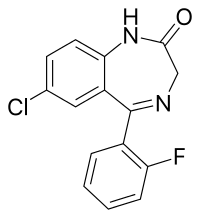
N-Desalkylflurazepam
N-Desalkylflurazepam (also known as norflurazepam) is a benzodiazepine analog and an active metabolite of several other benzodiazepine drugs including flurazepam,[2] flutoprazepam,[3] fludiazepam,[4] midazolam,[5] flutazolam[6], quazepam,[7] and ethyl loflazepate.[8][9] It is long-acting, prone to accumulation, and binds unselectively to the various benzodiazepine receptor subtypes.[7] It has been sold as a designer drug from 2016 onward.[10]
 | |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C15H10ClFN2O |
| Molar mass | 288.70 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 205 to 206 °C (401 to 403 °F) [1] |
SMILES
| |
Synonyms
- N-1-Desalkylflurazepam
- N-Desalkyl-2-oxoquazepam
- N-Desalkylflutoprazepam
- 7-Chloroflubromazepam
- Dealkylflurazepam
- Desalkylflurazepam
- Descarbethoxyloflazepate
- Norfludiazepam
- Norflurazepam
- Norflutoprazepam
- Ro 5-3367
- TL8002277
References
- SciFinder record for CAS#2886-65-9
- Riva, Roberto; De Anna, Marco; Albani, Fiorenzo; Baruzzi, Agostino (1981). "Rapid quantitation of flurazepam and its major metabolite, N-desalkylflurazepam, in human plasma by gas—liquid chromatography with electron-capture detection". Journal of Chromatography B. 222 (3): 491–495. doi:10.1016/S0378-4347(00)84153-5.
- Barzaghi, N; Leone, L; Monteleone, M; Tomasini, G; Perucca, E (1989). "Pharmacokinetics of flutoprazepam, a novel benzodiazepine drug, in normal subjects". European Journal of Drug Metabolism and Pharmacokinetics. 14 (4): 293–8. doi:10.1007/bf03190114. PMID 2633923.
- J. Descotes, ed. (1996). Human Toxicology. p. 43.
- Vogt, Susanne; Kempf, Jürgen; Buttler, Jürgen; Auwärter, Volker; Weinmann, Wolfgang (2013). "Desalkylflurazepam found in patients' samples after high-dose midazolam treatment". Drug Testing and Analysis. 5 (9–10): 745–7. doi:10.1002/dta.1484. PMID 23713025.
- Miyaguchi, Hajime; Kuwayama, Kenji; Tsujikawa, Kenji; Kanamori, Tatsuyuki; Iwata, Yuko T.; Inoue, Hiroyuki; Kishi, Tohru (10 February 2006). "A method for screening for various sedative-hypnotics in serum by liquid chromatography/single quadrupole mass spectrometry". Forensic Science International. 157 (1): 57–70. doi:10.1016/j.forsciint.2005.03.011. ISSN 0379-0738. PMID 15869852.
- Nikaido, AM; Ellinwood Jr, EH (1987). "Comparison of the effects of quazepam and triazolam on cognitive-neuromotor performance". Psychopharmacology. 92 (4): 459–64. doi:10.1007/bf00176478. PMID 2888152.
- B. B. Ba, A. Iliadis, J. P. Cano; Iliadis; Cano (1989). "Pharmacokinetic modeling of ethyl loflazepate (VictanR) and its main active metabolites". Annals of Biomedical Engineering. 17 (6): 633–646. doi:10.1007/bf02367467. PMID 2574017.CS1 maint: multiple names: authors list (link)
- Davi, H.; Guyonnet, J.; Necciari, J.; Cautreels, W. (1985). "Determination of circulating ethyl loflazepate metabolites in the baboon by radio-high-performance liquid chromatography with injection of crude plasma samples: Comparison with solvent extraction and thin-layer chromatography". Journal of Chromatography B. 342: 159–165. doi:10.1016/S0378-4347(00)84498-9.
- Manchester, Kieran R.; Maskell, Peter D.; Waters, Laura (2018). "Experimental versus theoretical log D7.4, pKa and plasma protein binding values for benzodiazepines appearing as new psychoactive substances". Drug Testing and Analysis. 10 (8): 1258–1269. doi:10.1002/dta.2387. ISSN 1942-7611. PMID 29582576.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.