Pazopanib
Pazopanib (trade name Votrient) is a potent and selective multi-targeted receptor tyrosine kinase inhibitor that blocks tumour growth and inhibits angiogenesis. It has been approved for renal cell carcinoma and soft tissue sarcoma by numerous regulatory administrations worldwide.[3][4][5][6]
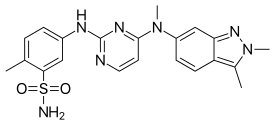 | |
| Clinical data | |
|---|---|
| Pronunciation | /pæˈzoʊpənɪb/ paz-OH-pə-nib |
| Trade names | Votrient |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610013 |
| License data | |
| Pregnancy category | |
| Routes of administration | By mouth (tablets) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 14–21%[1] |
| Protein binding | >99.5%[1][2] |
| Metabolism | Liver: CYP3A4 (major), 1A2 and 2C8 (minor)[2] |
| Elimination half-life | 30.9–31.9 hours[2] |
| Excretion | Faeces (primary), urine (<4%)[2] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C21H23N7O2S |
| Molar mass | 437.517 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Medical uses
It is approved by numerous regulatory administrations worldwide (including the FDA (19 October 2009), EMA (14 June 2010), MHRA (14 June 2010) and TGA (30 June 2010)) for use as a treatment for advanced/metastatic renal cell carcinoma and advanced soft tissue sarcomas.[2][3][4][5][6] In Australia and New Zealand, it is subsidised under the PBS and by Pharmac respectively, under a number of conditions, including:[7][8]
- The medication is used to treat clear cell variant renal cell carcinoma.
- The treatment phase is continuing treatment beyond 3 months.
- The patient has been issued an authority prescription for pazopanib
- The patient must have stable or responding disease according to the Response Evaluation Criteria In Solid Tumours (RECIST)
- This treatment must be the sole tyrosine kinase inhibitor subsidised for this condition.
It has also demonstrated initial therapeutic properties in patients with ovarian and non-small cell lung cancer,[9] though plans to apply to the EMA for a variation to include advanced ovarian cancer have been withdrawn and a license will not be sought in any country.[10][11]
Contraindications
The only contraindication is hypersensitivity to pazopanib or any of its excipients.[5] Cautions include:[2]
- Hypertension, including hypertensive crises reported
- QT interval prolongation and torsades de pointes reported.
- Thrombotic microangiopathy reported
- Thrombotic thrombocytopenic purpura reported
- Haemolytic uremic syndrome reported
- Haematologic parameter alterations reported in 31-37% of patients.
- Events of cardiac dysfunction (decreased LVEF and congestive heart failure) have been observed
- Fatal haemorrhage, arterial and venous thrombotic events and GI perforation have been observed in randomized clinical trials.
It has one black box warning by the US FDA, severe hepatotoxicity, including fatalities.[2]
Adverse effects
The most common side effects of pazopanib are nausea, vomiting, diarrhoea (occurs in about half of patients), changes in hair colour, hypertension (which usually occurs during the first few weeks of treatment), appetite loss, hyperglycaemia, hypoglycaemia, electrolyte abnormalities (including hypocalcaemia, hypomagnesemia, hypophosphatemia), lab anomalies (including increased AST, ALT and protein in the urine), oedema, hair loss or discolouration, taste changes, abdominal pain, rash, fatigue and myelosuppression (including leucopenia, neutropenia, thrombocytopenia and lymphopenia).[12] It has been associated with a low, but real risk of potentially fatal liver damage.[12]
Overdose
The treatment for overdose is purely supportive and the symptoms include grade 3 hypertension and fatigue.[5]
Interactions
Drug interactions include:[2]
- Coadministration with strong CYP3A4 inhibitors (e.g. ketoconazole, ritonavir, clarithromycin, grapefruit juice) may increase pazopanib serum levels as it is a CYP3A4 substrate.
- CYP3A4 inducers (e.g. rifampin, carbamazepine) decrease pazopanib serum levels.
- It is a p-glycoprotein (PGP) substrate and hence PGP inhibitors such as quinidine may interact with pazopanib.
- Pazopanib is not a substrate for either of the hepatic OATP1B1 and OATP1B3.[13]
- Pazopanib have inhibitory potency towards OATP-1B1 but not for OATP-1B3.[14]
Mechanism of action
Pazopanib is a multikinase inhibitor that limits tumor growth by targeting angiogenesis via inhibition of enzymes including vascular endothelial growth factor receptor (VEGFR), platelet-derived growth factory receptor (PDGFR), c-KIT and FGFR.[2][12][15][16][17][18]
References
- "CHMP Assessment Report: Votrient (pazopanib)" (PDF). European Medicines Agency. Retrieved 8 October 2016.
- "Votrient (pazopanib) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 27 January 2014.
- "VOTRIENT (pazopanib hydrochloride) tablet, film coated [GlaxoSmithKline LLC]" (PDF). DailyMed. GlaxoSmithKline LLC. November 2013. Retrieved 27 January 2014.
- "Votrient : EPAR - Product Information" (PDF). European Medicines Agency. Glaxo Group Ltd. 23 January 2014. Retrieved 27 January 2014.
- "Votrient 200 mg and 400 mg film coated tablets - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. GlaxoSmithKline UK. 20 December 2013. Retrieved 27 January 2014.
- "PRODUCT INFORMATION VOTRIENT® TABLETS" (PDF). TGA eBusiness Services. GlaxoSmithKline Australia Pty Ltd. 25 March 2013. Retrieved 27 January 2014.
- "Pharmaceutical Benefits Scheme (PBS) - Pazopanib". Pharmaceutical Benefits Scheme. Australian Government. Retrieved 27 January 2014.
- "Pazopanib - Online Pharmaceutical Schedule". Pharmaceutical Management Agency. Retrieved 9 June 2015.
- "Pazopanib shows encouraging activity in several tumour types, including soft tissue sarcoma and ovarian cancer". FierceBiotech. 2008-09-15. Retrieved 2010-08-10.
- "GSK pulls bid to extend use of kidney drug to ovarian cancer". Reuters. 31 March 2014. Retrieved 7 April 2014.
- "Regulatory update: Votrient (pazopanib) as maintenance therapy for advanced ovarian cancer in the EU". GlaxoSmithKline. 31 March 2014. Archived from the original on 8 April 2014. Retrieved 7 April 2014.
- Zivi, A; Cerbone, L; Recine, F; Sternberg, CN (September 2012). "Safety and tolerability of pazopanib in the treatment of renal cell carcinoma". Expert Opinion on Drug Safety. 11 (5): 851–859. doi:10.1517/14740338.2012.712108. PMID 22861374.
- Khurana V, Minocha M, Pal D, Mitra AK (March 2014). "Role of OATP-1B1 and/or OATP-1B3 in hepatic disposition of tyrosine kinase inhibitors". Drug Metabol Drug Interact. 29 (3): 1–11. doi:10.1515/dmdi-2013-0062. PMC 4407685. PMID 24643910.
- Khurana V, Minocha M, Pal D, Mitra AK (May 2014). "Inhibition of OATP-1B1 and OATP-1B3 by tyrosine kinase inhibitors". Drug Metabol Drug Interact. 29 (4): 1–11. doi:10.1515/dmdi-2014-0014. PMC 4407688. PMID 24807167.
- Verweij, J; Sleijfer, S (May 2013). "Pazopanib, a new therapy for metastatic soft tissue sarcoma". Expert Opinion on Pharmacotherapy. 14 (7): 929–935. doi:10.1517/14656566.2013.780030. PMID 23488774.
- Schöffski, P (June 2012). "Pazopanib in the treatment of soft tissue sarcoma". Expert Review of Anticancer Therapy. 12 (6): 711–723. doi:10.1586/era.12.41. PMID 22716487.
- Pick, AM; Nystrom, KK (March 2012). "Pazopanib for the treatment of metastatic renal cell carcinoma". Clinical Therapeutics. 34 (3): 511–520. doi:10.1016/j.clinthera.2012.01.014. PMID 22341567.
- Rimel, BJ (April 2015). "Antiangiogenesis agents in ovarian cancer". Contemporary Oncology. 7 (2): 16–19.