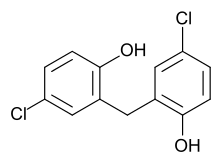
Dichlorophen
Dichlorophen is an anticestodal agent, fungicide, germicide, and antimicrobial agent.[2] It is used in combination with toluene for the removal of parasites such as ascarids, hookworms, and tapeworms from dogs and cats.[3]
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.002.335 |
| Chemical and physical data | |
| Formula | C13H10Cl2O2 |
| Molar mass | 269.12 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.5 g/cm3 g/cm3 |
| Melting point | 177.5 °C (351.5 °F) |
| Solubility in water | 0.003 g/100 mL[1] mg/mL (20 °C) |
SMILES
| |
InChI
| |
| | |
References
- Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 8–118, ISBN 0-8493-0594-2
- Milne, G.W.A. (Ed.). (2005). Gardner's commercially important chemicals: Synonyms, trade names, and properties. Hoboken, N.J.: Wiley-Interscience. Google Books
- "Code of Federal Regulations", Code of Federal Regulations, Title 21, Volume 6, U.S. Government Printing Office, 2005-04-01, retrieved 2009-05-01
- Helmut Fiege; Heinz-Werner Voges; Toshikazu Hamamoto; Sumio Umemura; Tadao Iwata; Hisaya Miki; Yasuhiro Fujita; Hans-Josef Buysch; Dorothea Garbe (2007). "Phenol Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_313.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.