Chloromorphide
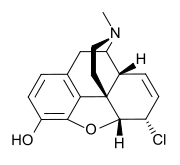
Chloromorphide (α-chloromorphide) is an opiate analog that is a derivative of morphine, where the 6-hydroxy group has been replaced by chlorine. Developed in 1933 in Germany, it has approximately ten times the potency of morphine.[1] It has similar effects to morphine, such as sedation, analgesia, and respiratory depression.
 | |
| Clinical data | |
|---|---|
| Other names | α-Chloromorphide |
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C17H18ClNO2 |
| Molar mass | 303.78 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Chloromorphide does not appear specifically in the Controlled Substances Act 1970 in the United States, but is presumably Schedule II controlled substance as a form of morphine or an analogue of morphine or morphinan. When halogenated morphides and codides are used for research or industrial uses, they are often synthesised on-site.
Chloromorphide is one of a series of opioids known as morphides and codides, which are important precursors and intermediates in the synthesis of semi-synthetic opioid analgesic drugs, especially those with additions, substitutions, or other modifications at the 7, 8, and/or 14 positions on the morphine carbon skeleton. Semisynthetics with changes at other positions can also be made from these compounds. The codeine analog of chloromorphide is α-chlorocodide (alphachlorcodide), an intermediate in one method of desomorphine synthesis which uses codeine as precursor.
During the 1930s, the entire series of alpha- and beta-halogenated codides, morphides, dihydromorphides, and dihydrocodides were produced and described, and α-bromomorphide and α-iodomorphide are sometimes currently used in research and manufacturing.
References
- Yeh HJC, Wilson RS, Klee WA, Jacobson AE (1976). "Alpha-and beta-halomorphides: Stereochemistry, analgesic potency, toxicity, and interaction with narcotic receptors in vitro". J Pharm Sci. 65 (6): 902–4. doi:10.1002/jps.2600650624. PMID 932978.