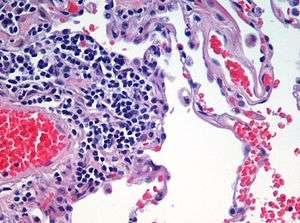
Histology
Histology,[help 1] also known as microscopic anatomy or microanatomy,[1] is the branch of biology which studies the microscopic anatomy of biological tissues.[2][3][4][5] Histology is the microscopic counterpart to gross anatomy, which looks at larger structures visible without a microscope.[5][6] Although one may divide microscopic anatomy into organology, the study of organs, histology, the study of tissues, and cytology, the study of cells, modern usage places these topics under the field of histology.[5] In medicine, histopathology is the branch of histology that includes the microscopic identification and study of diseased tissue.[5][6] In the field of paleontology, the term paleohistology refers to the histology of fossil organisms.[7][8]
| Look up histology in Wiktionary, the free dictionary. |

Biological tissues
Animal tissue classification
There are four basic types of animal tissues: muscle tissue, nervous tissue, connective tissue, and epithelial tissue.[5][9] All animal tissues are considered to be subtypes of these four principal tissue types (for example, blood is classified as connective tissue, since the blood cells are suspended in an extracellular matrix, the plasma).[9]
- Epithelium
- Muscle tissue
- Connective tissue
- General connective tissue
- Special connective tissue
- Nervous tissue
- Central nervous system
- Peripheral nervous system
- Special receptors
Plant tissue classification
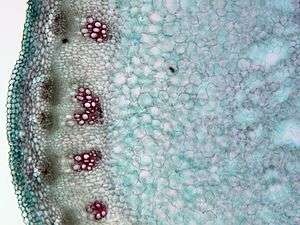
For plants, the study of their tissues falls under the field of plant anatomy, with the following four main types:
- Dermal tissue
- Vascular tissue
- Ground tissue
- Meristematic tissue
Medical histology
Histopathology is the branch of histology that includes the microscopic identification and study of diseased tissue.[5][6] It is an important part of anatomical pathology and surgical pathology, as accurate diagnosis of cancer and other diseases often requires histopathological examination of tissue samples.[10] Trained physicians, frequently licensed pathologists, perform histopathological examination and provide diagnostic information based on their observations.
Occupations
The field of histology that includes the preparation of tissues for microscopic examination is known as histotechnology. Job titles for the trained personnel who prepare histological specimens for examination are numerous and include histotechnicians, histotechnologists,[11] histology technicians and technologists, medical laboratory technicians, and biomedical scientists.
Sample preparation
Most histological samples need preparation before microscopic observation; these methods depend on the specimen and method of observation.[9]
Fixation

Chemical fixatives are used to preserve and maintain the structure of tissues and cells; fixation also hardens tissues which aids in cutting the thin sections of tissue needed for observation under the microscope.[5][12] Fixatives generally preserve tissues (and cells) by irreversibly cross-linking proteins.[12] The most widely used fixative for light microscopy is 10% neutral buffered formalin, or NBF (4% formaldehyde in phosphate buffered saline).[13][12][9]
For electron microscopy, the most commonly used fixative is glutaraldehyde, usually as a 2.5% solution in phosphate buffered saline.[9] Other fixatives used for electron microscopy are osmium tetroxide or uranyl acetate.[9]
The main action of these aldehyde fixatives is to cross-link amino groups in proteins through the formation of methylene bridges (-CH2-), in the case of formaldehyde, or by C5H10 cross-links in the case of glutaraldehyde. This process, while preserving the structural integrity of the cells and tissue can damage the biological functionality of proteins, particularly enzymes.
Formalin fixation leads to degradation of mRNA, miRNA, and DNA as well as denaturation and modification of proteins in tissues. However, extraction and analysis of nucleic acids and proteins from formalin-fixed, paraffin-embedded tissues is possible using appropriate protocols.[14][15]
Selection and trimming
Selection is the choice of relevant tissue in cases where it is not necessary to put the entire original tissue mass through further processing. The remainder may remain fixated in case it needs to be examined at a later time.
Trimming is the cutting of tissue samples in order to expose the relevant surfaces for later sectioning. It also creates tissue samples of appropriate size to fit into cassettes.[16]
Embedding
Tissues are embedded in a harder medium both as a support and to allow the cutting of thin tissue slices.[9][5] In general, water must first be removed from tissues (dehydration) and replaced with a medium that either solidifies directly, or with an intermediary fluid (clearing) that is miscible with the embedding media.[12]
Paraffin wax

For light microscopy, paraffin wax is the most frequently used embedding material.[12][13] Paraffin is immiscible with water, the main constituent of biological tissue, so it must first be removed in a series of dehydration steps.[12] Samples are transferred through a series of progressively more concentrated ethanol baths, up to 100% ethanol to remove remaining traces of water.[9][12] Dehydration is followed by a clearing agent (typically xylene[13] although other environmental safe substitutes are in use[13]) which removes the alcohol and is miscible with the wax, finally melted paraffin wax is added to replace the xylene and infiltrate the tissue.[9] In most histology, or histopathology laboratories the dehydration, clearing, and wax infiltration are carried out in tissue processors which automate this process.[13] Once infiltrated in paraffin, tissues are oriented in molds which are filled with wax; once positioned, the wax is cooled, solidifying the block and tissue.[13][12]
Other materials
Paraffin wax does not always provide a sufficiently hard matrix for cutting very thin sections (which are especially important for electron microscopy).[12] Paraffin wax may also be too soft in relation to the tissue, the heat of the melted wax may alter the tissue in undesirable ways, or the dehydrating or clearing chemicals may harm the tissue.[12] Alternatives to paraffin wax include, epoxy, acrylic, agar, gelatin, celloidin, and other types of waxes.[12][17]
In electron microscopy epoxy resins are the most commonly employed embedding media,[9] but acrylic resins are also used, particularly where immunohistochemistry is required.
For tissues to be cut in a frozen state, tissues are placed in a water-based embedding medium. Pre-frozen tissues are placed into molds with the liquid embedding material, usually a water-based glycol, OCT, TBS, Cryogel, or resin, which is then frozen to form hardened blocks.
Sectioning

For light microscopy, a knife mounted in a microtome is used to cut tissue sections (typically between 5-15 micrometers thick) which are mounted on a glass microscope slide.[9] For transmission electron microscopy (TEM), a diamond or glass knife mounted in an ultramicrotome is used to cut between 50-150 nanometer thick tissue sections.[9]
Staining
Biological tissue has little inherent contrast in either the light or electron microscope.[17] Staining is employed to give both contrast to the tissue as well as highlighting particular features of interest. When the stain is used to target a specific chemical component of the tissue (and not the general structure), the term histochemistry is used.[9]
Light microscopy
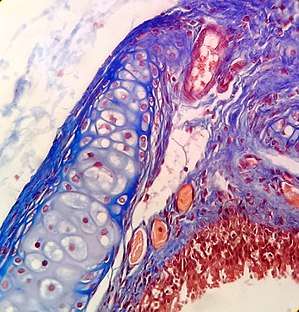
Hematoxylin and eosin (H&E stain) is one of the most commonly used stains in histology to show the general structure of the tissue.[9][18] Hematoxylin stains cell nuclei blue; eosin, an acidic dye, stains the cytoplasm and other tissues in different stains of pink.[9][12]
In contrast to H&E, which is used as a general stain, there are many techniques that more selectively stain cells, cellular components, and specific substances.[12] A commonly performed histochemical technique that targets a specific chemical is the Perls' Prussian blue reaction, used to demonstrate iron deposits[12] in diseases like hemochromatosis. The Nissl method for Nissl substance and Golgi's method (and related silver stains) are useful in identifying neurons are other examples of more specific stains.[12]
Historadiography
In historadiography, a slide (sometimes stained histochemically) is X-rayed. More commonly, autoradiography is used to visualize the locations to which a radioactive substance has been transported within the body, such as cells in S phase (undergoing DNA replication) which incorporate tritiated thymidine, or sites to which radiolabeled nucleic acid probes bind in in situ hybridization. For autoradiography on a microscopic level, the slide is typically dipped into liquid nuclear tract emulsion, which dries to form the exposure film. Individual silver grains in the film are visualized with dark field microscopy.
Immunohistochemistry
Recently, antibodies have been used to specifically visualize proteins, carbohydrates, and lipids. This process is called immunohistochemistry, or when the stain is a fluorescent molecule, immunofluorescence. This technique has greatly increased the ability to identify categories of cells under a microscope. Other advanced techniques, such as nonradioactive in situ hybridization, can be combined with immunochemistry to identify specific DNA or RNA molecules with fluorescent probes or tags that can be used for immunofluorescence and enzyme-linked fluorescence amplification (especially alkaline phosphatase and tyramide signal amplification). Fluorescence microscopy and confocal microscopy are used to detect fluorescent signals with good intracellular detail.
Electron microscopy
For electron microscopy heavy metals are typically used to stain tissue sections.[9] Uranyl acetate and lead citrate are commonly used to impart contrast to tissue in the electron microscope.[9]
Specialized techniques
Cryosectioning
Similar to the frozen section procedure employed in medicine, cryosectioning is a method to rapidly freeze, cut, and mount sections of tissue for histology. The tissue is usually sectioned on a cryostat or freezing microtome.[12] The frozen sections are mounted on a glass slide and may be stained to enhance the contrast between different tissues. Unfixed frozen sections can be used for studies requiring enzyme localization in tissues and cells. Tissue fixation is required for certain procedures such as antibody-linked immunofluorescence staining. Frozen sections are often prepared during surgical removal of tumors to allow rapid identification of tumor margins, as in Mohs surgery, or determination of tumor malignancy, when a tumor is discovered incidentally during surgery.
Ultramicrotomy
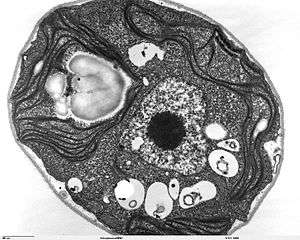
Ultramicrotomy is a method of preparing extremely thin sections for transmission electron microscope (TEM) analysis. Tissues are commonly embedded in epoxy or other plastic resin.[9] Very thin sections (less than 0.1 micrometer in thickness) are cut using diamond or glass knives on an ultramicrotome.[12]
Artifacts
Artifacts are structures or features in tissue that interfere with normal histological examination. Artifacts interfere with histology by changing the tissues appearance and hiding structures. Tissue processing artifacts can include pigments formed by fixatives,[12] shrinkage, washing out of cellular components, color changes in different tissues types and alterations of the structures in the tissue. An example is mercury pigment left behind after using Zenker's fixative to fix a section.[12] Formalin fixation can also leave a brown to black pigment under acidic conditions.[12]
History

In the 17th century, the Italian Marcello Malpighi used microscopes to study tiny biological entities and has been considered by some to be the founder of the fields of histology and microscopic pathology.[19][20] Malpighi analyzed several parts of the organs of bats, frogs and other animals under the microscope. Malpighi, while studying the structure of the lung, noticed its membranous alveoli and the hair-like connections between veins and arteries, which he named capillaries. His discovery established how the oxygen breathed in, enters the blood stream and serves the body.[21]
In the 19th century, histology was an academic discipline in its own right. The French anatomist Marie François Xavier Bichat introduced the concept of tissue in anatomy in 1801, and the term "histology" first appeared in a book of Karl Meyer in 1819.[22][23][24][19]
Bichat described twenty-one human tissues, which can be subsumed under the four categories currently accepted by histologists.[25] The usage of illustrations in histology, deemed as useless by Bichat, was promoted by Jean Cruveilhier.[26]
In the early 1830, Purkynĕ invented a microtome with high precision.[24]
During the 19th century, many fixation techniques were developed by Adolph Hannover (solutions of chromates and chromic acid), Franz Schulze and Max Schultze (osmic acid), Alexander Butlerov (formaldehyde) and Benedikt Stilling (freezing).[24]
Mounting techniques were developed by Rudolf Heidenhain (gum Arabic), Salomon Stricker (mixture of wax and oil), Andrew Pritchard (gum and isinglass) and Edwin Klebs (Canada balsam).
The 1906 Nobel Prize in Physiology or Medicine was awarded to histologists Camillo Golgi and Santiago Ramon y Cajal. They had conflicting interpretations of the neural structure of the brain based on differing interpretations of the same images. Cajal won the prize for his correct theory, and Golgi for the silver staining technique he invented to make it possible.
Notes
References
- "Microanatomy definition and meaning". Collins English Dictionary.
- "Histology | physiology". Encyclopedia Britannica. Retrieved 2018-10-29.
- "DefinedTerm: Histology". Defined Term. Retrieved 2018-10-29.
- Maximow, Alexander A.; Bloom, William (1957). A textbook of Histology (Seventh ed.). Philadelphia: W. B. Saunders Company.
- Leeson, Thomas S.; Leeson, C. Roland (1981). Histology (Fourth ed.). W. B. Saunders Company. p. 600. ISBN 978-0721657042.
- Stedman's medical dictionary (27th ed.). Lippincott Williams & Wilkins. ISBN 978-0683400076.
- Padian, Kevin; Lamm, Ellen-Thérèse, eds. (2013). Bone histology of fossil tetrapods : Advancing methods, analysis, and interpretation (1st ed.). University of California Press. p. 298. ISBN 978-0-520-27352-8.
- Canoville A, Chinsamy A (2015). "Bone Microstructure of the Stereospondyl Lydekkerina Huxleyi Reveals Adaptive Strategies to the Harsh Post Permian-Extinction Environment". Anat Rec (Hoboken). 298 (7): 1237–54. doi:10.1002/ar.23160. PMID 25857487.
- Ross, Michael H.; Pawlina, Wojciech (2016). Histology : a text and atlas : with correlated cell and molecular biology (7th ed.). Wolters Kluwer. pp. 984p. ISBN 978-1451187427.
- Rosai J (2007). "Why microscopy will remain a cornerstone of surgical pathology". Lab Invest. 87 (5): 403–8. doi:10.1038/labinvest.3700551. PMID 17401434.
- Titford, Michael; Bowman, Blythe (2012). "What May the Future Hold for Histotechnologists?". Laboratory Medicine. 43 (suppl 2): e5–e10. doi:10.1309/LMXB668WDCBIAWJL. ISSN 0007-5027.
- Bancroft, John; Stevens, Alan, eds. (1982). The Theory and Practice of Histological Techniques (2nd ed.). Longman Group Limited.
- Wick, Mark R. (2019). "The hematoxylin and eosin stain in anatomic pathology—An often-neglected focus of quality assurance in the laboratory". Seminars in Diagnostic Pathology. 36 (5): 303–311. doi:10.1053/j.semdp.2019.06.003. ISSN 0740-2570. PMID 31230963.
- Weiss AT, Delcour NM, Meyer A, Klopfleisch R (July 2011). "Efficient and cost-effective extraction of genomic DNA from formalin-fixed and paraffin-embedded tissues". Veterinary Pathology. 48 (4): 834–8. doi:10.1177/0300985810380399. PMID 20817894.
- Bennike TB, Kastaniegaard K, Padurariu S, Gaihede M, Birkelund S, Andersen V, Stensballe A (March 2016). "⿿Comparing the proteome of snap frozen, RNAlater preserved, and formalin-fixed paraffin-embedded human tissue samples". EuPA Open Proteomics. 10: 9–18. doi:10.1016/j.euprot.2015.10.001. PMC 5988570. PMID 29900094.
- Slaoui, Mohamed; Fiette, Laurence (2011). "Histopathology Procedures: From Tissue Sampling to Histopathological Evaluation". 691: 69–82. doi:10.1007/978-1-60761-849-2_4. ISSN 1064-3745. Cite journal requires
|journal=(help) - Drury, R. A. B.; Wallington, E. A. (1980). Carleton's Histological Technique (5th ed.). Oxford University Press. p. 520. ISBN 0-19-261310-3.
- Dapson RW, Horobin RW (2009). "Dyes from a twenty-first century perspective". Biotech Histochem. 84 (4): 135–7. doi:10.1080/10520290902908802. PMID 19384743.
- Bracegirdle B (1977). "The History of Histology: A Brief Survey of Sources". History of Science. 15 (2): 77–101. Bibcode:1977HisSc..15...77B. doi:10.1177/007327537701500201.
- Motta PM (1998). "Marcello Malpighi and the foundations of functional microanatomy". Anat Rec. 253 (1): 10–2. doi:10.1002/(SICI)1097-0185(199802)253:1<10::AID-AR7>3.0.CO;2-I. PMID 9556019.
- Adelmann HB, Malpighi M (1966). Marcello Malpighi and the Evolution of Embryology. 5. Ithaca, N.Y.: Cornell University Press. OCLC 306783.
- Bichat X (1801). "Considérations générales". Anatomie générale appliquée à la physiologie et à la médecine (in French). Paris: Chez Brosson, Gabon et Cie, Libraires, rue Pierre-Sarrazin, no. 7, et place de l'École de Médecine. pp. cvj–cxj.
- Mayer AF (1819). Ueber Histologie und eine neue Eintheilung der Gewebe des menschlichen Körpers (in German). Bonn: Adolph Marcus.
- Bock O (2015). "A history of the development of histology up to the end of the nineteenth century". Research. 2: 1283. doi:10.13070/rs.en.2.1283 (inactive 2019-08-20).
- Rather LJ (1978). The Genesis of Cancer: A Study in the History of Ideas. Baltimore: Johns Hopkins University Press. ISBN 9780801821035.
Most of Bichat's twenty-one tissues can be subsumed under the four categories generally accepted by contemporary histologists; epithelium, connective tissue, muscle, and nerve. Four of Bichat's tissues fall under the heading of epithelium (epidermoid, mucous, serous, and synovial); six under connective tissue (dermoid, fibrous, fibrocartilaginous, cartilaginous, osseous, and cellular); two under muscle; and two under nerve — the distinction between nervous governing "animal" life and nervous governing "organic" life corresponds with that between the voluntary and involuntary nervous systems. The arteries and the veins, long sources of contention, are classified today as compound tissues. The absorbents and the exhalants (which Bichat thought to be open-ended vessels) have dropped out or been replaced by the lymphatics. His medullary system has no counterpart among the present-day tissues.
- Meli DB (2017). Visualizing disease: the art and history of pathological illustrations. Chicago: The University of Chicago Press.