Fluticasone furoate
Fluticasone furoate is a corticosteroid for the treatment of non-allergic and allergic rhinitis administered by a nasal spray.[1] It is also available as an inhaled corticosteroid to help prevent and control symptoms of asthma. It is derived from cortisol.[2] Unlike fluticasone propionate, which is only approved for children 4 years and older, fluticasone furoate is approved in children as young as 2 years of age when used for allergies. [3]
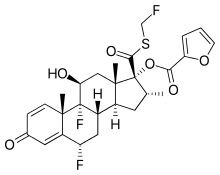 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| License data | |
| Pregnancy category |
|
| Routes of administration | Intranasal |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 0.51% (Intranasal) |
| Protein binding | 91% |
| Metabolism | Intranasal Hepatic (CYP3A4-mediated) |
| Elimination half-life | 15 hours |
| Excretion | Renal |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.158.130 |
| Chemical and physical data | |
| Formula | C27H29F3O6S |
| Molar mass | 538.576 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
It was approved by the FDA in 2007 and first marketed in the US in 2009.
Brand names
In the US it is marketed by GlaxoSmithKline for asthma as Arnuity Ellipta and is only available with a prescription.[4] It is marketed over-the-counter for allergic rhinitis as Flonase Sensimist.[5] The Veramyst brand name has been discontinued in the U.S. It is also marketed as Allermist (Japan, アラミスト) and Avamys (Australia, Canada, EU, South Africa, South America, Mexico, Israel, Italy, India and South Korea).
The combination drug fluticasone furoate/vilanterol, marketed as Breo Ellipta (US, Canada) and Relvar Ellipta (UK), is approved for use in the United States for long-term maintenance treatment of airflow obstruction in people with COPD.[6] It is also approved for the treatment of asthma.[7]
References
- Bruni FM, De Luca G, Venturoli V, Boner AL (2009). "Intranasal corticosteroids and adrenal suppression". Neuroimmunomodulation. 16 (5): 353–62. doi:10.1159/000216193. PMID 19571596.
- Kaliner, Michael A. (2011). Rhinitis, An Issue of Immunology and Allergy Clinics - E-Book. Elsevier Health Sciences. ISBN 9781455709328.
- https://www.drugs.com/pro/veramyst.html. Missing or empty
|title=(help) - https://www.arnuity.com/about-arnuity.html. Missing or empty
|title=(help) - https://www.flonase.com/products/childrens-sensimist/. Missing or empty
|title=(help) - "FDA approves Breo Ellipta to treat chronic obstructive pulmonary disease". Food and Drug Administration. May 10, 2013.
- "BREO® ELLIPTA® 100/25 (fluticasone furoate 100 mcg and vilanterol 25 mcg inhalation powder)" (PDF).