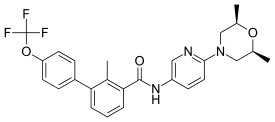
Sonidegib
Sonidegib (INN; also known as LDE225 and erismodegib; trade name Odomzo[3]) is a Hedgehog signaling pathway inhibitor (via smoothened antagonism) being developed as an anticancer agent by Novartis.[4][5]
 | |
| Clinical data | |
|---|---|
| Trade names | Odomzo[1] |
| AHFS/Drugs.com | Odomzo |
| License data | |
| Routes of administration | By mouth (capsules) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | <10% |
| Protein binding | >97% |
| Metabolism | Liver (CYP3A) |
| Elimination half-life | ~28 days |
| Excretion | Feces (~70%), urine (30%)[2] |
| Identifiers | |
IUPAC name
| |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| Chemical and physical data | |
| Formula | C26H26F3N3O3 |
| Molar mass | 485.498 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Approvals and indications
It was approved by the FDA for treating basal-cell carcinoma in July 2015[6][3] and is awaiting approval in the EU. In the EU, the agent has received positive opinion from the CHMP for approval.
It is indicated for the treatment of adult patients with locally advanced basal-cell carcinoma that has recurred following surgery or radiation therapy, or those who are not candidates for surgery or radiation therapy.[2]
Pharmacology
Sonidegib is administered orally. Common side effects include muscle spasms, hair loss, fatigue, abdominal pain, nausea, headache, and weight loss.[7]
Sonidegib binds to and inhibits smoothened to inhibit activation of the Hedgehog pathway. Sonidegib is primarily metabolized by CYP3A and is eliminated hepatically.[8]
Development
It has been investigated as a potential treatment for:
- Pancreatic cancer[9][10][11][12]
- Breast cancer[13][14]
- Basal cell carcinoma of the skin[15][16][17]
- Small cell lung cancer[18]
- Medulloblastoma[19][20]
- Advanced solid tumors (including ovarian, breast, pancreatic, stomach, oesophageal cancers and glioblastoma multiforme)[21][22][23]
- Acute leukemia[24] and chronic myeloid leukemia[25]
- Myelofibrosis and essential thrombocythaemia[26]
It has demonstrated significant efficacy against melanoma in vitro and in vivo.[27] It also demonstrated efficacy in a mouse model of pancreatic cancer.[28]
Reference list
- FDA Approves Odomzo (sonidegib) for Locally Advanced Basal Cell Carcinoma
- "Odomzo (sonidegib) Capsules, for Oral Use. Full Prescribing Information" (PDF). Novartis Pharmaceuticals Corporation, East Hanover, New Jersey 07936. Retrieved 2 October 2016.
- "FDA approves Novartis's advanced skin cancer drug". Retrieved 2015-07-24.
- "LDE225 - PubChem". PubChem. National Institutes of Health. Retrieved 16 February 2014.
- Pan, S; Wu, X; Jiang, J; Gao, W; Wan, Y; Cheng, D; Han, D; Liu, J; Englund, NP; Wang, Y; Peukert, S; Miller-Moslin, K; Yuan, J; Guo, R; Matsumoto, M; Vattay, A; Jiang, Y; Tsao, J; Sun, F; Pferdekamper, AC; Dodd, S; Tuntland, T; Maniara, W; Kelleher, JF; Yao, Y; Warmuth, M; Williams, J; Dorsch, M (10 June 2010). "Discovery of NVP-LDE225, a Potent and Selective Smoothened Antagonist". ACS Medicinal Chemistry Letters. 1 (3): 130–134. doi:10.1021/ml1000307. PMC 4007689. PMID 24900187.
- "FDA approves new treatment for most common form of advanced skin cancer". www.fda.gov. Retrieved 2015-07-24.
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/205266s000lbl.pdf
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/205266s000lbl.pdf
- "A Biomarker Study to Identify Predictive Signatures of Response to LDE225 (Hedgehog Inhibitor) In Patients With Resectable Pancreatic Cancer". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Gemcitabine + Nab-paclitaxel With LDE-225 (Hedgehog Inhibitor) as Neoadjuvant Therapy for Pancreatic Adenocarcinoma". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Dose-escalation, and Safety Study of LDE225 and Gemcitabine in Locally Advanced or Metastatic Pancreatic Cancer Patients". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "A Pilot Study of a Hedgehog Pathway Inhibitor (LDE-225) in Surgically Resectable Pancreas Cancer". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Study With LDE225 in Combination With Docetaxel in Triple Negative (TN) Advanced Breast Cancer (ABC) Patients (EDALINE)". ClinicalTrials.gov. National Institutes of Health. 13 February 2014.
- "LDE225 in Treating Patients With Stage II-III Estrogen Receptor- and HER2-Negative Breast Cancer". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "A Phase II Study of Efficacy and Safety in Patients With Locally Advanced or Metastatic Basal Cell Carcinoma (BOLT)". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "To Evaluate the Safety, Local Tolerability, PK and PD of LDE225 on Sporadic Superficial and Nodular Skin Basal Cell Carcinomas(sBCC)". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "A Trial to Evaluate the Safety, Local Tolerability, Pharmacokinetics and Pharmacodynamics of LDE225 on Skin Basal Cell Carcinomas in Gorlin Syndrome Patients". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Combination of the Hedgehog Inhibitor, LDE225, With Etoposide and Cisplatin in the First-Line Treatment of Patients With Extensive Stage Small Cell Lung Cancer (ES-SCLC)". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "A Phase III Study of Oral LDE225 Versus (vs) Temozolomide (TMZ) in Patients With Hedge-Hog (Hh)-Pathway Activated Relapsed Medulloblastoma (MB)". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "A Phase I Dose Finding and Safety Study of Oral LDE225 in Children and a Phase II Portion to Assess Preliminary Efficacy in Recurrent or Refractory MB". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Phase Ib, Dose Escalation Study of Oral LDE225 in Combination With BKM120 in Patients With Advanced Solid Tumors". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Dose Finding and Safety of Oral LDE225 in Patients With Advanced Solid Tumors". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "LDE225 and Paclitaxel in Solid Tumors". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Study of Efficacy and Safety of LDE225 in Adult Patients With Relapsed/Refractory Acute Leukemia". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "Nilotinib and LDE225 in the Treatment of Chronic or Accelerated Phase Myeloid Leukemia in Patients Who Developed Resistance to Prior Therapy". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- "A Phase Ib/II Dose-finding Study to Assess the Safety and Efficacy of LDE225 + INC424 in Patients With MF". ClinicalTrials.gov. National Institutes of Health. 13 February 2014. Retrieved 16 February 2014.
- Jalili, A; Mertz, KD; Romanov, J; Wagner, C; Kalthoff, F; Stuetz, A; Pathria, G; Gschaider, M; Stingl, G; Wagner, SN (30 July 2013). "NVP-LDE225, a potent and selective SMOOTHENED antagonist reduces melanoma growth in vitro and in vivo". PLoS ONE. 8 (7): e69064. Bibcode:2013PLoSO...869064J. doi:10.1371/journal.pone.0069064. PMC 3728309. PMID 23935925.
- Fendrich, V; Wiese, D; Waldmann, J; Lauth, M; Heverhagen, AE; Rehm, J; Bartsch, DK (November 2011). "Hedgehog inhibition with the orally bioavailable Smo antagonist LDE225 represses tumor growth and prolongs survival in a transgenic mouse model of islet cell neoplasms". Annals of Surgery. 254 (5): 818–23. doi:10.1097/SLA.0b013e318236bc0f. PMID 22042473.