Somatosensory system
The somatosensory system is a part of the sensory nervous system. The somatosensory system is a complex system of sensory neurons and neural pathways that responds to changes at the surface or inside the body. The axons (as afferent nerve fibers) of sensory neurons connect with, or respond to, various receptor cells. These sensory receptor cells are activated by different stimuli such as heat and nociception, giving a functional name to the responding sensory neuron, such as a thermoreceptor which carries information about temperature changes. Other types include mechanoreceptors, chemoreceptors, and nociceptors which send signals along a sensory nerve to the spinal cord where they may be processed by other sensory neurons and then relayed to the brain for further processing. Sensory receptors are found all over the body including the skin, epithelial tissues, muscles, bones and joints, internal organs, and the cardiovascular system.

Somatic senses are sometimes referred to as somesthetic senses,[1] with the understanding that somesthesis includes the sense of touch, proprioception (sense of position and movement), and (depending on usage) haptic perception.[2]
The mapping of the body surfaces in the brain is called somatotopy. In the cortex, it is also referred to as the cortical homunculus. This brain-surface ("cortical") map is not immutable, however. Dramatic shifts can occur in response to stroke or injury.
System overview
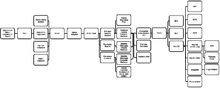
Sensory receptors
The four mechanoreceptors in the skin each respond to different stimuli for short or long periods.
Merkel cell nerve endings are found in the basal epidermis and hair follicles; they react to low vibrations (5–15 Hz) and deep static touch such as shapes and edges. Due to having a small receptive field (extremely detailed info), they are used in areas like fingertips the most; they are not covered (shelled) and thus respond to pressures over long periods.
Tactile corpuscles react to moderate vibration (10–50 Hz) and light touch. They are located in the dermal papillae; due to their reactivity, they are primarily located in fingertips and lips. They respond in quick action potentials, unlike Merkel nerve endings. They are responsible for the ability to read Braille and feel gentle stimuli.
Lamellar corpuscles determine gross touch and distinguish rough and soft substances. They react in quick action potentials, especially to vibrations around 250 Hz (even up to centimeters away). They are the most sensitive to vibrations and have large receptor fields. Pacinian reacts only to sudden stimuli so pressures like clothes that are always compressing their shape are quickly ignored.
Bulbous corpuscles react slowly and respond to sustained skin stretch. They are responsible for the feeling of object slippage and play a major role in the kinesthetic sense and control of finger position and movement. Merkel and bulbous cells - slow-response - are myelinated; the rest - fast-response - are not. All of these receptors are activated upon pressures that squish their shape causing an action potential.[3][4][5][6]
Somatosensory cortex
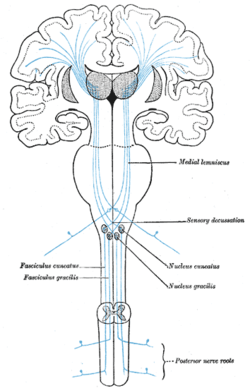
The postcentral gyrus includes the primary somatosensory cortex (Brodmann areas 3, 2 and 1) collectively referred to as S1.
BA3 receives the densest projections from the thalamus. BA3a is involved with the sense of relative position of neighboring body parts and amount of effort being used during movement. BA3b is responsible for distributing somatosensory information, it projects texture information to BA1 and shape and size information to BA2.
Region S2 (secondary somatosensory cortex) divides into Area S2 and parietal ventral area. Area S2 is involved with specific touch perception and is thus integrally linked with the amygdala and hippocampus to encode and reinforce memories.
Parietal ventral area is the somatosensory relay to the premotor cortex and somatosensory memory hub, BA5.
BA5 is the topographically organized somato memory field and association area.
BA1 processes texture info while BA2 processes size + shape info.
Area S2 processes light touch, pain, visceral sensation, and tactile attention.
S1 processes the remaining info (crude touch, pain, temperature).[7][8][9]
BA7 integrates visual and proprioceptive info to locate objects in space.[10][11]
The insular cortex (insula) plays a role in the sense of bodily-ownership, bodily self-awareness, and perception. Insula also plays a role in conveying info about sensual touch, pain, temperature, itch, and local oxygen status. Insula is a highly connected relay and thus is involved in numerous functions.
Structure
The somatosensory system is spread through all major parts of the vertebrate body. It consists both of sensory receptors and afferent neurons in the periphery (skin, muscle and organs for example), to deeper neurons within the central nervous system.
General somatosensory pathway
All afferent touch/vibration info ascends the spinal cord via the posterior (dorsal) column-medial lemniscus pathway via gracilis (T7 and below) or cuneatus (T6 and above). Cuneatus sends signals to the cochlear nucleus indirectly via spinal grey matter, this info is used in determining if a perceived sound is just villi noise/irritation. All fibers cross (left becomes right) in the medulla.
A somatosensory pathway will typically have three neurons:[12] first-order, second-order, and third-order.
- The first-order neuron always has its cell body in the dorsal root ganglion of the spinal nerve (if sensation is in parts of the head or neck not covered by the cervical nerves, it will be the trigeminal nerve ganglia or the ganglia of other sensory cranial nerves).
- The second-order neuron has its cell body either in the spinal cord or in the brainstem. This neuron's ascending axons will cross (decussate) to the opposite side either in the spinal cord or in the brainstem.
- In the case of touch and certain types of pain, the third-order neuron has its cell body in the VPN of the thalamus and ends in the postcentral gyrus of the parietal lobe.

Photoreceptors, similar to those found in the retina of the eye, detect potentially damaging ultraviolet radiation (ultraviolet A specifically), inducing increased production of melanin by melanocytes.[13] Thus tanning potentially offers the skin rapid protection from DNA damage and sunburn caused by ultraviolet radiation (DNA damage caused by ultraviolet B). However, whether this offers protection is debatable, because the amount of melanin released by this process is modest in comparison to the amounts released in response to DNA damage caused by ultraviolet B radiation.[13]
Tactile feedback
The tactile feedback from proprioception is derived from the proprioceptors in the skin, muscles, and joints.[14]
Balance
The receptor for the sense of balance resides in the vestibular system in the ear (for the three-dimensional orientation of the head, and by inference, the rest of the body). Balance is also mediated by the kinesthetic reflex fed by proprioception (which senses the relative location of the rest of the body to the head).[15] In addition, proprioception estimates the location of objects which are sensed by the visual system (which provides confirmation of the place of those objects relative to the body), as input to the mechanical reflexes of the body.
Fine touch and crude touch
Fine touch (or discriminative touch) is a sensory modality that allows a subject to sense and localize touch. The form of touch where localization is not possible is known as crude touch. The posterior column–medial lemniscus pathway is the pathway responsible for the sending of fine touch information to the cerebral cortex of the brain.
Crude touch (or non-discriminative touch) is a sensory modality that allows the subject to sense that something has touched them, without being able to localize where they were touched (contrasting "fine touch"). Its fibres are carried in the spinothalamic tract, unlike the fine touch, which is carried in the dorsal column. [16] As fine touch normally works in parallel to crude touch, a person will be able to localize touch until fibres carrying fine touch (Posterior column–medial lemniscus pathway) have been disrupted. Then the subject will feel the touch, but be unable to identify where they were touched.
Neural processing of social touch
The somatosensory cortex encodes incoming sensory information from receptors all over the body. Affective touch is a type of sensory information that elicits an emotional reaction and is usually social in nature, such as a physical human touch. This type of information is actually coded differently than other sensory information. Intensity of affective touch is still encoded in the primary somatosensory cortex and is processed in a similar way to emotions invoked by sight and sound, as exemplified by the increase of adrenaline caused by the social touch of a loved one, as opposed to the physical inability to touch someone you don't love.
Meanwhile, the feeling of pleasantness associated with affective touch activates the anterior cingulate cortex more than the primary somatosensory cortex. Functional magnetic resonance imaging (fMRI) data shows that increased blood-oxygen-level contrast (BOLD) signal in the anterior cingulate cortex as well as the prefrontal cortex is highly correlated with pleasantness scores of an affective touch. Inhibitory transcranial magnetic stimulation (TMS) of the primary somatosensory cortex inhibits the perception of affective touch intensity, but not affective touch pleasantness. Therefore, the S1 is not directly involved in processing socially affective touch pleasantness, but still plays a role in discriminating touch location and intensity.[16]
Individual variation
A variety of studies have measured and investigated the causes for differences between individuals in the sense of fine touch. One well-studied area is passive tactile spatial acuity, the ability to resolve the fine spatial details of an object pressed against the stationary skin. A variety of methods have been used to measure passive tactile spatial acuity, perhaps the most rigorous being the grating orientation task.[17] In this task subjects identify the orientation of a grooved surface presented in two different orientations,[18] which can be applied manually or with automated equipment.[19] Many studies have shown a decline in passive tactile spatial acuity with age;[20][21][22] the reasons for this decline are unknown, but may include loss of tactile receptors during normal aging. Remarkably, index finger passive tactile spatial acuity is better among adults with smaller index fingertips;[23] this effect of finger size has been shown to underlie the better passive tactile spatial acuity of women, on average, compared to men.[23] The density of tactile corpuscles, a type of mechanoreceptor that detects low-frequency vibrations, is greater in smaller fingers;[24] the same may hold for Merkel cells, which detect the static indentations important for fine spatial acuity.[23] Among children of the same age, those with smaller fingers also tend to have better tactile acuity.[25] Many studies have shown that passive tactile spatial acuity is enhanced among blind individuals compared to sighted individuals of the same age,[22][26][27][28][29] possibly because of cross modal plasticity in the cerebral cortex of blind individuals. Perhaps also due to cortical plasticity, individuals who have been blind since birth reportedly consolidate tactile information more rapidly than sighted people.[30]
Clinical significance
A somatosensory deficiency may be caused by a peripheral neuropathy involving peripheral nerves of the somatosensory system. This may present as numbness or paresthesia.
Society and culture
Haptic technology can provide touch sensation in virtual and real environments.[31] In the field of speech therapy, tactile feedback can be used to treat speech disorders.
See also
- Allochiria
- Cell signalling
- Golgi tendon organ
- Haptic communication
- Haptic perception
- Muscle spindle
- Molecular cellular cognition
- Phantom limb
- Sensory maps
- Somatosensory Rehabilitation of Pain
- Special senses
- Supramarginal gyrus
- Tactile illusion
- Vibratese, method of communication through touch
- Tactile imaging
References
- O. Franzen, R. Johansson, L. Terenius (1996) Somesthesis and the Neurobiology of the Somatosensory Cortex
- Robles-De-La-Torre G (2006). "The Importance of the Sense of Touch in Virtual and Real Environments" (PDF). IEEE Multimedia. 13 (3): 24–30. doi:10.1109/MMUL.2006.69.
- Paré, Michel, and Catherine Behets. "Paucity of Presumptive Ruffini Corpuscles in the Index Finger Pad of Humans." Wiley Online Library. 10 Feb. 2003. Web. 27 Mar. 2016.
- Scheibert J, Leurent S, Prevost A, Debrégeas G (March 2009). "The role of fingerprints in the coding of tactile information probed with a biomimetic sensor". Science. 323 (5920): 1503–6. arXiv:0911.4885. Bibcode:2009Sci...323.1503S. doi:10.1126/science.1166467. PMID 19179493.
- Biswas A, Manivannan M, Srinivasan MA (2015). "Vibrotactile sensitivity threshold: nonlinear stochastic mechanotransduction model of the Pacinian Corpuscle". IEEE Transactions on Haptics. 8 (1): 102–13. doi:10.1109/TOH.2014.2369422. PMID 25398183.
- Paré, Michel, and Robert Elde. "The Meissner Corpuscle Revised: A Multiafferented Mechanoreceptor with Nociceptor Immunochemical Properties." JNeurosci. 15 Sept. 2001. Web. 27 Mar. 2016.
- Hashim IH, Kumamoto S, Takemura K, Maeno T, Okuda S, Mori Y (November 2017). "Tactile Evaluation Feedback System for Multi-Layered Structure Inspired by Human Tactile Perception Mechanism". Sensors (Basel, Switzerland). 17 (11): 2601. doi:10.3390/s17112601. PMC 5712818. PMID 29137128.
- Buccino G, Binkofski F, Fink GR, Fadiga L, Fogassi L, Gallese V, Seitz RJ, Zilles K, Rizzolatti G, Freund HJ (January 2001). "Action observation activates premotor and parietal areas in a somatotopic manner: an fMRI study". The European Journal of Neuroscience. 13 (2): 400–4. doi:10.1111/j.1460-9568.2001.01385.x. PMID 11168545.
- Seelke AM, Padberg JJ, Disbrow E, Purnell SM, Recanzone G, Krubitzer L (August 2012). "Topographic Maps within Brodmann's Area 5 of macaque monkeys". Cerebral Cortex. 22 (8): 1834–50. doi:10.1093/cercor/bhr257. PMC 3388892. PMID 21955920.
- Geyer S, Schleicher A, Zilles K (July 1999). "Areas 3a, 3b, and 1 of Human Primary Somatosensory Cortex". NeuroImage. 10 (1): 63–83. doi:10.1006/nimg.1999.0440. PMID 10385582.
- Disbrow E (June 2002). "Thalamocortical connections of the parietal ventral area (PV) and the second somatosensory area (S2) in macaque monkeys". Thalamus & Related Systems. 1 (4): 289–302. doi:10.1016/S1472-9288(02)00003-1.
- Saladin KS. Anatomy and Physiology 3rd edd. 2004. McGraw-Hill, New York.
- Zukerman, Wendy. "Skin 'sees' the light to protect against sunshine". newscientist.com. New Scientist. Retrieved 2015-01-22.
- Proske U, Gandevia SC (October 2012). "The proprioceptive senses: their roles in signaling body shape, body position and movement, and muscle force". Physiological Reviews. 92 (4): 1651–97. doi:10.1152/physrev.00048.2011. PMID 23073629.
- Proske U, Gandevia SC (September 2009). "The kinaesthetic senses". The Journal of Physiology. 587 (Pt 17): 4139–46. doi:10.1113/jphysiol.2009.175372. PMC 2754351. PMID 19581378.
- Case LK, Laubacher CM, Olausson H, Wang B, Spagnolo PA, Bushnell MC (May 2016). "Encoding of Touch Intensity But Not Pleasantness in Human Primary Somatosensory Cortex". The Journal of Neuroscience. 36 (21): 5850–60. doi:10.1523/JNEUROSCI.1130-15.2016. PMC 4879201. PMID 27225773.
- Van Boven, R. W.; Johnson, K. O. (1 December 1994). "The limit of tactile spatial resolution in humans: Grating orientation discrimination at the lip, tongue, and finger". Neurology. 44 (12): 2361–6. doi:10.1212/WNL.44.12.2361. PMID 7991127.
- Craig JC (1999). "Grating orientation as a measure of tactile spatial acuity". Somatosensory & Motor Research. 16 (3): 197–206. doi:10.1080/08990229970456. PMID 10527368.
- Goldreich D, Wong M, Peters RM, Kanics IM (June 2009). "A Tactile Automated Passive-Finger Stimulator (TAPS)". Journal of Visualized Experiments (28). doi:10.3791/1374. PMC 2726582. PMID 19578327.
- Stevens JC, Alvarez-Reeves M, Dipietro L, Mack GW, Green BG (2003). "Decline of tactile acuity in aging: a study of body site, blood flow, and lifetime habits of smoking and physical activity". Somatosensory & Motor Research. 20 (3–4): 271–9. doi:10.1080/08990220310001622997. PMID 14675966.
- Manning H, Tremblay F (2006). "Age differences in tactile pattern recognition at the fingertip". Somatosensory & Motor Research. 23 (3–4): 147–55. doi:10.1080/08990220601093460. PMID 17178550.
- Goldreich D, Kanics IM (April 2003). "Tactile acuity is enhanced in blindness". The Journal of Neuroscience. 23 (8): 3439–45. doi:10.1523/jneurosci.23-08-03439.2003. PMC 6742312. PMID 12716952.
- Peters RM, Hackeman E, Goldreich D (December 2009). "Diminutive digits discern delicate details: fingertip size and the sex difference in tactile spatial acuity". The Journal of Neuroscience. 29 (50): 15756–61. doi:10.1523/JNEUROSCI.3684-09.2009. PMC 3849661. PMID 20016091.
- Dillon YK, Haynes J, Henneberg M (November 2001). "The relationship of the number of Meissner's corpuscles to dermatoglyphic characters and finger size". Journal of Anatomy. 199 (Pt 5): 577–84. doi:10.1046/j.1469-7580.2001.19950577.x. PMC 1468368. PMID 11760888.
- Peters RM, Goldreich D (2013). "Tactile spatial acuity in childhood: effects of age and fingertip size". PLOS ONE. 8 (12): e84650. Bibcode:2013PLoSO...884650P. doi:10.1371/journal.pone.0084650. PMC 3891499. PMID 24454612.
- Stevens, Joseph C.; Foulke, Emerson; Patterson, Matthew Q. (1996). "Tactile acuity, aging, and braille reading in long-term blindness". Journal of Experimental Psychology: Applied. 2 (2): 91–106. doi:10.1037/1076-898X.2.2.91.
- Van Boven RW, Hamilton RH, Kauffman T, Keenan JP, Pascual-Leone A (June 2000). "Tactile spatial resolution in blind braille readers". Neurology. 54 (12): 2230–6. doi:10.1212/wnl.54.12.2230. PMID 10881245.
- Goldreich D, Kanics IM (November 2006). "Performance of blind and sighted humans on a tactile grating detection task". Perception & Psychophysics. 68 (8): 1363–71. doi:10.3758/bf03193735. PMID 17378422.
- Wong M, Gnanakumaran V, Goldreich D (May 2011). "Tactile spatial acuity enhancement in blindness: evidence for experience-dependent mechanisms". The Journal of Neuroscience. 31 (19): 7028–37. doi:10.1523/JNEUROSCI.6461-10.2011. PMC 6703211. PMID 21562264.
- Bhattacharjee A, Ye AJ, Lisak JA, Vargas MG, Goldreich D (October 2010). "Vibrotactile masking experiments reveal accelerated somatosensory processing in congenitally blind braille readers". The Journal of Neuroscience. 30 (43): 14288–98. doi:10.1523/JNEUROSCI.1447-10.2010. PMC 3449316. PMID 20980584.
- Gabriel Robles-De-La-Torre. "International Society for Haptics: Haptic technology, an animated explanation". Isfh.org. Archived from the original on 2010-03-07. Retrieved 2010-02-26.
Further reading
- Boron WF, Boulpaep EL (2003). Medical Physiology. Saunders. pp. 352–358. ISBN 0-7216-3256-4.
- Flanagan, J.R., Lederman, S.J. Neurobiology: Feeling bumps and holes, News and Views, Nature, 2001 Jul. 26;412(6845):389-91.
- Hayward V, Astley OR, Cruz-Hernandez M, Grant D, Robles-De-La-Torre G (2004). "Haptic interfaces and devices" (PDF). Sensor Review. 24 (1): 16–29. doi:10.1108/02602280410515770.
- Purves, Dale (2012). Neuroscience, Fifth Edition. Sunderland, MA: Sinauer Associates, Inc. pp. 202–203. ISBN 978-0-87893-695-3.
- Robles-De-La-Torre G, Hayward V (July 2001). "Force can overcome object geometry in the perception of shape through active touch" (PDF). Nature. 412 (6845): 445–8. doi:10.1038/35086588. PMID 11473320.
- Robles-De-La-Torre, G (2006). "The Importance of the Sense of Touch in Virtual and Real Environments" (PDF). IEEE Multimedia. 13 (3): 24–30. doi:10.1109/mmul.2006.69.
- Grunwald, M. (Ed.) Human Haptic Perception – Basics and Applications. Boston/Basel/Berlin: Birkhäuser, 2008, ISBN 978-3-7643-7611-6
- Encyclopedia of Touch Scholarpedia Expert articles
External links
