Mespirenone
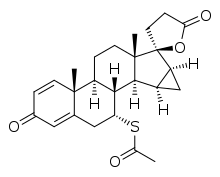
Mespirenone (INN) (developmental code name ZK-94679), also known as Δ1-15β,16β-methylenespironolactone, is a steroidal antimineralocorticoid of the spirolactone group related to spironolactone that was never marketed.[1][2] Animal research found that it was 3.3-fold more potent as an antimineralocorticoid relative to spironolactone.[3] In addition to its antimineralocorticoid properties, mespirenone is also a progestogen, antigonadotropin, and antiandrogen.[2][4] It is 2- to 3-fold as potent as spironolactone as a progestogen and antigonadotropin but its antiandrogenic activity is markedly reduced and weak (though still of significance) in comparison.[4][5] Mespirenone is also a potent and specific enzyme inhibitor of 18-hydroxylase and thus of mineralocorticoid biosynthesis.[6] The drug was under development by Schering (now Bayer Schering Pharma) and reached phase II clinical trials but was discontinued in 1989.[7]
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C25H30O4S |
| Molar mass | 426.5683 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
See also
References
- J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 775–. ISBN 978-1-4757-2085-3.
- Losert W, Bittler D, Buse M, Casals-Stenzel J, Haberey M, Laurent H, Nickisch K, Schillinger E, Wiechert R (1986). "Mespirenone and other 15,16-methylene-17-spirolactones, a new type of steroidal aldosterone antagonists". Arzneimittelforschung. 36 (11): 1583–600. PMID 3028435.
- Arzneimittel-Forschung. Editio Cantor. 1991.
- Nishino Y, Schröder H, el Etreby MF (1988). "Experimental studies on the endocrine side effects of new aldosterone antagonists". Arzneimittelforschung. 38 (12): 1800–5. PMID 3245852.
- Opoku J, Kalimi M, Agarwal M, Qureshi D (1991). "Effect of a new mineralocorticoid antagonist mespirenone on aldosterone-induced hypertension". Am. J. Physiol. 260 (2 Pt 1): E269–71. PMID 1996630.
- Weindel K, Lewicka S, Vecsei P (1991). "Inhibitory effects of the novel anti-aldosterone compound mespirenone on adrenocortical steroidogenesis in vitro". Arzneimittelforschung. 41 (9): 946–9. PMID 1796922.
- Eckhard Ottow; Hilmar Weinmann (8 September 2008). Nuclear Receptors as Drug Targets. John Wiley & Sons. pp. 410–. ISBN 978-3-527-62330-3.