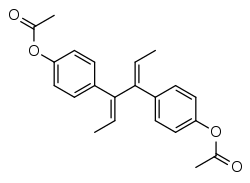
Dienestrol diacetate
Dienestrol diacetate (brand names Faragynol, Gynocyrol, others) is a synthetic nonsteroidal estrogen of the stilbestrol group related to diethylstilbestrol.[1] It is an ester of dienestrol.[1]
| Estrogen | Type | Class | ETD (mg/14 days) | EPD (mg/14 days) | EPD (mg/day) | MSD (mg/14 days) | MSD (mg/day) | TSD (mg/day) |
|---|---|---|---|---|---|---|---|---|
| Estradiol (non-micronized) | Bioidentical | Steroidal | ? | ≥120–300 | ? | ? | ? | ? |
| Estradiol (micronized) | Bioidentical | Steroidal | ? | 60–80 | 4.3 | 14–28 | 1.0–2.0 | >8 |
| Estradiol valerate | Bioidentical | Steroidal | 6–10 | 60–80 | 4.3 | 14–28 | 1.0–2.0 | >8 |
| Estradiol benzoate | Bioidentical | Steroidal | ? | 60–140 | 4.5 | ? | ? | ? |
| Estriol | Bioidentical | Steroidal | 20a | 120–150b | 10.0–10.7b | 28–84 | 1.0–6.0 | ? |
| Estriol succinate | Bioidentical | Steroidal | ? | 140–150b | 10.0–10.7b | 28–84 | 2.0–6.0 | ? |
| Conjugated estrogens | Natural | Steroidal | 5–12 | 60–80 | 4.3 | 8.4–17.5 | 0.625–1.25 | 7.5 |
| Ethinylestradiol | Synthetic | Steroidal | 0.2 | 1.0–2.0 | 0.071–0.11 | 0.28 | 0.02–0.04 | 0.1 |
| Mestranol | Synthetic | Steroidal | 0.3 | 1.5–3.0 | 0.11–0.13 | 0.3–0.5 | 0.025 | ? |
| Quinestrol | Synthetic | Steroidal | 0.3 | 2.0–4.0 | 0.14–0.29 | ? | 0.025–0.05 | ? |
| Methylestradiol | Synthetic | Steroidal | ? | 2.0 | ? | ? | ? | ? |
| Diethylstilbestrol | Synthetic | Nonsteroidal | 2.5 | 20–30 | 1.4–2.1 | ? | 0.5–2.0 | 3 |
| Diethylstilbestrol dipropionate | Synthetic | Nonsteroidal | ? | 15–30 | 1.1–1.4 | ? | ? | ? |
| Dienestrol | Synthetic | Nonsteroidal | ? | 30 | ? | ? | 0.5–4.0 | ? |
| Dienestrol diacetate | Synthetic | Nonsteroidal | 3–5 | 30–60 | 2.9–4.3 | ? | ? | ? |
| Hexestrol | Synthetic | Nonsteroidal | ? | 70–110 | ? | ? | ? | ? |
| Hexestrol diacetate | Synthetic | Nonsteroidal | ? | 45 | ? | ? | ? | ? |
| Chlorotrianisene | Synthetic | Nonsteroidal | ? | >100 | ? | ? | ? | ? |
| Methallenestril | Synthetic | Nonsteroidal | ? | 400 | ? | ? | ? | ? |
| Note: The OID of EE is 0.1 mg/day. Footnotes: a = Very variable, often higher. b = In divided doses, 3x/day; irregular and atypical proliferation. Sources: See template. | ||||||||
| Estrogen | Form | Major brand name(s) | EPD (14 days) | Duration | |
|---|---|---|---|---|---|
| Diethylstilbestrol (DES) | Oil solution | ? | 20 mg | 3 mg ≈ 3 days | |
| Diethylstilbestrol dipropionate | Oil solution | Cyren B | 12.5–15 mg | 2.5 mg ≈ 5 days | |
| Aqueous suspension | ? | 5 mg | ? mg = 21–28 days | ||
| Dimestrol (DES dimethyl ether) | Oil solution | Depot-Cyren, Depot-Oestromon, Retalon Retard | 20–40 mg | ? | |
| Fosfestrol (DES diphosphate)a | Aqueous solution | Honvan | ? | <1 day | |
| Dienestrol diacetate | Aqueous suspension | Farmacyrol-Kristallsuspension | 50 mg | ? | |
| Hexestrol dipropionate | Oil solution | Hormoestrol, Retalon Oleosum | 25 mg | ? | |
| Hexestrol diphosphatea | Aqueous solution | Cytostesin, Pharmestrin, Retalon Aquosum | ? | Very short | |
| Note: All by intramuscular injection unless otherwise noted. Footnotes: a = By intravenous injection. Sources: See template. | |||||
 | |
| Clinical data | |
|---|---|
| Trade names | Faragynol, Gynocyrol |
| Drug class | Nonsteroidal estrogen; Estrogen ester |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C22H22O4 |
| Molar mass | 350.414 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
See also
References
- J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 390. ISBN 978-1-4757-2085-3.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.