Firmicutes
The Firmicutes (Latin: firmus, strong, and cutis, skin, referring to the cell wall) are a phylum of bacteria, most of which have gram-positive cell wall structure.[3] A few, however, such as Megasphaera, Pectinatus, Selenomonas and Zymophilus, have a porous pseudo-outer membrane that causes them to stain gram-negative. Scientists once classified the Firmicutes to include all gram-positive bacteria, but have recently defined them to be of a core group of related forms called the low-G+C group, in contrast to the Actinobacteria. They have round cells, called cocci (singular coccus), or rod-like forms (bacillus).
| Firmicutes | |
|---|---|
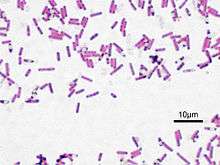 | |
| Bacillus subtilis, Gram-stained | |
| Scientific classification | |
| Domain: | Bacteria |
| (unranked): | Terrabacteria |
| Phylum: | Firmicutes Gibbons and Murray 1978,[1] Murray, 1984[2] |
| Classes | |
| |
| Synonyms | |
| |
Many Firmicutes produce endospores, which are resistant to desiccation and can survive extreme conditions. They are found in various environments, and the group includes some notable pathogens. Those in one family, the heliobacteria, produce energy through anoxygenic photosynthesis. Firmicutes play an important role in beer, wine, and cider spoilage.
Classes
The group is typically divided into the Clostridia, which are anaerobic, and the Bacilli, which are obligate or facultative aerobes.
On phylogenetic trees, the first two groups show up as paraphyletic or polyphyletic, as do their main genera, Clostridium and Bacillus.[4]
Phylogeny
The phylogeny is based on 16S rRNA-based LTP release 132 by the All-Species Living Tree Project,[5] with the currently accepted taxonomy based on the List of Prokaryotic names with Standing in Nomenclature (LPSN),[6] National Center for Biotechnology Information (NCBI),[7] and some non-validated clade names from Genome Taxonomic Database.[8]
| Firmicutes classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Unassigned Clostridia s.s.
|
Notes:
♥ Clade names not lodged at National Center for Biotechnology Information (NCBI) or listed in the List of Prokaryotic names with Standing in Nomenclature (LPSN)
The family Synergistaceae (Clostridiales Family XV Incertae Sedis) and Thermodesulfobiaceae have been removed from Firmicutes based on the fact, they do not form a monophyletic clade with it.
Genera
More than 274 genera were considered as of 2016 to be within the Firmicutes phylum, notable genera of Firmicutes include:
Bacilli, order Bacillales
Bacilli, order Lactobacillales
- Acetobacterium
- Clostridioides
- Clostridium
- Eubacterium
- Heliobacteria
- Heliospirillum
- Megasphaera
- Pectinatus
- Selenomonas
- Zymophilus
- Sporohalobacter
- Sporomusla
Health implications
Firmicutes make up the largest portion of the mouse and human gut microbiome.[9] The division Firmicutes as part of the gut flora has been shown to be involved in energy resorption, and potentially related to the development of diabetes and obesity.[10][11][12][13] Within the gut of healthy human adults, the most abundant bacterium: Faecalibacterium prausnitzii (F. prausnitzii), which makes up 5% of the total gut microbiome, is a member of the Firmicutes phylum. This species is directly associated with reduced low-grade inflammation in obesity.[14] F. prausnitzii has been found in higher levels within the guts of obese children than in non-obese children.
In multiple studies a higher abundance of Firmicutes has been found in obese individuals than in lean controls. A higher level of Lactobacillus (of the Firmicutes phylum) has been found in obese patients and in one study, obese patients put on weight loss diets showed a reduced amount of Firmicutes within their guts.[15]
Diet changes in mice have also been shown to promote changes in Firmicutes abundance. A higher relative abundance of Firmicutes was seen in mice fed a western diet (high fat/high sugar) than in mice fed a standard low fat/ high polysaccharide diet. The higher amount of Firmicutes was also linked to more adiposity and body weight within mice.[16] Specifically, within obese mice, the class Mollicutes (within the Firmicutes phylum) was the most common. When the microbiota of obese mice with this higher Firmicutes abundance was transplanted into the guts of germ-free mice, the germ-free mice gained a significant amount of fat as compared to those transplanted with the microbiota of lean mice with lower Firmicutes abundance.[17]
The presence of Christensenella (Firmicutes, in class Clostridia), isolated from human faeces, has been found to correlate with lower body mass index.[18]
Laboratory detection
The presence of Firmicutes can be reliably detected with polymerase chain reaction (PCR) techniques.[19]
References
- Gibbons, N. E. & Murray, R. G. E. 1978. Proposals concerning the higher taxa of bacteria. Int J Syst Bacteriol 28:1–6, (PDF)
- Murray, R. G. E. (1984). The higher taxa, or, a place for everything...?. In: N. R. Krieg & J. G. Holt (ed.) Bergey's Manual of Systematic Bacteriology, vol. 1, The Williams & Wilkins Co., Baltimore, p. 31–34.
- "Firmicutes" at Dorland's Medical Dictionary
- Wolf M, Müller T, Dandekar T, Pollack JD (May 2004). "Phylogeny of Firmicutes with special reference to Mycoplasma (Mollicutes) as inferred from phosphoglycerate kinase amino acid sequence data". Int. J. Syst. Evol. Microbiol. (Comparative Study). 54 (Pt 3): 871–5. CiteSeerX 10.1.1.126.3863. doi:10.1099/ijs.0.02868-0. PMID 15143038.
- "16S rRNA-based LTP release 132 (full tree)". All-Species Living Tree Project. Silva Comprehensive Ribosomal RNA Database. Retrieved 2013-03-20.
- J. P. Euzéby. "Firmicutes". List of Prokaryotic names with Standing in Nomenclature (LPSN). Archived from the original on January 27, 2013. Retrieved 2013-03-20.
- Sayers; et al. "Firmicutes". National Center for Biotechnology Information (NCBI) taxonomy database. Retrieved 24 April 2019.
- "GTDB taxonomy". Genome Taxonomic Database. Archived from the original on 2018-07-29. Retrieved 2018-07-20.
- Ley RE, Peterson DA, Gordon JI (2006). "Ecological and evolutionary forces shaping microbial diversity in the human intestine". Cell (Review). 124 (4): 837–848. doi:10.1016/j.cell.2006.02.017. PMID 16497592.
- Ley RE, Turnbaugh PJ, Klein S, Gordon JI (2006). "Microbial ecology: human gut microbes associated with obesity". Nature (Clinical Trial). 444 (7122): 1022–1023. Bibcode:2006Natur.444.1022L. doi:10.1038/4441022a. PMID 17183309.
- Henig, Robin Marantz (2006-08-13). "Fat Factors". New York Times Magazine. Retrieved 2008-09-28.
- Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI (August 2005). "Obesity alters gut microbial ecology". Proc. Natl. Acad. Sci. USA (Research Support). 102 (31): 11070–11075. Bibcode:2005PNAS..10211070L. doi:10.1073/pnas.0504978102. PMC 1176910. PMID 16033867.
- Komaroff AL. The Microbiome and Risk for Obesity and Diabetes. JAMA. Published online December 22, 2016. doi:10.1001/jama.2016.20099
- Chakraborti, Chandra Kanti (15 November 2015). "New-found link between microbiota and obesity". World Journal of Gastrointestinal Pathophysiology. 6 (4): 110–119. doi:10.4291/wjgp.v6.i4.110. PMC 4644874. PMID 26600968.
- Million, M.; Lagier, J.-C; Yahav, D.; Paul, M. (April 2013). "Gut bacterial microbiota and obesity". Clinical Microbiology and Infection. 19 (4): 305–313. doi:10.1111/1469-0691.12172. PMID 23452229.
- Turnbaugh, Peter J. (17 April 2008). "Diet-Induced Obesity Is Linked to Marked but Reversible Alterations in the Mouse Distal Gut Microbiome". Cell Host & Microbe. 3 (4): 213–223. doi:10.1016/j.chom.2008.02.015. PMC 3687783. PMID 18407065.
- Million, M. (April 2013). "Gut bacterial microbiota and obesity". Cell Microbiology and Infection. 19 (4): 305–313. doi:10.1111/1469-0691.12172. PMID 23452229.
- Goodrich, Julia K.; Waters, Jillian L.; Poole, Angela C.; Sutter, Jessica L.; Koren, Omry; Blekhman, Ran; Beaumont, Michelle; Van Treuren, William; Knight, Rob; Bell, Jordana T.; Spector, Timothy D.; Clark, Andrew G.; Ley, Ruth E. (2014). "Human Genetics Shape the Gut Microbiome". Cell. 159 (4): 789–799. doi:10.1016/j.cell.2014.09.053. ISSN 0092-8674. PMC 4255478. PMID 25417156.

- Haakensen M, Dobson CM, Deneer H, Ziola B (July 2008). "Real-time PCR detection of bacteria belonging to the Firmicutes Phylum". Int. J. Food Microbiol. (Research Support). 125 (3): 236–41. doi:10.1016/j.ijfoodmicro.2008.04.002. PMID 18501458.
External links
- Phylum "Firmicutes" - J.P. Euzéby: List of Prokaryotic names with Standing in Nomenclature