Cardiac neural crest complex
Neural crest cells are multipotent cells required for the development of cells, tissues and organ systems.[1] A subpopulation of neural crest cells are the cardiac neural crest complex. This complex refers to the cells found amongst the midotic placode and somite 3 destined to undergo epithelial-mesenchymal transformation and migration to the heart via pharyngeal arches 3, 4 and 6.[2]
| Cardiac neural crest complex | |
|---|---|
| Details | |
| Identifiers | |
| Latin | complexus cristae neuralis cardiacus |
| TE | E4.0.3.5.0.3.1 |
| Anatomical terminology | |
The cardiac neural crest complex plays a vital role in forming connective tissues that aid in outflow septation and modelling of the aortic arch arteries during early development.[2] Ablation of the complex often leads to impaired myocardial functioning similar to symptoms present in DiGeorge syndrome.[3] Consequently, the removal of cardiac crest cells that populate in pharyngeal arches has flow on effects on the thymus, parathyroid and thyroid gland.[4]
Development
The cardiac neural crest originates from the region of cells between somite 3 and the midotic placode that migrate towards and into the cardiac outflow tract.[5] The cells migrate from the neural tube to populate pharyngeal arches 3, 4 and 6 with the largest population of the outflow tract originating from those in pharyngeal arches 4.[2] From here, a subpopulation of cells will develop into the endothelium of the aortic arch arteries while others will migrate into the outflow tract to form the aorticopulmonary and truncal septa.[2][6] Other ectomesenchymal cells will form the thymus and parathyroid glands.[7]
Epithelial-mesenchymal transition
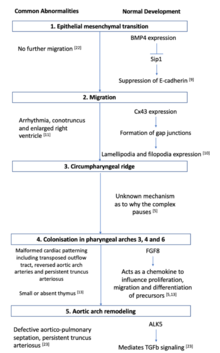
Prior to migration, during a process known as epithelial-mesenchymal transition, there is a loss of cell contact, remodelling of the cytoskeleton and increased motility and interaction with extracellular components in the matrix.[8] An important step in this process is the suppression of adhesion protein E-cadherin present on epithelial cells to initiate the migration process. This suppression mechanism occurs via the growth factor BMP signalling to turn on a transcriptional repressor Smad-interacting protein 1 (Sip1) and marks the beginning of the epithelial-mesenchymal transition.[9]
Early migration
During migration, crest cells destined for pharyngeal arches maintain contact with each other via lamellipodia and filopodia. Short range local contact is maintained with lamellipodia whilst long range non-local contact is maintained with filopodia.[10] During this process, connexin 43 (Cx43) regulates cell interaction by regulating the formation of channels known as gap junctions.[1] Impaired Cx43 function in transgenic mice leads to altered coronary artery patterns and abnormal outflow tracts.[11] Further gap junction signalling is dependent on a cadherin mediated cell adhesion formed during cross talking with p120 catenin signalling.[12]
Appropriate outflow tract formation relies on a morphogen concentration gradient set up by fibroblast growth factor (FGF) secreting cells. Cardiac crest cells furthest away from FGF secreting cells will receive lower concentrations of FGF8 signalling than cells closer to FGF secreting cells. This allows for appropriate formation of the outflow tract.[13] Cells located in rhombomeres 3and 5 undergo programmed cell death under signalling cues from semaphorins. The lack of cells in this region results in the formation of crest-free zones.[14]
The process of migration requires a permissive extracellular matrix.[2] The enzyme arginyltransferase creates this environment by adding an arginyl group onto newly synthesised proteins during post-translational modification.[15] This process aids cells motility and ensures proteins the proteins contained within the actin cytoskeleton is prepped for migration.[2]
Circumpharyngeal ridge
Cell migration towards the circumpharyngeal ridge is forced to paused to allow for the formation of the caudal pharyngeal arches.[2] Little is known about this pausing mechanism, but studies conducted in chicks have uncovered the role of mesoderm expressed factors EphrinB3 and EphrinB4 in forming fibronectin attachments.[16]
Caudal pharynx and arch artery condensation
Pharyngeal arches are tissues composed of mesoderm-derived cells enclosed by an external ectoderm and an internal endoderm.[2] Once the caudal pharyngeal arches are formed, cardiac neural crest complexes will migrate towards these and colonise in arches 3, 4 and 6. Cells leading this migration maintain contact with the extracellular matrix and contain the protein filopedia that acts as extensions towards the ectodermal pharyngeal arches.[2][10] A range of secreted factors ensure appropriate directionality of the cells. FGF8 acts as a chemotactic attraction in directing cellular movement towards pharyngeal arch 4.[1][10]
A second signalling pathway that directs crest cell movement are the family of endothelin ligands. Migrating cardiac neural crest cells will populate at the correct pharyngeal arches under signalling guidance from EphrinA and Ephrin B variations. This corresponds with receptor expression at the pharyngeal arches. Pharyngeal arch 3 expresses EphrinA and EphrinB1 receptors and pharyngeal arch 2 expresses EphrinB2 and allows for the binding of EphrinA and EphrinB variations to guide migration of the cardiac neural crest cells.[2]
Aortic arch remodelling
The aortic arch arteries transport blood from the aorta to the head and trunk of the embryo.[17] Normally, early development of the outflow tract begins with a single vessel that forms bilateral symmetrical branches at the aortic sac within pharyngeal arches. This process requires the elongation of the outflow tract as a prerequisite to ensure the correct series of looping and cardiac alignment.[1] The cardiac neural crest complex then colonises in the truncal cushion and is localised to the subendothelial layer prior to spiralisation of the endocardarial cushion to form the conotruncal ridges. This later undergoes remodelling to form the left-sided aortic pattern present in adult hearts.[1] The group of cells found in the third aortic arch gives rise to common carotid arteries. Cells found in the fourth aortic arch differentiates to form the distal aortic arch and right subclavian artery, whilst cells in the sixth aortic arch develops into the pulmonary arteries. Cardiac neural crest cells express Hox genes that supports the development of arteries 3, 4 and 6 and the simultaneous regression of arteries 1 and 2. The ablation of Hox genes on cardiac neural crest cells causes defective outflow septation.[17]
Ablation of cardiac neural crest complex
Cardiac outflow anomalies
One of the main cardiac outflow anomalies present during cardiac neural crest complex ablation is persistent truncus arteriosus.[5] This arises when the arterial trunk fails to divide and cause the separation of pulmonary artery and aorta.[1] This results in a lack of aorticopulmonary septum as the vessels which would normally disappear during normal development remains and interrupts the carotid vessels.[5] The malformation of the heart and its associated great vessels depends on the extent and location of the cardiac neural crest complex ablation.[5] Complete removal of cardiac neural crests results in persistent truncus arteriosus characterised in most cases by the presence of just one outflow valve and a ventricular septal defect.[18] Mesencephalic neural crest cells interfere with normal development of cardiac outflow septation as its presence leads to persistent truncus arteriosus.[19] However, the addition of trunk neural crest cells results in normal heart development.[5]
Other outcomes of cardiac outflow anomalies includes Tetralogy of Fallot, Eisenmenger’s complex, transposition of the great vessels and double outlet right ventricle.[5]
Aortic arch arteries anomalies
Overriding aorta is caused by the abnormal looping during early development of the heart and is accompanied with ventricular septal defects.[3] Instead of abnormal formation of the aorticopulmonary septum, partial removal of cardiac neural crest results in an overriding aorta, whereby the misplacement of the aorta is found over the ventricular septum as opposed to the left ventricle.[18] This results in a reduction of oxygenated blood as the aorta receives some deoxygenated blood from the flow of the right ventricle. There is a reduction in the quantity of endothelial tubes of ectomesenchyme in pharyngeal arches that surround the aortic arch arteries.[5]
Other outcomes of aortic arch artery anomalies includes a double aortic arch, variable absence of the carotid arteries and left aortic arch.[5]
Functional changes to the heart
Functional changes to the heart becomes apparent well before structural changes are observed in the phenotype of ablated chicks. This is due to the embryo compromising morphological changes to the heart to maintain cardiac functioning via vasodilation. Despite an increase in embryonic stroke volume and cardiac output, this compensation of decreased contraction results in misalignment of the development vessels due to incomplete looping of the cardiac tube.[5]
In an adult heart, myocardium contraction occurs via excitation-contraction coupling whereby cellular depolarisation occurs and allows an influx of calcium via voltage-gated calcium channels. A subsequent reuptake of calcium into the sarcoplasmic reticulum causes a decrease in intracellular calcium to cause myocardium relaxation.[17] The removal of the cardiac neural crest complex causes a reduction in contractility of the myocardium. In embryos containing persistent truncus arteriosus, there is a significant 2-fold reduction in calcium currents, thereby interrupting the cardiac excitation-contraction coupling process to cause a reduction in contractility.[5][17]
Pulmonary venous system
During cardiogenesis, migration of the cardiac neural crest complex occurs prior to the development of the pulmonary system. There is no visible difference in the pulmonary veins of chick embryos that developed persistent truncus arteriosus and embryos with an intact cardiac neural crest complex. Ablation of the cardiac neural crest complex do not play a role in the systemic or pulmonary venous system as no visible venous defects is observed.[20]
Derivative development
Due to its population in pharyngeal arches, removal of the cardiac neural crest complex has flow on effects on the thymus, parathyroid and thyroid gland.[7]
Location
Into the pharyngeal arches and Truncus arteriosus (embryology), forming the aorticopulmonary septum[21] and the smooth muscle of great arteries.
Anterior of the aorta to become the four pre-aortic ganglia: (celiac ganglion, superior mesenteric ganglion, inferior mesenteric ganglion and aortical renal ganglia).
References
- Snider P, Olaopa M, Firulli AB, Conway SJ (2007). "Cardiovascular development and the colonizing cardiac neural crest lineage". The Scientific World Journal. 7: 1090–1113. doi:10.1100/tsw.2007.189. PMC 2613651. PMID 17619792.
- Kirby ML, Hutson MR (2010). "Factors controlling cardiac neural crest cell migration". Cell Adhesion & Migration. 4 (4): 609–621. doi:10.4161/cam.4.4.13489. PMC 3011257. PMID 20890117.
- Hutson MR, Kirby ML (2007). "Model systems for the study of heart development and disease: cardiac neural crest and conotruncal malformations". Seminars in Cell & Developmental Biology. 18 (1): 101–110. doi:10.1016/j.semcdb.2006.12.004. PMC 1858673. PMID 17224285.
- Le Lièvre CS, Le Douarin NM (1975). "Mesenchymal derivatives of the neural crest: analysis of chimaeric quail and chick embryos". Development. 34 (1): 124–154. PMID 1185098.
- Le Lièvre CS, Le Douarin NM (1990). "Role of neural crest in congenital heart disease". Circulation. 82 (2): 332–340. doi:10.1161/01.CIR.82.2.332. PMID 2197017.
- Bajolle F, Zaffran S, Meilhac SM, Dandonneau M, Chang T, Kelly RG (2008). "Myocardium at the base of the aorta and pulmonary trunk is prefigured in the outflow tract of the heart and in subdomains of the second heart field". Developmental Biology. 313 (1): 25–34. doi:10.1016/j.ydbio.2007.09.023. PMID 18005956.
- Bockman DE, Kirby ML (1984). "Dependence of thymus development on derivatives of the neural crest". Science. 223 (4635): 498–500. Bibcode:1984Sci...223..498B. doi:10.1126/science.6606851. PMID 6606851.
- Hay ED (1995). "An overview of epithelio-mesenchymal trans-formation". Actaanatomica. 154 (1): 8–20. doi:10.1159/000147748. PMID 8714286.
- Comijn J, Berx G, Vermassen P, Verschueren K, van Grunsven L, Bruyneel E, Mareel M, Huylebroeck D, van Roy F (2007). "The two-handed E box binding zinc finger protein SIP1 downregulates E-cadherin and induces invasion". Molecular Cell. 7 (6): 1267–1278. doi:10.1016/S1097-2765(01)00260-X. PMID 11430829.
- Teddy JM, Kulesa PM (2004). "In vivo evidence for short-and long-range cell communication in cranial neural crest cells". Development. 131 (24): 6141–6151. doi:10.1242/dev.01534. PMID 15548586.
- Huang GY, Wessels A, Smith BR, Linask KK, Ewart JL, Lo CW (1998). "Alteration in connexin 43 gap junction gene dosage impairs conotruncal heart development". Developmental Biology. 198 (1): 32–44. doi:10.1006/dbio.1998.8891. PMID 9640330.
- Xu X, Li WE, Huang GY, Meyer R, Chen T, Luo Y, Thomas MP, Radice GL, Lo CW (2001). "Modulation of mouse neural crest cell motility by N-cadherin and connexin 43 gap junctions". Journal of Cell Biology. 154 (1): 217–230. doi:10.1083/jcb.200105047. PMC 2196865. PMID 11449002.
- Abu-Issa R, Smyth G, Smoak I, Yamamura K, Meyers EN (2002). "Fgf8 is required for pharyngeal arch and cardiovascular development in the mouse". Development. 129 (19): 4613–4625. PMID 12223417.
- Toyofuku T, Yoshida J, Sugimoto T, Yamamoto M, Makino N, Takamatsu H, Takegahara N, Suto F, Hori M, Fujisawa H, Kumanogoh A, Kukutani H (2007). "Repulsive and attractive semaphorins cooperate to direct the navigation of cardiac neural crest cells". The Scientific World Journal. 7 (1): 1090–1113. doi:10.1016/j.ydbio.2008.06.028. PMID 18625214.
- Kurosaka S, Leu NA, Zhang F, Bunte R, Saha S, Wang J, Guo C, He W, Kashina A (2010). "Arginylation-dependent neural crest cell migration is essential for mouse development". PLoS Genetics. 6 (3): e1000878. doi:10.1371/journal.pgen.1000878. PMC 2837401. PMID 20300656.
- Santiago A, Erickson CA (2002). "Ephrin-B ligands play a dual role in the control of neural crest cell migration". Development. 129 (15): 3621–3623. PMID 12117812.
- Creazzo TL, Godt RE, Leatherbury L, Conway SJ, Kirby ML (1998). "Role of cardiac neural crest cells in cardiovascular development". Annual Review of Physiology. 60 (1): 267–286. doi:10.1146/annurev.physiol.60.1.267. PMID 9558464.
- van den Hoff MJ, Moorma AF (2000). "Cardiac neural crest: the holy grail of cardiac abnormalities?". Cardiovascular Research. 47 (2): 212–216. doi:10.1016/s0008-6363(00)00127-9. PMID 10946058.
- Kirby ML (1989). "Plasticity and predetermination of mesencephalic and trunk neural crest transplanted into the region of the cardiac neural crest". Developmental Biology. 134 (2): 402–412. doi:10.1016/0012-1606(89)90112-7. PMID 2744240.
- Phillips III MT, Waldo K, Kirby ML (1989). "Neural crest ablation does not alter pulmonary vein development in the chick embryo.". The Anatomical Record. 223 (3): 292–298. doi:10.1002/ar.1092230308. PMID 2923280.
- Jiang X, Rowitch DH, Soriano P, McMahon AP, Sucov HM (April 2000). "Fate of the mammalian cardiac neural crest". Development. 127 (8): 1607–16. PMID 10725237.
External links
- Embryology at UNSW Notes/ncrest