Elfazepam
Elfazepam[1] is a drug which is a benzodiazepine derivative.[2] Presumably it has sedative and anxiolytic actions like those of other benzodiazepines.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
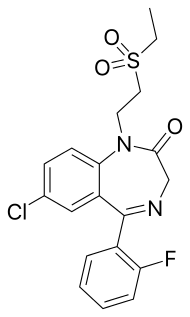
| Formula | C19H18ClFN2O3S |
| Molar mass | 408.87 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Orexigenic properties in animals.[3][4] The mechanism for increasing feed intake is not clear and has been subject of investigation.[5] It has been found that elfazepam suppresses gastric acid secretion.[6]
Synthesis
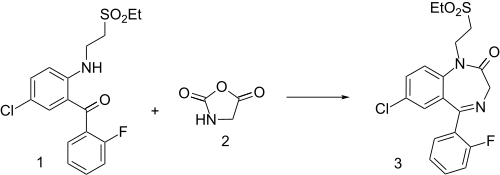
Elfazepam synthesis: U.S. Patent 4,010,154
Benzophenone derivative 1 is reacted with a glycine equivalent masked as an oxazolidine-2,5-dione 2 to give the final product 3 Elfazepam.
References
- U.S. Patent 4,010,154
- Psychotropics.dk
- Baile, CA.; McLaughlin, CL. (Nov 1979). "A review of the behavioral and physiological responses to elfazepam, a chemical feed intake stimulant". J Anim Sci. 49 (5): 1371–95. PMID 396294.
- Baile, CA.; Naylor, J.; McLaughlin, CL.; Catanzaro, CA. (Aug 1981). "Endotoxin-elicited fever and anorexia and elfazepam-stimulated feeding in sheep". Physiol Behav. 27 (2): 271–7. doi:10.1016/0031-9384(81)90269-9. PMID 7029576.
- Keim, DA.; Baile, CA.; Bolton, JR.; Wangsness, PJ.; Della Fera, MA. (Jan 1979). "Abomasal function following injections of elfazepam and 9-aza-cannabinol". Pharmacol Biochem Behav. 10 (1): 63–70. doi:10.1016/0091-3057(79)90170-9. PMID 35793.
- Van Den Broek, GW.; Robertson, J.; Keim, DA.; Baile, CA. (Jul 1979). "Feeding and depression of abomasal secretion in sheep elicited by elfazepam and 9-aza-cannabinol". Pharmacol Biochem Behav. 11 (1): 51–6. doi:10.1016/0091-3057(79)90296-x. PMID 493298.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.