaniu narkomanii ( Dz.U. 2011 nr 105 poz. 614 )". Internetowy System Aktów Prawnych. Retrieved 17 June 2011.
- Soholing WE (1982). "Therapy of the orthostatic syndrome. Studies using dimepropion-HCl". Fortschritte der Medizin (in German). 100 (7): 289–293.
- Judy Siegel-Itzkovich (2006-02-22). "Recreational drug 'rakefet' banned". Jpost.com. Retrieved 2010-09-13.
- Thevis, M; Sigmund, G; Thomas, A; Gougoulidis, V; Rodchenkov, G; Schänzer, W (2009). "Doping control analysis of metamfepramone and two major metabolites using liquid chromatography-tandem mass spectrometry". European Journal of Mass Spectrometry. 15 (4): 507–15. doi:10.1255/ejms.1010. PMID 19661559.
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.