Rimegepant
Rimegepant (BHV-3000; formerly BMS-927711) is an investigational drug candidate for the treatment of migraine. Originally discovered at Bristol-Myers Squibb,[1] it is currently under development by Biohaven Pharmaceuticals.[2]
 | |
| Clinical data | |
|---|---|
| Other names | BHV-3000, BMS-927711 |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
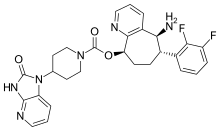
| Formula | C28H28F2N6O3 |
| Molar mass | 534.568 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Mechanism of action
Rimegepant is a small molecule calcitonin gene-related peptide (CGRP) receptor antagonist.[3]
References
- "Drug Profile: Rimegepant". Adis Insight.
- "Rimegepant (BHV-3000) – for acute treatment of Migraine". Biohaven Pharmaceuticals.
- Diener, Hans-Christoph; Charles, Andrew; Goadsby, Peter J; Holle, Dagny (2015). "New therapeutic approaches for the prevention and treatment of migraine". The Lancet Neurology. 14 (10): 1010. doi:10.1016/S1474-4422(15)00198-2. PMID 26376968.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.