Estradiol cypionate/medroxyprogesterone acetate
Estradiol cypionate/medroxyprogesterone acetate (EC/MPA), sold under the brand name Cyclofem among others, is a form of combined injectable birth control.[2] It contains estradiol cypionate (EC), an estrogen, and medroxyprogesterone acetate (MPA), a progestin.[2] It is recommended for short-term use and is given once a month by injection into a muscle.[3]
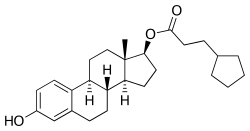 | |
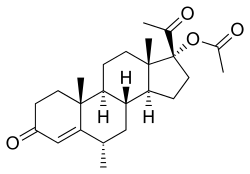 Estradiol cypionate (top) and medroxy- progesterone acetate (bottom) | |
| Combination of | |
|---|---|
| Estradiol cypionate | Estrogen |
| Medroxyprogesterone acetate | Progestogen |
| Clinical data | |
| Trade names | Cyclofem, Lunelle, others[1] |
| Other names | EC/MPA; Cyclo-Provera; HRP-112 |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | Intramuscular injection |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
Common side effects of EC/MPA include irregular menstrual periods which typically improves with time.[3] Other side effects include blood clots, headache, hair loss, depression, nausea, and breast pain.[3][4] Use during pregnancy is not recommended.[2] Use during breastfeeding is likely safe.[5] It works mainly by preventing ovulation.[2]
EC/MPA came into medical use in 1993 with approval in the United States in 2000.[6][2] It is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system.[7] The medication is approved for use in 18 countries.[8] It is used in Mexico, Thailand, and Indonesia, among other countries.[6] It is no longer commercially available in the United States.[2]
Medical uses
EC/MPA is used as a once-monthly combined injectable contraceptive to prevent pregnancy in women.[9]
Available forms
EC/MPA is available in the form of a microcrystalline aqueous suspension of 5 mg EC and 25 mg MPA given in a 0.5 mL aqueous solution for intramuscular injection once per month.[9] It is provided in the form of single-dose vials and ampoules.[9] The particle sizes of the formulation are 93% within a range of 5 to 16 μm.[10]
History
Depot MPA (DMPA) and EC/MPA were developed by Upjohn in the 1960s.[11][12] DMPA (brand name Depo-Provera) was introduced for use as a progestogen-only injectable contraceptive for the first time outside of the United States in 1969 and was subsequently approved for use in birth control in the United States in 1992.[13][14][12] A variety of preliminary studies on EC/MPA as a contraceptive were published between 1968 and 1978.[9][15][16] In the late 1970s, the World Health Organization (WHO) began an initiative known as the Special Programme of Research, Development and Research Training in Human Reproduction (WHO/HRP) to develop combined injectable contraceptives as part of its efforts to increase the availability and choices of birth control for women and men throughout the world.[15] They sought to remedy the side effect of menstrual irregularity that is the major reason for discontinuation of progestogen-only injectable contraceptives like DMPA via incorporation of an estrogen.[9] Funding for the initiative was provided by the United Nations Development Programme (UNDP), United Nations Population Fund (UNFPA), and World Bank.[15]
In 1980, the WHO conducted a pharmacokinetic study of estradiol cypionate, estradiol valerate, and estradiol benzoate to determine which ester(s) would be most suitable for use in combined injectable contraceptives.[9][15][17] Following the initial development of the medication by Upjohn under the code name Cyclo-Provera previously, EC/MPA was developed as a combined injectable contraceptive by the World Health Organization under the code name HRP-112 in the 1980s and early 1990s.[15][9] The WHO also developed estradiol valerate/norethisterone enantate (code name HRP-102) as a combined injectable contraceptive.[15][9] Development of EC/MPA was completed by the early 1990s and the medication was licensed to the WHO/HRP-established Concept Foundation as well as several pharmaceutical companies.[15] EC/MPA was introduced for use as a combined injectable contraceptive under the brand name Cyclofem and others in Mexico, Thailand, and Indonesia in 1993.[6][9][18] It was available in 18 countries, mostly in Latin America and Asia, by 1998.[15] In late 2000, EC/MPA was introduced for use as a combined injectable contraceptive by Upjohn under the brand name Lunelle in the United States.[15][11] However, the formulation was discontinued in the United States in late 2003.[19]
Society and culture
Generic names
EC/MPA was originally known as Cyclo-Provera (or Cycloprovera), and is also known by its former developmental code name HRP-112.[20]
Brand names
Brand names of EC/MPA include Ciclofem, Ciclofemina, Cyclofem, Cyclofemina, Cyclogeston, Femelin, Femydrol, Gestin, Harmonis, Lunella, Lunelle, and Novafem.[21][22]
Availability
EC/MPA has been approved for use in at least 18 countries.[8] It is or has been used in Bolivia, Brazil, Chile, China, Colombia, Costa Rica, the Dominican Republic, Ecuador, El Salvador, Guatemala, Hong Kong, Indonesia, Malaysia, Mexico, Panama, Peru, Thailand, the United States, and Zimbabwe, among other countries.[21][22][23][24][25][8][6][26][27] The medication is no longer available in the United States.[2]
See also
References
- Humans, IARC Working Group on the Evaluation of Carcinogenic Risks to; Organization, World Health; Cancer, International Agency for Research on (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. p. 431. ISBN 9789283212911. Archived from the original on 2017-01-02.
- "Estradiol and medroxyprogesterone Intramuscular Advanced Patient Information - Drugs.com". www.drugs.com. Archived from the original on 1 January 2017. Retrieved 1 January 2017.
- WHO Model Formulary 2008 (PDF). World Health Organization. 2009. pp. 368–370. ISBN 9789241547659. Archived (PDF) from the original on 13 December 2016. Retrieved 8 December 2016.
- "Estradiol/medroxyprogesterone Side Effects in Detail - Drugs.com". www.drugs.com. Archived from the original on 1 January 2017. Retrieved 1 January 2017.
- "Estradiol / medroxyprogesterone (Lunelle) Use During Pregnancy". www.drugs.com. Archived from the original on 1 January 2017. Retrieved 1 January 2017.
- Senanayake, Pramilla; Potts, Malcolm (2008). Atlas of Contraception, Second Edition (2 ed.). CRC Press. p. 51. ISBN 9780203347324. Archived from the original on 2017-01-02.
- "WHO Model List of Essential Medicines (19th List)" (PDF). World Health Organization. April 2015. Archived (PDF) from the original on 13 December 2016. Retrieved 8 December 2016.
- Bagade O, Pawar V, Patel R, Patel B, Awasarkar V, Diwate S (2014). "Increasing use of long-acting reversible contraception: safe, reliable, and cost-effective birth control" (PDF). World J Pharm Pharm Sci. 3 (10): 364–392. ISSN 2278-4357.
- Newton JR, D'arcangues C, Hall PE (1994). "A review of "once-a-month" combined injectable contraceptives". J Obstet Gynaecol (Lahore). 4 Suppl 1: S1–34. doi:10.3109/01443619409027641. PMID 12290848.
- Sang GW (April 1994). "Pharmacodynamic effects of once-a-month combined injectable contraceptives". Contraception. 49 (4): 361–85. doi:10.1016/0010-7824(94)90033-7. PMID 8013220.
The exact formulation and the size of the microcrystals is most important for duration of action. The smaller particles are more rapidly dissolved than larger ones and, hence, MPA appears more rapidly in the circulation, with more rapid elimination from the body. This is also true for the once-a-month formulation, Cyclofem. From our laboratory’s data, the distribution of particle size of Cyclofem is showed in Table 2. [...] TABLE 2. Distribution of particle size of crystalline steroids in Cyclofem (aqueous suspension). Size of particle (μm): ≤4: 0.3%. 5–6: 7.3%. 6–8: 16.7%. 8–10: 29.0%. 10–13: 29.7%. 13–16: 9.9%. 16–20: 0.5%. >20: 0.1%.
- Kaunitz AM (December 2000). "Injectable contraception. New and existing options". Obstet. Gynecol. Clin. North Am. 27 (4): 741–80. doi:10.1016/S0889-8545(05)70171-6. PMID 11091987.
- Kaunitz AM (March 2001). "Injectable long-acting contraceptives". Clin Obstet Gynecol. 44 (1): 73–91. doi:10.1097/00003081-200103000-00011. PMID 11219248.
- Anne Nadakavukaren (28 February 2011). Our Global Environment: A Health Perspective, Seventh Edition. Waveland Press. pp. 63–. ISBN 978-1-4786-0976-6.
- Levitt JI (30 April 2015). Black Women and International Law: Deliberate Interactions, Movements and Actions. Cambridge University Press. pp. 230–231. ISBN 978-1-316-29840-4.
- Hall PE (August 1998). "New once-a-month injectable contraceptives, with particular reference to Cyclofem/Cyclo-Provera". Int J Gynaecol Obstet. 62 Suppl 1: S43–56. doi:10.1016/S0020-7292(98)00090-3. PMID 9806239.
- Coutinho EM, Carlos de Souza J (March 1968). "Conception control by monthly injections of medroxyprogesterone suspension and long-acting oestrogen". J. Reprod. Fertil. 15 (2): 209–14. doi:10.1530/jrf.0.0150209. PMID 5643482.
- Oriowo MA, Landgren BM, Stenström B, Diczfalusy E (April 1980). "A comparison of the pharmacokinetic properties of three estradiol esters". Contraception. 21 (4): 415–24. doi:10.1016/S0010-7824(80)80018-7. PMID 7389356.
- d'Arcangues C (1993). "Once-a-month injectable contraceptives". World Health Forum. 14 (4): 439–40. PMID 8185807.
- Nagrath Arun; Malhotra Narendra; Seth Shikha (15 December 2012). Progress in Obstetrics and Gynecology--3. Jaypee Brothers Medical Publishers Pvt. Ltd. pp. 416–. ISBN 978-93-5090-575-3.
- Koetsawang S (April 1994). "Once-a-month injectable contraceptives: efficacy and reasons for discontinuation". Contraception. 49 (4): 387–98. doi:10.1016/0010-7824(94)90034-5. PMID 8013221.
- https://www.drugs.com/international/estradiol.html
- https://www.drugs.com/international/medroxyprogesterone.html
- Sweetman, Sean C., ed. (2009). "Sex hormones and their modulators". Martindale: The Complete Drug Reference (36th ed.). London: Pharmaceutical Press. p. 2082. ISBN 978-0-85369-840-1.
- http://www.micromedexsolutions.com/micromedex2/librarian/
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer (1 January 1999). Hormonal Contraception and Post-menopausal Hormonal Therapy (PDF). IARC. p. 65. ISBN 978-92-832-1272-0.
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. pp. 431–. ISBN 978-92-832-1291-1.
- Klitsch M (1995). "Still waiting for the contraceptive revolution". Fam Plann Perspect. 27 (6): 246–53. doi:10.2307/2136177. JSTOR 2136177. PMID 8666089.