Valofane
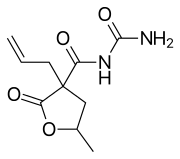
Valofane is a sedative drug structurally related to the barbiturates[1] and similar drugs such as primidone. It is metabolised once inside the body to form the barbiturate proxibarbital (proxibarbal) and is thus a prodrug.[2]
 | |
| Clinical data | |
|---|---|
| Other names | N-carbamoyl-5-methyl-2-oxo-3-prop-2-enyloxolane-3-carboxamide |
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| ECHA InfoCard | 100.019.871 |
| Chemical and physical data | |
| Formula | C10H14N2O4 |
| Molar mass | 226.229 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
References
- Traversa U, Puppini P, Jacquot C, Vertua R. Effect of an atypical barbiturate, the 2-allophanyl-2-allyl-4-valerolactone (valofan), on exploratory behaviour and brain serotonin concentrations in mice. Journal de Pharmacologie. 1985 Jul-Sep;16(3):279-90.
- Lambrey B, Compagnon PL, Jacquot C. Pharmacokinetics of 14C-2-allophanyl-2-allyl -gamma-valero-lactone: a prodrug of proxibarbal in rats. European Journal of Drug Metabolism and Pharmacokinetics. 1981;6(3):161-9.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.