Pregnenolone (medication)
Pregnenolone is a medication and supplement as well as a naturally occurring and endogenous steroid.[2][1][3][4][5] It is a neurosteroid and anti-inflammatory drug, and has been used historically in the treatment of rheumatoid arthritis.[2] Pregnenolone can be taken by mouth or as a topical medication.[2]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Arthenolone, Bina-Skin, Enelone, Natolone, Pregneton, Regnosone, Sharmone, Skinostelon[1] |
| Other names | P5; 5-Pregnenolone; δ5-Pregnene-3β-ol-20-one; Pregn-5-en-3β-ol-20-one; NSC-1616 |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth, transdermal |
| Drug class | Neurosteroid; Anti-inflammatory |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C21H32O2 |
| Molar mass | 316.485 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Pregnenolone is promoted online with false claims it can treat a variety of health conditions including cancer, arthritis and multiple sclerosis.[6]
Alternative medicine
Pregnenolone has been promoted online with claims it can treat a variety of diseases including multiple sclerosis, arthritis and cancer, but such claims are not backed by evidence.[6]
Pharmacology
Pregnenolone is a neurosteroid.[4][5] It is a negative allosteric modulator of the CB1 receptor,[4][7] a ligand of the microtubule-associated protein 2 (MAP2),[8][9] and an agonist of the pregnane X receptor.[10] Pregnenolone has no progestogenic, corticosteroid, estrogenic, androgenic, or antiandrogenic activity.[2] In addition to its own activities, pregnenolone is a precursor for other neurosteroids such as pregnenolone sulfate, allopregnanolone, and pregnanolone and for steroid hormones.[11][12][13][14]
Pregnenolone has low bioavailability and is subject to high metabolism.[4] Oral administration of 50 or 100 mg pregnenolone has been found to have minimal or negligible effect on urinary levels of testosterone and testosterone metabolites, including of androsterone, etiocholanolone, 5β-androstanediol, androstadienol, and androstenol (and/or their conjugates), and this suggests that only a small amount of pregnenolone is converted into testosterone.[12][13] This is in accordance with findings on the conversion of DHEA into testosterone, in which only 1.5% of an oral dose of DHEA was found to be converted into testosterone.[12] In contrast to the androstanes, 50 or 100 mg oral pregnenolone has been found to significantly and in fact "strongly" increase urinary levels of the progesterone metabolites pregnanediol and pregnanolone (and/or their conjugates), whereas pregnanetriol was unaffected.[12][13] Unlike the case of oral administration, transdermal administration of 30 mg/day pregnenolone cream has not been found to affect urinary levels of metabolites of any other steroids, including of progesterone.[13] Intranasal administration of pregnenolone was found to have low bioavailability of around 23%.[4]
Sripada et al. reported that oral pregnenolone is preferentially metabolized into the neurosteroid allopregnanolone rather than into other steroids such as DHEA or cortisol.[14] In further research by their group, a single 400 mg dose of oral pregnenolone at 3 hours post-administration was found to result in a 3-fold elevation in serum levels of pregnenolone and a 7-fold increase in allopregnanolone levels.[14] Pregnanolone levels increased by approximately 60% while DHEA levels decreased non-significantly by approximately 5% and cortisol levels were not affected.[14] Another study found that allopregnanolone levels were increased by 3-fold at 2 hours post-administration following a single 400 mg oral dose of pregnenolone.[14]
In addition to allopregnanolone, pregnenolone acts as a prodrug of pregnenolone sulfate.[11] However, pregnenolone sulfate does not cross the blood–brain barrier.[15][16]
Chemistry
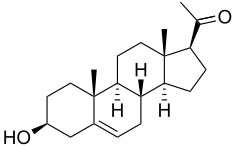
Pregnenolone, also known as 5-pregnenolone or as pregn-5-en-3β-ol-20-one, is a naturally occurring pregnane steroid and a derivative of cholesterol.[2][1][3] Related steroids include pregnenolone sulfate, 3β-dihydroprogesterone (4-pregnenolone), progesterone, allopregnanolone, and pregnanolone.[2][1][3]
Derivatives
A few synthetic ester derivatives of pregnenolone exist.[1] These include pregnenolone acetate (Antofin, Previsone, Pregno-Pan) and pregnenolone succinate (Panzalone, Formula 405).[1] Prebediolone acetate (Acetoxanon, Acetoxy-Prenolon, Artisone, Artivis, Pregnartrone, Sterosone), the 21-acetate ester of 21-hydroxypregnenolone, also exists.[1] These esters are all described as glucocorticoids similarly to pregnenolone.[1]
The 3β-methyl ether of pregnenolone, 3β-methoxypregnenolone (MAP-4343), retains similar activity to pregnenolone in regard to interaction with MAP2,[8][9] and is under development for potential clinical use for indications such as the treatment of brain and spinal cord injury and depressive disorders.[17][18][19][20]
History
Pregnenolone was first synthesized by Adolf Butenandt and colleagues in 1934.[2] It was first used in medicine, as an anti-inflammatory medication, in the 1940s.[4]
Society and culture
Research directions
As of 2016 pregnenolone is being researched for possible therapeutic applications, but its poor bioavailability makes its prospects for usefulness low.[4] Pregnenolone is available as an over-the-counter supplement, for instance in the United States.[22]
References
- J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 665–. ISBN 978-1-4757-2085-3.
- Henderson E, Weinberg M, Wright WA (April 1950). "Pregnenolone". J. Clin. Endocrinol. Metab. 10 (4): 455–74. doi:10.1210/jcem-10-4-455. PMID 15415436.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 872–873. ISBN 978-3-88763-075-1.
- Vallée M (June 2016). "Neurosteroids and potential therapeutics: Focus on pregnenolone". J. Steroid Biochem. Mol. Biol. 160: 78–87. doi:10.1016/j.jsbmb.2015.09.030. PMID 26433186.
- Weng JH, Chung BC (July 2016). "Nongenomic actions of neurosteroid pregnenolone and its metabolites". Steroids. 111: 54–59. doi:10.1016/j.steroids.2016.01.017. PMID 26844377.
- Russell J, Rovere A, eds. (2009). "Pregnenolone". American Cancer Society Complete Guide to Complementary and Alternative Cancer Therapies (2nd ed.). American Cancer Society. pp. 807-810. ISBN 9780944235713.CS1 maint: uses editors parameter (link)
- Pertwee, Roger G. (2015), "Endocannabinoids and Their Pharmacological Actions", Endocannabinoids, Handbook of Experimental Pharmacology, 231, Springer International Publishing, pp. 1–37, doi:10.1007/978-3-319-20825-1_1, ISBN 9783319208244, PMID 26408156
- Mellon SH (2007). "Neurosteroid regulation of central nervous system development". Pharmacol. Ther. 116 (1): 107–24. doi:10.1016/j.pharmthera.2007.04.011. PMC 2386997. PMID 17651807.
- Fontaine-Lenoir V, Chambraud B, Fellous A, David S, Duchossoy Y, Baulieu EE, Robel P (2006). "Microtubule-associated protein 2 (MAP2) is a neurosteroid receptor". Proc. Natl. Acad. Sci. U.S.A. 103 (12): 4711–6. doi:10.1073/pnas.0600113103. PMC 1450236. PMID 16537405.
- Kliewer SA, Lehmann JM, Milburn MV, Willson TM (1999). "The PPARs and PXRs: nuclear xenobiotic receptors that define novel hormone signaling pathways". Recent Prog. Horm. Res. 54: 345–67, discussion 367–8. PMID 10548883.
- Ducharme N, Banks WA, Morley JE, Robinson SM, Niehoff ML, Mattern C, Farr SA (2010). "Brain distribution and behavioral effects of progesterone and pregnenolone after intranasal or intravenous administration". Eur. J. Pharmacol. 641 (2–3): 128–34. doi:10.1016/j.ejphar.2010.05.033. PMC 3008321. PMID 20570588.
- Saudan C, Desmarchelier A, Sottas PE, Mangin P, Saugy M (2005). "Urinary marker of oral pregnenolone administration". Steroids. 70 (3): 179–83. doi:10.1016/j.steroids.2004.12.007. PMID 15763596.
- Piper T, Schlug C, Mareck U, Schänzer W (2011). "Investigations on changes in ¹³C/¹²C ratios of endogenous urinary steroids after pregnenolone administration". Drug Test Anal. 3 (5): 283–90. doi:10.1002/dta.281. PMID 21538944.
- Sripada RK, Marx CE, King AP, Rampton JC, Ho SS, Liberzon I (2013). "Allopregnanolone elevations following pregnenolone administration are associated with enhanced activation of emotion regulation neurocircuits". Biol. Psychiatry. 73 (11): 1045–53. doi:10.1016/j.biopsych.2012.12.008. PMC 3648625. PMID 23348009.
- Jong Rho; Raman Sankar; Carl E. Stafstrom (18 June 2010). Epilepsy: Mechanisms, Models, and Translational Perspectives. CRC Press. pp. 479–. ISBN 978-1-4200-8560-0.
- CIBA Foundation Symposium (30 April 2008). Steroids and Neuronal Activity. John Wiley & Sons. pp. 101–. ISBN 978-0-470-51399-6.
- "Pregnenolone methyl ether - AdisInsight".
- Duchossoy Y, David S, Baulieu EE, Robel P (2011). "Treatment of experimental spinal cord injury with 3β-methoxy-pregnenolone". Brain Res. 1403: 57–66. doi:10.1016/j.brainres.2011.05.065. PMID 21704982.
- Bianchi M, Baulieu EE (2012). "3β-Methoxy-pregnenolone (MAP4343) as an innovative therapeutic approach for depressive disorders". Proc. Natl. Acad. Sci. U.S.A. 109 (5): 1713–8. doi:10.1073/pnas.1121485109. PMC 3277154. PMID 22307636.
- Baulieu ÉÉ (2015). "From steroid hormones to depressive states and senile dementias: New mechanistic, therapeutical and predictive approaches". Comptes Rendus Biologies. 338 (8–9): 613–6. doi:10.1016/j.crvi.2015.06.003. PMID 26251072.
- "ChemIDplus - 145-13-1 - ORNBQBCIOKFOEO-QGVNFLHTSA-N - Pregnenolone [INN:BAN] - Similar structures search, synonyms, formulas, resource links, and other chemical information".
- Meieran SE, Reus VI, Webster R, Shafton R, Wolkowitz OM (May 2004). "Chronic pregnenolone effects in normal humans: attenuation of benzodiazepine-induced sedation". Psychoneuroendocrinology. 29 (4): 486–500. doi:10.1016/S0306-4530(03)00056-8. PMID 14749094.