Current Intelligence Bulletin 55
On this Page
- Foreword
- Abstract
- Abbreviations
- Acknowledgments
- Introduction
- Physical and Chemical Properties
- Production, Use, and Potential for Occupational Exposure
- Exposure Limits, Standards, and Recommendations
- Evidence of Carcinogenicity in Animals
- Mutagenicity
- Human Health Effects
- Epidemiologic Studies
- Conclusions
- Recommendations
- Guidelines for Minimizing Worker Exposure
- Appendix A
- Appendix B
- Notes
- References
September 1991
DHHS (NIOSH) Publication Number 91-112
Carcinogenicity of Acetaldehyde and Malonaldehyde, and Mutagenicity of Related Low-Molecular-Weight Aldehydes
Foreword
Current Intelligence Bulletins (CIBs) are issued by the National Institute for Occupational Safety and Health (NIOSH), Centers for Disease Control, Atlanta, Georgia, to disseminate new scientific information about occupational hazards. A CIB may draw attention to a formerly unrecognized hazard, report new data on a known hazard, or disseminate information about hazard control.
CIBs are distributed to representatives of academia, industry, organized labor, public health agencies, and public interest groups as well as to Federal agencies responsible for ensuring the safety and health of workers. Copies are available to individuals upon request from the Division of Standards Development and Technology Transfer, NIOSH (Robert A. Taft Laboratories, 4676 Columbia Parkway, Cincinnati, OH 45226). We welcome suggestions concerning the content, style, and distribution of these documents.
The purpose of this bulletin is to disseminate recent information about the potential carcinogenicity of acetaldehyde and malonaldehyde. Also discussed is the mutagenicity of nine related low-molecular-weight aldehydes (acrolein, butyraldehyde, crotonaldehyde, glutaraldehyde, glyoxal, paraformaldehyde, propiolaldehyde, propionaldehyde, and valeraldehyde).
Results of recent studies in animals indicate an increased incidence of laryngeal cancer in hamsters and nasal cancer in rats following exposure to acetaldehyde. Malonaldehyde administered to rats produced an increased incidence of adenomas and carcinomas of the thyroid gland and pancreatic islet cell adenomas. Acetaldehyde is a probable metabolite in the metabolism of malonaldehyde by rat liver mitochondria. Acetaldehyde and malonaldehyde both meet the criteria of the Occupational Safety and Health Administration (OSHA) for classifying substances as potential occupational carcinogens [Title 29 of the Code of Federal Regulations, Part 1990]. NIOSH therefore considers acetaldehyde and malonaldehyde to be potential occupational carcinogens and recommends that exposures to each be reduced to the lowest feasible concentration. The potential for acetaldehyde or malonaldehyde to produce cancer in humans has not been determined, but reducing occupational exposures to these chemicals should lower the risk.
The carcinogenic potential of the nine related low-molecular-weight aldehydes has not been adequately evaluated by appropriate experimentation. However, some studies indicate that their chemical reactivity and mutagenicity are similar to those of acetaldehyde and malonaldehyde. Thus NIOSH is also concerned about occupational exposure to these nine aldehydes because they (in addition to acetaldehyde and malonaldehyde) may be used as substitutes for formaldehyde, a regulated carcinogen [Title 29 of the Code of Federal Regulations, Part 1910.1048]. For example, acetaldehyde and glyoxal have been used to replace formaldehyde in embalming fluids. Therefore, careful consideration should be given to reducing exposures to these nine related aldehydes.
NIOSH recommends that the producers and users of acetaldehyde, malonaldehyde, and the other low-molecular-weight aldehydes disseminate this information to workers and customers. NIOSH also recommends that professional and trade associations and unions inform their members about the potential hazards of working with these aldehydes and that they use all available preventive measures (including available engineering controls and work practices) to minimize the exposure of workers. Readers seeking more detailed information about the studies cited in this bulletin are urged to consult the original publications.
[signature]
J. Donald Millar, M.D., D.T.P.H. (Lond.)
Assistant Surgeon General
Director, National Institute for
Occupational Safety and Health
Centers for Disease Control
Abstract
The National Institute for Occupational Safety and Health (NIOSH) has determined that acetaldehyde and malonaldehyde are potentially carcinogenic to occupationally exposed workers. NIOSH is also concerned about exposure to nine related low-molecular-weight aldehydes: acrolein, butyraldehyde, crotonaldehyde, glutaraldehyde, glyoxal, paraformaldehyde , propiolaldehyde, propionaldehyde, and valeraldehyde.
Long-term inhalation studies of acetaldehyde produced laryngeal cancers in hamsters and nasal cancers in rats. A long-term gavage study of malonaldehyde produced adenomas and carcinomas of the thyroid gland and adenomas of the pancreatic islet cells in rats. Acetaldehyde and malonaldehyde have also been shown to be mutagenic in a variety of assays.
Adequate epidemiologic data are not available from workers exposed to acetaldehyde or malonaldehyde. However, both chemicals meet the criteria of the Occupational Safety and Administration (OSHA) for potential carcinogens [Title 29 of the Code of Federal Regulations, Part 1990]. NIOSH therefore considers acetaldehyde and malonaldehyde to be potential occupational carcinogens and recommends that worker exposure to acetaldehyde and malonaldehyde be reduced to the lowest feasible concentration.
NIOSH is concerned about the nine related aldehydes because their chemical reactivity and mutagenicity are similar to those of acetaldehyde and malonaldehyde, and they may be used as substitutes for formaldehyde, a regulated carcinogen. Although their carcinogenic potential has not been adequately evaluated by in vitro research or studies in experimental animals, careful consideration should be given to reducing occupational exposures to these low-molecular-weight aldehydes.
Abbreviations
| BP | benzo[a]pyrene |
| Ca | potential occupational carcinogen |
| CAS | Chemical Abstracts Service |
| CFR | Code of Federal Regulations |
| CHO | Chinese hamster ovary cell culture |
| CIB | Current Intelligence Bulletin |
| CL | ceiling limit |
| CPC | chemical protective clothing |
| °C | degree Celsius |
| DENA | diethylnitrosamine |
| DHHS | Department of Health and Human Services |
| DMBA | dimethylbenz[a]anthracene |
| DNA | deoxyribonucleic acid |
| DSDTT | Division of Standards Development and Technology Transfer |
| °F | degree Fahrenheit |
| g | gram |
| Hg | mercury |
| HGPRT | hypoxanthine guanine phosphoribosyl transferase |
| hr | hour |
| IARC | International Agency for Research on Cancer |
| kg | kilogram |
| LC50 | lethal concentration for 50% of the exposed animals |
| LD50 | lethal dose for 50% of the exposed animals |
| m | meter |
| MCA | Manufacturing Chemists Association |
| mg | milligram |
| min | minute |
| ml | milliliter |
| mm | millimeter |
| MSHA | Mine Safety and Health Administration |
| Na | sodium |
| NIOSH | National Institute for Occupational Safety and Health |
| NTP | National Toxicology Program |
| ODC | ornithine decarboxylation activity |
| OSHA | Occupational Safety and Health Administration |
| P | probability |
| PEL | permissible exposure limit |
| ppm | parts per million |
| RD50 | concentration eliciting a 50% decrease in respiratory rate |
| REL | recommended exposure limit |
| RNA | ribonucleic acid |
| RTECS | Registry of Toxic Effects of Chemical Substances |
| SCE | sister chromatid exchange |
| SRI | Stanford Research Institute |
| STEL | short-term exposure limit |
| TWA | time-weighted average |
| UDS | unscheduled DNA synthesis |
| USITC | United States International Trade Commission |
| wk | week |
| yr | year |
Acknowledgments
This document was prepared by the staff of the Division of Standards Development and Technology Transfer, Richard W. Niemeier, Ph.D., Director. Brenda K. Boutin developed the document. The contributions of other National Institute for Occupational Safety and Health (NIOSH) personnel are gratefully acknowledged: David D. Bayse, Ph.D.; Philip J. Bierbaum; Alexander Cohen, Ph.D.; Michael L. Colligan, Ph.D.; Jerome P.Flesch; Bryan D. Hardin, Ph.D.; Kent Hatfield, Ph.D.; Robert F. Herrick, Ph.D.; Richard A.Lemen, Ph.D.; Howard R. Ludwig; Pervis C. Major, Ph.D.; Dannie C. Middleton, M.D.; William H. Perry; Laurence Reed; Joseph A. Seta; David M. Votaw; Joann A. Wess; John J.Whalen; and Ralph D. Zumwalde.
Ruth E. Grubbs and Anne C. Hamilton performed editorial review and coordinated production; Vanessa L. Becks, Geoffrey Buesing, and Susan R. Kaelin provided editorial assistance and produced camera-ready copy; and Judith G. Curless, Linda Dodgen, Denise Hill, and Susan Sebree formatted and typed the draft.
We also wish to thank the following former NIOSH employees for their contributions to this document: Burt J. Cooper; Hugh J. Hansen, Ph.D.; Jane B. McCammon; Robert H. Schutz, Ph.D.; and William D. Wagner.
Carcinogenicty of Acetaldehyde and Malonaldehyde, and Mutagenicity of Related Low-Molecular-Weight Aldehydes
September 1991
Introduction
This Current Intelligence Bulletin (CIB) presents recent information about the potential carcinogenicity and mutagenicity of acetaldehyde and malonaldehyde (synonym, propanedial); the document also discusses the chemical reactivity and mutagenicity of nine related low-molecular-weight aldehydes: acrolein, butyraldehyde, crotonaldehyde, glutaraldehyde, glyoxal, propiolaldehyde, propionaldehyde, and valeraldehyde. Guidelines are included for minimizing occupational exposures.
Published inhalation studies of acetaldehyde have shown the production of laryngeal carcinomas in male and female hamsters and carcinomas and adenocarcinomas, of the nasal cavity in male and female rats. Malonaldehyde administered to rats by gavage produced an increase in adenomas and carcinomas of the thyroid gland in males and females and an increase in adenomas of the pancreatic islet cells in male rats. Research on the carcinogenic potential of the nine related aldehydes is not complete, but data suggest that their chemical reactivity and mutagenicity are similar to those of acetaldehyde, malonaldehyde, and formaldehyde. NIOSH is therefore concerned about possible increases in the occupational use of all nine compounds as substitutes for formaldehyde, a carcinogen recently regulated by the Occupational Safety and Health Administration (OSHA) [29 CFR* 1910.1048]. For example, acetaldehyde and glyoxal have been used to replace formaldehyde in embalming fluids [CRCS 1984]. Aldehydes such as glyoxal and malonaldehyde [Ura et al. 1983] may be used to replace formaldehyde as an intermediate for resins employed to treat paper [IARC 1982]. In addition, acetaldehyde, malonaldehyde, acrolein, butyraldehyde, crotonaldehyde, glutaraldehyde, and propionaldehyde have been used as reagents in laboratories [Auerbach et al. 1977]. Therefore, careful consideration should be given to reducing exposure to these aldehydes.
Physical and Chemical Properties
Acetaldehyde is a two-carbon compound with a carbonyl group (HC=O) (see Figure 1) and a molecular weight of 44.05. This clear liquid boils at 20.8°C (69.5°F). Its pleasant, fruity odor at dilute concentrations becomes pungent and suffocating at high concentrations. Acetaldehyde is readily oxidized within the body to acetate and acetic acid by liver mitochondrial acetaldehyde dehydrogenase [Lubin and Westerfeld 1945; Parrilla et al. 1974].
Monoaldehyde is a three-carbon compound with two carbonyl groups (see Figure 1) and a molecular weight of 72.06 (propanedial, sodium salt with a molecular weight of 94.04). The chemical and physical properties of malonaldehyde are similar to acetaldehyde and other low-molecular-weight monoaldehydes and dialdehydes [Brabec 1981]. Pure malonaldehyde is unstable and is precipitated as a sodium salt immediately before its use in bioassays [Huttel 1941; Marnett and Tuttle 1980]. Recent studies of purity analysis by ultraviolet spectroscopy established that malonaldehyde (propanedial, sodium salt) was stable with no notable degradation for 2 yr when stored at -20°C [NTP 1988]. Malonaldehyde is metabolized in vivo and in vitro by oxidation to malonic semialdehyde and by decarboxylation to acetaldehyde [Siu and Draper 1982]. The physical and chemical properties of acetaldehyde and malonaldehyde are presented in Table 1.
Figure 1.
Structures for Acetaldehyde, Molonaldehyde, and Nine Related Low-Molecular-Weight Aldehydes.
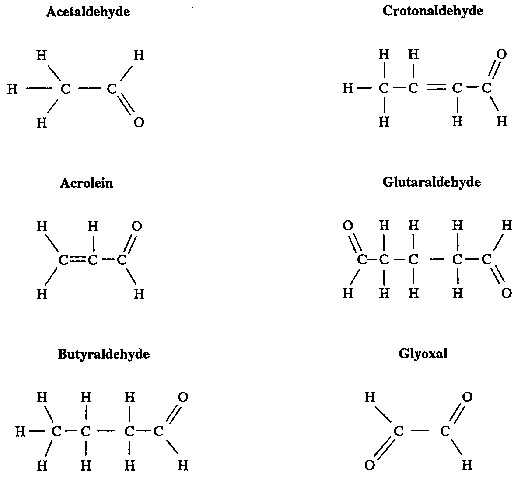
Figure 1 (Continued)
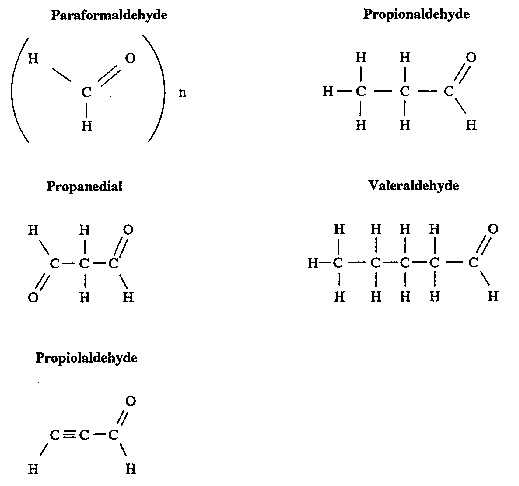
Acetaldehyde, malonaldehyde, and the other low-molecular-weight aldehydes included in this CIB are highly reactive intermediates in the oxidation of alcohols to acids [Henson 1959]. The carbonyl groups of aldehydes (see Figure 1) can react by condensation with endogenous primary amines or amines generated by metabolic N-dealkylation to form azomethine compounds (-C=N-, Schiff bases) [Hawley 1977; Hemminki and Suni 1984; O'Donnell 1982]. The reactions of aldehydes with biogenic compounds can generate products with altered reactivity or toxicity. For example, salsolinol (an enzyme inhibitor) is the condensation product of dopamine and acetaldehyde [Collins and Bigdeli 1975; Ellingboe 1978]. Acetaldehyde has been reported to bind covalently to a variety of proteins [Tuma and Sorrell 1985] and to form adducts with phospholipids [Kenney 1982, 1984] and nucleosides, [Hemminki and Suni 1984].
The physical and chemical properties of the other nine low-molecular-weight aldehydes are presented in Appendix A.
Production, Use, and Potential for Occupational Exposure
Acetaldehyde
In 1982, 280,000 tons of acetaldehyde were produced in the United States [IARC 1985a]. The Hoechst-Wacker two-stage process is the main production method used in the United States for preparing acetaldehyde from ethylene. In this process, acetaldehyde is generated during the oxidation of ethylene by aqueous palladium chloride. The reduced palladium is reoxidized by cupric chloride to palladium chloride, and the reduced cuprous chloride that is formed is reoxidized by oxygen. The overall reaction yields acetaldehyde [SRI 1985].
Acetaldehyde is primarily used as a substrate for acetic acid manufacture, although this use is declining as more economical chemical intermediates are replacing acetaldehyde [SRI 1979]. This chemical is also used in the synthesis of pyridine and pyridine bases, peracetic acid, pentaerythritol, 1,3-butylene glycol, and chloral. In addition, acetaldehyde is used in the silvering of mirrors, in leather tanning, as a denaturant for alcohol, in fuel compositions, as a hardener for gelatin fibers, in glue and casein products, as a preservative for fish, in the paper industry, and in the manufacture of cosmetics, aniline dyes, plastics, and synthetic rubber [Fishbein 1979; IARC 1985a; Hoff 1982; Windholz et al. 1983].
NIOSH estimates that approximately 14,000 workers are exposed to acetaldehyde [NIOSH 1983a]. This estimate is based on field survey data from workers potentially exposed during the handling of acetaldehyde. The total number of exposed workers may be underestimated because this figure omits any estimate of potential exposure in workplaces where acetaldehyde is used in tradenamed or proprietary products.
Malonaldehyde
Purified malonaldehyde is subject to polymerization and degradation [Huttel 1941] and is usually generated shortly before use [Marnett and Tuttle 1980]. Malonaldehyde or its enolic sodium salt is primarily used in research laboratories where it may be generated by acid hydrolysis of 1,1,3,3-tetraethoxypropane or 1,1,3,3-tetramethoxypropane. Malonaldehyde may be purified by column separation and acetone precipitation [Bird et al. 1982; Marnett et al. 1979; NTP 1988; Protopopova and Skoldinov 1958]. Although this process can generate a 99 % pure malonaldehyde (propanedial, sodium salt), hydrolytic intermediates (ß-ethoxyacrolein, ß-methoxy-acrolein, 3,3-diethoxypropionaldehyde, and 3,3-dimethoxypropionaldehyde) may also be generated [Marnett and Tuttle 1980]. The United States International Trade Commission (USITC) has no separate listing for the production of malonaldehyde or its precursors in the United States. The annual production of malonaldehyde varies because it is produced on an as-needed basis by several U.S. chemical manufacturers [USITC 1986].
No figures are available for the number of workers exposed to malonaldehyde in the United States.
Table 1
Physical and Chemical Properties of Acetaldehyde and Malonaldehyde *
| Item | Acetaldehyde | Malonaldehyde | Propanedial (sodium salt) |
|---|---|---|---|
| CAS** registry no. | 75-07-0 | 542-78-9 | 24382-04-5 |
| RTECS*** no. | AB1925000 | TX6475000 | ON8930000 |
| Odor threshold | 0.21 ppm | NA† | NA |
| Molecular formula | C2H4O | C3H4O2 | C3H3O2Na |
| Molecular weight | 44.05 | 72.06 | 94.04 |
| Boiling point (760 mm Hg) | 20.8°C (69.5°F) | NA | NA |
| Melting point | -121°C (-185.8°F) | 72°C (161°F) | NA |
| Specific gravity (g/ml) | 0.788 at 20°C | NA | NA |
| Refractive index | 1.3316 | NA | NA |
| Vapor pressure (mm Hg @ 20°C) | 740 | NA | NA |
| Flash point: | |||
| Open cup | -40°C (-40°F) | NA | NA |
| Closed cup | -38.89°C (-38°F) | NA | NA |
| Temperature of autoignition | 175°C (347°F) | NA | NA |
| Explosive limit in air, vol. % | 4-57 | NA | NA |
| Solvents | Water, alcohol, ether, acetone, benzene, toluene, solvent naphtha, turpentine. gasoline |
Water | Water |
Related Low-Molecular-Weight Aldehydes
Appendix B lists the production estimates and potential number of workers exposed to the nine low-molecular-weight aldehydes discussed in this CIB.
Exposure Limits, Standards, and Recommendations
Acetaldehyde
In recent rulemaking on air contaminants [54 Fed. Reg.** 2445 (1989)], OSHA established a permissible exposure limit (PEL) of 100 parts of acetaldehyde per million parts of air (100 ppm, or 180mg/m3) as an 8-hr time-weighted average (TWA) with a short-term exposure limit (STEL) of 150 ppm (270 mg/m3). The PEL is based on the results of an acute toxicity study [Silverman et al. 1946] and is intended to protect workers against the risk of conjunctivitis and sensory irritation.
NIOSH has not established a recommended exposure limit (REL) for acetaldehyde. In testimony at the OSHA hearings on this rulemaking, NIOSH [1988a] did not concur with the proposed PEL, citing acetaldehyde in a group of 53 chemicals that should be designated as potential occupational carcinogens for which a substantial level of risk remains at the proposed PEL.
The Mine Safety and Health Administration (MSHA) has proposed a PEL of 100 ppm (180 mg/m3) as an 8-hr TWA for acetaldehyde with a STEL of 150 ppm (270 mg/m3) [MSHA 1989].
The International Agency for Research on Cancer (IARC) has concluded that there is sufficient evidence that acetaldehyde is carcinogenic to experimental animals but that there is inadequate evidence that acetaldehyde is carcinogenic to humans [IARC 1987]. The OSHA Hazard Communication Standard [29 CFR 1910.1200, 1915.98, 1917.28, 1918.90, 1926.59] requires that chemical manufacturers, distributors, employers, and importers provide appropriate hazard warnings for carcinogens or potential carcinogens listed in the latest editions of IARC Monographs.
Malonaldehyde
NIOSH, OSHA, and MSHA have not established exposure limits for malonaldehyde. In 1985, IARC concluded that there was inadequate evidence to establish the carcinogenicity of malonaldehyde in experimental animals and that there were no epidemiologic data to evaluate the carcinogenic risk of malonaldehyde to humans [IARC 1985b].
Related Low-Molecular-Weight Aldehydes
Appendix B lists the OSHA and MSHA standards and the NIOSH recommendations for the nine low-molecular-weight aldehydes discussed in this CIB
Evidence of Carcinogenicity in Animals
Acetaldehyde
Woutersen et al. [1986]
Woutersen et al. [1986] reported evidence for the carcinogenicity of acetaldehyde in their chronic inhalation study. Three groups (each composed of 55 male and 55 female Wistar rats) were exposed to acetaldehyde vapor 6 hr/day, 5 days/wk for 28 months at mean concentrations of 0, 735, or 1,412 ppm. A fourth group of the same size was initially exposed to 3,033 ppm. By day 360, exposure of the latter group was gradually reduced to a final concentration of 977 ppm because of severe growth retardation, loss of body weight, and early mortality. The study was terminated after 120 to 122 wk. A concentration-related incidence of squamous cell carcinoma of the nasal cavity occurred in both male and female rats. This increased incidence was statistically significant (P<0.001) in males and females exposed to 977 to 3,033 ppm and in males exposed to 1,412 ppm (P<0.05). These carcinomas infiltrated the submucosa of the nasal epithelium, filled one or both sides of the nasal cavity, destroyed turbinates, invaded the nasal bones, and extended into the subcutis and brain. Adenocarcinomas occurred in statistically significant numbers in the nasal cavities of rats of both sexes exposed to all three concentrations of acetaldehyde (P<0.001, except P<0.05 in female rats exposed to 735 ppm). The adenocarcinomas ranged in size from small groups of atypical cells in the olfactory epithelium that invaded only nerve bundles, to large osteolytic tumors that grew into the subcutis outside the nose and invaded the cerebrum through the olfactory lobe. The authors concluded that the nasal tumors arose from the respiratory and olfactory epithelia that were damaged by acetaldehyde.
Feron et al. [1982]
Feron et al. [1982] conducted inhalation studies in which male and female Syrian golden hamsters were exposed (7 hr/day, 5 days/wk) to a gradual reduction of acetaldehyde vapor as follows:
| Average vapor concentration, in ppm (mg/m3) | Exposure period (study weeks) |
|---|---|
| 2,500 (4,500) | 1-9 |
| 2,250 (4,050) | 10-20 |
| 2,000 (3,600) | 21-29 |
| 1,800 (3,240) | 30-44 |
| 1,650 (2,970) | 45-52 |
An equal number of control animals received filtered and conditioned air. Hamsters of the acetaldehyde-exposed and the air-control groups were further subdivided into five treatment groups (Table 2). Inhalation exposure to acetaldehyde and benzo[a]pyrene (BP) or diethylnitrosamine (DENA) treatments were conducted simultaneously for 52 wk, and the study ended at wk 81 after a 29-wk recovery period. A statistically significant increase (P<0.05) occurred in the incidence of combined laryngeal tumors (carcinoma in situ, squamous cell carcinoma, and adeno-squamous carcinoma) in hamsters of both sexes exposed to acetaldehyde vapor when compared with unexposed, pooled controls (Groups 1 and 2).
A statistically significant increase (P<0.05) occurred in the incidence of squamous cell carcinoma in the trachea, larynx, and bronchi of males instilled with a total BP dose of 36.4 mg/hamster and exposed to acetaldehyde compared with males instilled with the same dose of BP and exposed to air (Group 4). An increased incidence of squamous cell carcinoma of the trachea (statistical significance was not given) occurred in females instilled with 0.35% BP and exposed to acetaldehyde compared with females instilled with the same dose of BP and exposed to air (Group 4). Males and females exposed to acetaldehyde and instilled with a total BP dose of 18.2 mg/hamster had a statistically increased incidence (P<0.05) of squamous cell carcinoma of the larynx compared with controls instilled with the same dose of BP (Group 3).
Table 2
Acetaldehyde Inhalation, BP Instillations, and DENA Subcutaneous Injections of Hamsters for 52 Weeks
| Treatment group | Number of hamsters* |
Type of weekly instillation |
Subcutaneous injections (total of 17) |
| Group 1: | |||
| Group exposed to acetaldehyde | 36 | -- | -- |
| Control group | 36 | -- | -- |
| Group 2: | |||
| Group exposed to acetaldehyde | 36 | Saline | -- |
| Control Group | 36 | Saline | -- |
| Group 3: | |||
| Group exposed to acetaldehyde | 60 | 0.175% BP** | -- |
| Control group | 60 | 0.175% BP | -- |
| Group 4: | |||
| Group exposed to acetaldehyde | 60 | 0.35% BP*** | -- |
| Control group | 60 | 0.35% BP | -- |
| Group 5: | |||
| Group exposed to acetaldehyde | 60 | -- | 0.0625% DENA† |
| Control group | 60 | -- | 0.0625% DENA |
* Each group consisted of equal numbers of male and female hamsters [Feron et al. 1982]. [return to table]
**Total of 18.2 mg BP/hamster. [return to table]
***Total of 36.4 mg BP/hamster. [return to table]
†Total of 0.0021 mg DENA/hamster. [return to table]
In hamsters exposed to acetaldehyde and subcutaneously injected with a total of 0.0021 mg DENA/hamster, the incidence of carcinoma in situ of the larynx was increased in males and the incidences of carcinoma in situ and squamous cell carcinoma of the larynx in females were statistically increased (P<0.05) compared with the controls exposed to air and injected with an equivalent dose of DENA (Group 5). The incidence of tumors in the control animals treated with BP or DENA and exposed to air were not different from the controls treated with saline and exposed to air.
Malonaldehyde
NTP [1988]
The National Toxicology Program (NTP) [1988] conducted a 2-yr study of malonaldehyde exposure in F344/N rats and B6C3F1 mice. Highly purified anhydrous malonaldehyde (propanedial, sodium salt) was administered by gavage 5 days/wk for 103 wk at doses of 0, 50, and 100 mg/kg (in distilled water) to groups of 50 male and 50 female rats. Doses of 0, 60, and 120 mg/kg (in distilled water) were administered by gavage to groups of 50 male and 50 female mice on the same schedule. There was no evidence of carcinogenic activity in the mice. The incidence of thyroid gland follicular cell hyperplasia was statistically increased (P<0.001) in the female rats given 100 mg malonaldehyde/kg compared with the vehicle controls. In male and female rats that received 100 mg malonaldehyde/kg, the incidence of follicular cell adenomas and carcinomas (combined) was statistically increased (P<0.05) when compared with that of the corresponding vehicle controls. The incidence of pancreatic islet cell adenomas was statistically increased (P=0.001) in male rats receiving 50 mg malonaldehyde/kg compared with the vehicle controls. Incidences of pancreatic islet cell tumors in female rats were not significantly different from those observed in the female vehicle controls.
The NTP study concluded that the increased incidences of follicular cell adenomas and carcinomas (combined) of the thyroid gland provided clear evidence of carcinogenic activity for male and female F344/N rats administered malonaldehyde (propanedial, sodium salt). Pancreatic islet cell adenomas were also observed at a significantly increased incidence in low-dosed male rats.
Shamberger et al. [1974, 1975]
In a two-stage mouse-skin initiation assay [Shamberger et al. 1974, 1975], groups of 30 female Swiss mice, 55 days old, were treated with one application of 6 or 12 mg malonaldehyde in acetone or with 0.125 mg of 7,12-dimethyl-benz[a]anthracene (DMBA) in acetone to their shaved backs. After 3 wk, the backs of the mice were treated for 27 wk (5 days/wk) with 0.1% croton oil in acetone. Control groups received either no treatment or one application of the following: malonaldehyde, DMBA, or acetone. By the end of the 30-wk experiment, 52% of the mice receiving malonaldehyde and croton oil had tumors, and 95% of the mice treated with DMBA and croton oil had tumors. No tumors appeared in the control groups. Statistical significance was not given.
In a second experiment, 30 female Swiss mice were treated with 12 mg malonaldehyde daily for 9 wk [Shamberger et al. 1974]. Because this concentration was toxic, daily skin application was reduced to 0.36 mg malonaldehyde for an additional 39 wk. No tumors occurred in the 12 mice that died during the first 4 to 6 wk of the experiment. Five of the six mice that died during wk 7 through 9 had tumors, including four liver carcinomas (three of which were metastatic to the kidney, rectum, or lung) and one rectal carcinoma. No other tumors were seen through wk 48, and no tumors were seen in the control animals. Statistical significance was not given.
Mutagenicity
Acetaldehyde and Malonaldehyde
Most chemicals known to cause cancer are also capable of causing a change within the genetic material of a cell (mutation) [Ames et al. 1975]. Acetaldehyde and malonaldehyde have been shown to induce mutagenic changes in a variety of assays. Both have produced sister chromatid exchanges (SCEs), chromosomal breaks and chromosomal aberrations in in vitro mammalian assays; and both have caused mutations in Escherichia coli bacterial systems and Drosophila melanogaster. The mutagenic responses listed in Tables 3 and 4 provide supporting evidence for the carcinogenic potential of acetaldehyde and malonaldehyde reported in animals.
Related Low-Molecular-Weight Aldehydes
Overview
The carcinogenic potential of the nine related aldehydes has not been adequately evaluated by in vivo and in vitro experimentation. However, NIOSH selected these aldehydes of five or fewer carbon atoms to be included in this CIB because
- their chemical structures, chemical reactivity, and mutagenicity are similar to acetaldehyde, malonaldehyde, and formaldehyde, and
- their commercial production levels and their potential for worker exposure are significant.
Also, the potential exists for simultaneous exposure to several aldehydes in the same occupational setting. Furthermore, any of the related aldehydes, might be substituted for acetaldehyde, or formaldehyde.
Although formaldehyde is also a low-molecular-weight aldehyde similar to the aldehydes discussed in this CIB, it was not included in this discussion because it has been extensively reviewed [NIOSH 1976a, NIOSH/OSHA 1980]. NIOSH considers formaldehyde to be a potential occupational carcinogen [Lemen 1987]. In 1988, OSHA promulgated a revised occupational safety and health standard for occupational exposure to for maldehyde. This standard states that formaldehyde has the potential to cause cancer in humans [ 29 CFR 1910.1048].
This subsection summarizes the mutagenicity studies for the nine low-molecular-weight aldehydes. Appendix C contains additional information about their toxic effects.
Mutagenicity Summaries
The highest concentration of acrolein administered in the drinking water of male and female rats produced a statistically significant in crease (P=0.022) in adenomas and hyperplastic nodules of the adrenal cortex in female rats [Lijinsky and Reuber 1987]. In another study [Feron and Kruysse 1977], exposure to acrolein vapor induced epithelial metaplasia in the nasal cavities of hamsters. Hamsters exposed to acrolein vapor and instilled with BP had more respiratory tract tumors than BP-instilled hamsters exposed to air (statistical value was not given). The authors concluded that their study did not provide sufficient evidence to indicate that acrolein enhanced the carcinogenic effect of BP [Feron and Kruysse 1977].
Acrolein was also mutagenic in Salmonella typhimurium assays [Haworth et al. 1983; Lutz et al. 1982; Marnett et al. 1985], and it inhibited the activity of DNA polymerases [Munsch et al. 1973]. NTP [1991] is currently evaluating several aldehydes for toxicity testing, and acrolein has been selected for general toxicity testing.
Butyraldehyde induced chromosomal damage and meiotic anomalies in male mice during spermatogenesis [Moutschen-Dahmen et al. 1976]. NTP [1991] has completed a prechronic gavage and a prechronic inhalation study of butyraldehyde in mice and rats. Isobutyraldehyde, an isomer of butyraldehyde, has been tested in a prechronic inhalation study, and a 2-yr inhalation study in rats and mice is now in progress.
Table 3
Summary of Positive Mutagenic Responses to Acetaldehyde
| Type of mutation | Organism | References |
| SCE* | Human lymphocytes | He and Lambert [1985] Jansson [1982] Knadle [1985] Norppa at al. [1985] Ristow and Obe [1978] Veghelyi and Osztovics [1977] |
| CHO** | Brambilla et al. [1986] Obe and Beek [1979] |
|
| Chinese hamster bone marrow | Korte and Obe [1981] | |
| CBA mouse chromosomes | Obe et al. [1979] | |
| Chromosomal aberrations | Rat skin fibroblasts Human lymphocytes Rat fetuses |
Bird et al. [1982] Obe et al. [1979] Barilyak and Kozachuk [1983] |
| DNA cross-links | Human lymphocytes Rat nasal mucosa CHO Calf thymus |
Lambert et al. [1985] Lain et al. [1986] Marinari et al. [1984] Ristow and Obe [1978] |
| Zone inhibition | Escherichia coli pol A1- | Rosenkranz [1977] |
| Sex-linked recessive lethal mutation | Drosophila melanogaster | Woodruff et al. [1985] |
| Inhibited RNA and DNA synthesis | Human bronchial epithelial cells | Saladino et al. [1985] |
| Inhibited DNA synthesis | Reuber rat hepatoma (H35)and (Hep 10C) | Dreosti et al. [1981] |
| Inhibited protein and RNA synthesis | Rat pancreatic acini | Majumdar et al. [1996] |
| Initiation | C3H/10T1/2 mouse cells | Abernethy et al. [1982] |
**CHO: Chinese hamster ovary cell culture. [ return to table]
Table 4
Summary of Positive Mutagenic Responses to Malonaldehyde
| Type of mutation | Organism | References |
| SCE* | CHO** | NTP [1988] |
| Increased reversion and forward mutation | Escherichia coli H/r30 | Yonei and Furui [1981] |
| Frameshift or base-pair | Salmonella typhimurium histidine-deficient mutants | Goldstein et al. [1975] |
| HisD3052, hisC3076, hisC207, TA1976, TA1977, TA1978 | Mukai and Goldstein [1976] | |
| TA1975, hisC3076, hisD3052, TA1978, hisC207, TA1977 | Shamberger [1980] | |
| HisC207, hisC3076, hisD3052, TA1977 | Marnett and Tuttle [1980] | |
| TA102, TA104 | Marnett et al. [1985] | |
| HisD3052 | Basu and Marnett [1983] | |
| Thymidine resistance | Murine lymphoma L5178Y | NTP [1988] |
| Thymidine and methotrexate resistance | Murine lymphoma L5178Y | Yau [1979] |
| Chromosomal aberrations | Primary rat skin fibroblasts | Bird et al. [1982] Draper et al. [1986] |
| Inhibited DNA, RNA and protein synthesis | Primary rat skin fibroblasts | Bird and Draper [1980] |
| DNA cross-links | CHO-K1 E. coli Plasmid pBR322 |
Marinari et al. [1984] Yonei and Furui [1981] Basu et al. [1984] |
| Inhibited DNA and protein synthesis | Human fibroblasts | Brooks and Klamerth [1968] |
| Wing mosaicism | Drosophila melanogaster | Szabad et al. [1983] |
**CHO: Chinese hamster ovary cell culture. [ return to table]
Crotonaldehyde administered in the drinking water of rats induced a statistically significant incidence (P<0.001) of well circumscribed, noninvasive, and nonmetastatic hepatocellular neoplasm [Chung et al. 1986]. Crotonaldehyde was mutagenic in S. typhimurium assays [Marnett et al. 1985; Neudecker et al. 1981; Haworth et al. 1983; Lutz et al. 1982]. This aldehyde induced chromosomal damage and meiotic anomalies in male mice during spermatogenesis [Moutschen-Dahmen et al. 1976] and sex-linked recessive lethal mutations in D. melanogaster [Woodruff et al. 1985]. NTP [1990] has completed the prechronic testing of crotonaldehyde by corn oil gavage in rats and mice.
Glutaraldehyde was mutagenic in S. typhimurium assays [Marnett et al. 1985; Haworth et al. 1983; Levin et al. 1982]. NTP [1990] canceled the original skin painting study of rats and mice with glutaraldehyde. Chronic testing of glutaraldehyde is planned by NTP following a prechronic inhalation study.
Glyoxal induced unscheduled DNA synthesis and increased ornithine decarboxylase activity in the pyloric mucosa, of rats, indicating a potential initiating activity in carcinogenesis of the stomach [Furihata. et al. 1985]. Glyoxal was mutagenic in S. typhimurium assays [Sasaki and Endo 1978; Yamaguchi and Nakagawa 1983; Levin et al. 1982; Marnett et al. 1985] and in an E. coli mutagenesis assay [Chopra 1966]. Glyoxal also induced sex-linked recessive lethal mutations in D. melanogaster [Barnett and Munoz 1969]. A prechronic study is in progress with glyoxal by the oral water route in rats and mice [NTP 1991].
Paraformaldehyde induced transformation in rat embryo cells infected with Rauscher leukemia virus [Traul et al. 1981].
Propiolaldehyde was mutagenic in S. typhimurium assays [Basu and Marnett 1983].
Propionaldehyde and valeraldehyde induced single-strand breaks in Chinese hamster ovary cells [Marinari et al. 1984]. Valeraldehyde tested negative in the S. typhimurium assay, and no other testing is scheduled [NTP 1990].
There is evidence that the nine related aldehydes produce DNA breaks and alterations. Acetaldehyde and malonaldehyde have been shown to induce similar alterations [Lambert et al. 1985; Marinari et al. 1984]. Most chemicals known to cause cancer are also capable of causing a change in the genetic material within a cell (mutation) [Ames et al. 1975]. DNA damage has been related to mutagenicity and is a sensitive indicator of potential chemical carcinogenesis [Goldstein et al. 1975; Lutz 1979]. The interaction of the aldehyde chemical group with DNA, RNA, or enzymes involved in DNA replication, resulting in a change in genetic material, is a possible mechanism for the carcinogenicity of acetaldehyde, malonaldehyde, and formaldehyde [Feron et al. 1982; Chio and Tappel 1969; Auerbach et al. 1977]. The mutagenic effects of the nine related aldehydes are similar to those produced by acetaldehyde and malonaldehyde and thus may indicate carcinogenic potential.
Human Health Effects
Acute exposure of humans to 50 ppm acetaldehyde vapor for 15 min produced mild eye irritation; 200 ppm for 15 min produced bloodshot eyes and reddened eyelids; and 135 ppm. for 30 min produced mild irritation of the upper respiratory tract [Silverman et al. 1946; Sim and Pattle 1957]. Acetaldehyde splashed in the eyes is reported to cause painful but superficial injury to the cornea [Grant 1974; McLaughlin 1946]. Systemic effects resulting from chronic acetaldehyde exposure in the workplace have not been reported, but prolonged exposure to acetaldehyde may produce drowsiness [MCA, Inc. 1952].
Young male students infused intravenously with a 5 % solution of acetaldehyde experienced a linear increase in acetaldehyde concentration in the blood. The infusion of acetaldehyde resulted in a marked increase in ventilation and pulse rate and a decrease in alveolar carbon dioxide [Asmussen et al. 1948].
No human health effects have been reported for malonaldehyde.
Epidemiologic Studies
Between 1967 and 1972, an epidemiologic study was conducted in an East German chemical plant to assess the risk of cancer associated with exposure to aldol and aliphatic aldehydes produced from the dimerization of acetaldehyde [Bittersohl 1975]. Workers were reported to have suffered from repeated irritation of the eyes and upper respiratory tract as a result of exposure to acetaldehyde, butyraldehyde, crotonaldehyde, higher condensed aldehydes, and traces of acrolein. Investigators examined the medical records of 150 workers who had worked for the company for 20 yr, and 9 cases of malignant neoplasm were identified. Although specific mortality data (observed versus expected) were not presented, deaths resulting from malignant neoplasm of the respiratory tract and oral cavity occurred at rates above that of the national average of the German Democratic Republic and at rates higher than those observed for other chemical industries in that nation. The increased risk of cancer was attributed to exposure to acetaldehyde and possibly to other aldehydes (e.g., acrolein, butyraldehyde, and crotonaldehyde) in the work environment. However, the study had a number of limitations: the sample size was small; all cancer patients identified were smokers; and although records of 150 workers were initially examined, the loss to followup and the actual number of workers included in the study were not stated. IARC considered the study to be inadequate for evaluating the carcinogenicity of acetaldehyde to humans [IARC 1985a].
In NIOSH health hazard evaluations conducted from 1976 to 1988, surveys of workers potentially exposed to aldehydes and other chemical contaminants revealed that the airborne concentrations of acetaldehyde in these workplaces were below the analytical detection limit [NIOSH 1976b, 1977, 1980a, 1980b, 1980c, 1981, 1982, 1983b, 1988b; NIOSH/OSHA 1981].
Conclusions
The data presented in this CIB on the production of nasal tumors in rats and laryngeal tumors in hamsters exposed to acetaldehyde provide sufficient evidence to conclude that acetaldehyde is carcinogenic in laboratory animals. NTP reported that there was clear evidence of carcinogenic activity in rats administered malonaldehyde, as demonstrated by the increased incidence of follicular cell adenomas and carcinomas (combined) of the thyroid gland and pancreatic islet cell adenomas. The numerous mutagenic responses reported for both acetaldehyde and malonaldehyde are in accordance with the evidence for carcinogenicity.
Evidence for the potential carcinogenicity of the other nine aldehydes is inconclusive. All have been shown to produce cellular mutation, cause chromosomal damage, induce cellular transformation, or induce neoplasms. These mutagenic effects are similar to those produced by acetaldehyde and malonaldehyde, and they may indicate carcinogenic potential. Additional experimental research is needed to clarify the carcinogenic potential of these nine aldehydes.
Recommendations
Several systems exist for classifying a substance as a carcinogen. Such classification systems have been developed by NTP [1989], IARC [1985a], and OSHA [29 CFR 1990]. NIOSH considers the OSHA classification system ("Identification, Classification, and Regulation of Potential Occupational Carcinogens" [29 CFR 1990], also known as the OSHA carcinogen policy) the most appropriate for use in identifying potential occupational carcinogens.***
Exposure to acetaldehyde has produced nasal tumors in rats and laryngeal tumors in hamsters, and exposure to malonaldehyde has produced thyroid gland and pancreatic islet cell tumors in rats. NIOSH therefore recommends that acetaldehyde and malonaldehyde be considered potential occupational carcinogens in conformance with the OSHA carcinogen policy. The excess cancer risk for workers exposed to acetaldehyde or malonaldehyde has not been established, but the probability of developing cancer should be decreased by minimizing exposure. As a matter of prudent public health policy, employers should assess the conditions under which workers may be exposed to acetaldehyde or malonaldehyde and take reasonable precautions (such as appropriate engineering and work practice controls) to reduce exposures to the lowest feasible concentration.
Testing has not been completed to determine the carcinogenicity of the nine related low-molecular-weight aldehydes discussed in this CIB (acrolein, butyraldehyde, crotonaldehyde, glutaraldehyde, glyoxal, paraformaldehyde, propiolaldehyde, propionaldehyde, and valeraldehyde). However, the limited studies that have been conducted indicate that these chemicals have chemical reactivity and mutagenicity similar to acetaldehyde, malonaldehyde, and formaldehyde. Therefore, careful consideration should be given to reducing exposures to these nine related aldehydes.
Guidelines for Minimizing Worker Exposure
The following guidelines for minimizing worker exposure to the aldehydes discussed here are general and should be adapted to specific work situations as required.
Exposure Monitoring
NIOSH recommends that each employer who manufactures, transports, packages, stores, or uses aldehydes in any capacity determine whether a potential exists for any worker to be exposed to the chemical. In work areas where exposures may occur, an initial survey should be done to determine the extent of worker exposure.
Samples should be collected over a full shift to determine exposures. When the potential for exposure is periodic, short-term sampling may be needed to replace or supplement full-shift sampling. Personal sampling is preferred to area sampling. If personal sampling is not feasible, area sampling can be substituted only if the results can be used to approximate the workers' exposure. Sampling should be used to (1) identify the sources of emissions so that effective engineering or work practice controls can be instituted, and (2) ensure that controls already in place are operational and effective.
The NIOSH Manual of Analytical Methods provides detailed descriptions of sampling and analytical techniques for aldehydes (screening) (Method 2539 [NIOSH 1989]), acetaldehyde (Method 3507 [NIOSH 1986b] and Method 2538 [NIOSH 1989]), acrolein (Method 2501 [NIOSH 1986b]), glutaraldehyde (Method 2532 [NIOSH 1986b] and Method 2531 [NIOSH 1989]), and valeraldehyde (Method 2536 [NIOSH 1989]).
The NIOSH Occupational Exposure Sampling Strategy Manual [Leidel et al. 1977] provides guidance for developing efficient strategies to monitor worker exposures to toxic chemicals. The manual contains information about determining the need for exposure monitoring, the number of samples to be collected, and the appropriate sampling times.
Controlling Worker Exposure
Equipment maintenance and worker education are vital aspects of a good exposure control program. Workers should be informed about any materials that may contain or be contaminated with aldehydes, the nature of the potential hazard, and methods for minimizing exposure. Every attempt should be made to minimize exposure to aldehydes by using the following work practices and controls: emergency procedures, product substitution, closed systems and ventilation, worker isolation, personal protective equipment (such as chemical protective clothing [CPC] and equipment, and respiratory protection devices), and proper decontamination and waste disposal. Medical screening is also recommended under certain circumstances.
Emergency Procedures
In the event of an accidental exposure to an aldehyde, the victim should be removed from further exposure, medical assistance should be sought, and appropriate emergency procedures should be followed as described below [NIOSH/OSHA 1981]:
- Eye exposure: If an aldehyde or a solution containing an aldehyde gets into the eyes, immediately flush them with large amounts of water for a minimum of 15 min, lifting the lower and upper lids occasionally. Get medical attention.
- Skin exposure If an aldehyde or a solution containing an aldehyde contacts the skin, gently wash the contaminated skin with soap and water. Get medical attention if irritation persists.
- Inhalation: If aldehyde vapors are inhaled, move the victim at once to fresh air. If respiration has stopped, begin cardiopulmonary resuscitation. Administer oxygen if possible. Get medical attention.
- Ingestion: If an aldehyde or a solution containing an aldehyde is ingested, give the victim several glum of water to drink, dim induce vomiting by having the victim touch the back of his throat with his finger or by giving syrup of ipecac as directed on the package. Do not force an unconscious or convulsing person to drink liquids or to vomit. Get medical attention. Keep the victim warm and quiet until medical help arrives.
- Chronic exposure: Evaluate chronic exposure to aldehydes that results in dermatitis and conjunctivitis or chronic intoxication (e.g., symptoms similar to those of chronic alcoholism such as weight loss, anemia, delirium, apparent loss of intelligence, and psychological disturbances).
- Rescue: Remove an incapacitated worker from further exposure and implement appropriate emergency procedures (e.g., those listed on the Material Safety Data Sheet required by OSHA's Hazard Communication Standard [29 CFR 1910.1200]). Make all workers familiar with emergency procedures and the location and proper use of emergency equipment.
Product Substitution
When feasible, employers should substitute a less hazardous material for aldehydes. However, extreme care must be used when selecting substitutes. Possible adverse health effects of the substitute should be evaluated before selection.
Closed Systems and Ventilation
Engineering controls should be the principal method for minimizing skin and respiratory exposure to aldehydes, in the workplace. Achieving and maintaining reduced concentrations of airborne aldehydes depend on adequate engineering controls such as closed-system operations and ventilation systems that are properly constructed and maintained.
Closed-system operations provide the most effective means for minimizing worker exposures to aldehydes. Closed systems should be used for producing. storing, transferring, packaging, and processing aldehydes. For quality control laboratories or laboratories where production samples are prepared for analyses, exhaust ventilation systems should be designed to capture and contain vapors. Guidance for designing local exhaust ventilation systems can be found in Recommended Industrial Ventilation Guidelines [Hagopian and Bastress 1976], Industrial Ventilation A Manual of Recommended Practice [ACGIH 1986], and Fundamentals Governing the Design and Operation of Local Exhaust Systems [ANSI 1988].
Worker Isolation
The area in which aldehydes are produced or used should be restricted to workers essential to the process or operation. If feasible, these workers should be isolated from direct contact with aldehydes by the use of automated equipment operated from a closed control booth or room. This room should be maintained at greater air pressure than that surrounding the process equipment so that air flows out rather than in. When workers must enter the general work area to perform process checks, adjustment, maintenance, assembly line tasks, and related operations, they should take special precautions such as the use of personal protective equipment.
Personal Protective Equipment
The use of personal protective equipment (CPC, other protective equipment, and respiratory protection devices) is the least desirable method of controlling worker exposures to aldehydes and should not be used as the primary control method during routine operations.
CPC and other protective equipment.--To prevent repeated or prolonged skin contact and absorption, workers who may handle aldehydes should wear appropriate CPC and protective equipment such as gloves and face shields (8-in. length minimum). CPC made from butyl rubber should provide adequate protection for at least 1 hr [Schwope et al. 1985]. Note, however, that the quality of gloves may vary significantly among glove producers [Mickelsen and Hall 1987]. Product-specific chemical permeation data should therefore be obtained from the glove manufacturer. Splashproof goggles or face shields should be worn if there is any possibility that liquid aldehydes will contact the eyes. Safety showers and eye wash stations should be located close to operations that involve aldehydes.
Respiratory protection devices.-- NIOSH recognizes that respirators may be required to provide protection in certain situations such as implementation of engineering controls, certain short-duration maintenance procedures, and emergencies. NIOSH maintains that only the most protective respirators should be used for situations involving carcinogens. These respirators include
- any self-contained breathing apparatus equipped with a full facepiece and operated in a pressure-demand or other positive-pressure mode, and
- any supplied-air respirator equipped with a full facepiece and operated in a pressure-demand or other positive-pressure mode in combination with an auxiliary self-contained breathing apparatus operated in a pressure- demand or other positive-pressure mode.
Any respiratory protection program must, at a minimum , meet the requirements of 29 CFR 1910.134. Respirators should be approved by NIOSH and MSHA [NIOSH 1987a]. A complete respiratory protection program should include regular training and medical evaluation of personnel, fit testing, periodic environmental monitoring, and maintenance, inspection, and cleaning of equipment The program should be evaluated regularly. The following publications contain additional information about medical evaluations of respirator users and the selection, fit testing, use, storage, and cleaning of respiratory equipment: Guide to Industrial Respiratory Protection [NIOSH 1987b] and NIOSH Respirator Decision Logic [NIOSH 1987c].
Decontamination and Waste Disposal
The following steps should be taken in the event of an aldehyde spill. Persons not wearing protective clothing or equipment should be restricted from the areas of aldehyde contamination [Mackison et al. 1981].
- Remove all ignition sources.
- Ventilate the area of the spill or leak to disperse gas.
- If the spill is gaseous, stop the flow of gas.
- If the spill is liquid, absorb small quantities on paper towels. Allow towels to dry in a safe place such as a fume hood. Allow sufficient time for vapors to completely clear the hood ductwork, then bum the paper in a suitable location away from combustible materials. Reclaim or collect large quantities of spilled liquid in sealable containers and atomize the contents in a suitable combustion chamber. Absorb any remaining liquid in sand or inert absorbent and remove the absorbed material to a safe place.
- Do not allow aldehydes to enter a confined space such as a sewer unless the sewer is designed to preclude the formation of explosive concentrations of aldehyde vapors.
Medical Screening
Potential cancer sites in humans.--On the basis of the animal data presented in this CIB, NIOSH has determined that acetaldehyde and malonaldehyde meet the OSHA criteria for potential occupational carcinogens. However, screening recommendations depend on human evidence to identify appropriate screening sites. Currently, no definitive epidemiologic evidence suggests what cancer sites may be associated with exposure to acetaldehyde or malonaldehyde.
Medical screening may be appropriate if (1) animal data strongly suggest the site where the cancer risk may be increased in humans, or (2) epidemiologic evidence establishes the site for other substances in the same chemical class. For example, formaldehyde, which is chemically related to acetaldehyde and malonaldehyde, is a nasal carcinogen in rats and mice [NIOSH/OSHA 1980] and is associated with an increased risk of cancers of the upper respiratory tract (buccal, nasal, and pharyngeal) in exposed workers [Stayner et al. 1988; Stayner 1989; Lemen 1987]. On the basis of these considerations, it is prudent to assume that workers exposed to these aldehydes (formaldehyde, acetaldehyde, and malonaldehyde) have an increased risk of cancer in the upper respiratory tract The limited studies conducted with the nine related aldehydes indicate that their chemical reactivity and mutagenicity are similar to those of acetaldehyde, malonaldehyde, and formaldehyde.
Screening effectiveness.-- An effective medical screening test must detect the target condition before the worker would normally have sought treatment, and it must do so with sufficient accuracy to avoid producing large numbers of false-positive and false-negative results. In addition, the conditions targeted by screening should be those that will have a better clinical outcome with early detection.
Epidemiologic evidence about the effectiveness of cancer screening programs in improving clinical outcomes can be established only for common cancers such as lung, colon, or breast cancer. Most other cancers do not occur frequently enough for the effectiveness of screening to be evaluated epidemiologically. NIOSH therefore recommends screening when (1) screening tests are widely available, safe, and effective, and (2) limited evidence suggests that screening may be beneficial (at least in high-risk groups) or that state-of-the-art treatment may improve survival.
Several consensus groups have considered the potential usefulness of screening for oral cancer. The Canadian Task Force on the Periodic Health Examination [1979] recommends annual visual inspection of the mouth for males and all smokers. The National Cancer Institute [NCI 1987] and the American Cancer Society [1980] recommend periodic oral examinations for these groups. The Report of the U. S. Preventive Services Task Force [1989] cites evidence that patients whose oral cancer was discovered at an early stage have a better prognosis than those whose cancer was detected later. Because further research is necessary to confirm the value of screening, the task force did not recommend routine screening of asymptomatic persons for oral cancer by primary care clinicians. However, they noted that it "may be prudent for clinicians to perform careful examinations for cancerous lesions of the oral cavity in patients who use tobacco or excessive amounts of alcohol, as well as in those with suspicious symptoms or lesions detected through self-examination." On the basis of these recommendations NIOSH concludes that workers exposed to acetaldehyde and malonaldehyde are likely to benefit from screening.
Cancer screening recommendations.-- NIOSH recommends that workers exposed to acetaldehyde or malonaldehyde be offered medical screening (including cancer screening) along with appropriate counseling. Such counseling should include a discussion of (1) the evidence suggesting that such screening may be beneficial, and (2) the action to be taken if the screening test is positive (e.g., referral for further workup).
The cancer screening program should include the following:
- A medical history with emphasis on the ears, nose, mouth, and throat (e.g., history of dysphagia, sore throat, otalgia, recurrent otitis media, cranial nerve dysfunction, blocked eustachian tubes, nasal obstruction, chronic sinusitis, bleeding, hoarseness, neck lumps, or cough)
- Examination of the nasal passages, buccal cavity, and pharynx (including the nasopharynx) for evidence of cancer or precancerous lesions
- Examination of the cranial nerves (a neurological deficit could result from tumor encroachment in the nasopharyngeal area)
- Examination of the cervical lymph nodes (cervical adenopathy is frequently the first sign of pharyngeal carcinoma) [@@!--PLACEHOLDER:4f41f4aa41c74fec7446969a6895ae23dba1827--@@]
NIOSH is not aware of studies showing routine cytology of the nasal and oral passages to be beneficial to screened individuals, but any suspicious lesions noted on visual examination should be adequately evaluated (including biopsy and pathological examination if appropriate).
Appendix A
Physical and Chemical Properties of Acetaldehyde, Malonaldehyde, and Nine Related Aldehydes*
*Data from Brabec [1981], CRCS [1984], Fassett [1963], Hawley [1977], IARC [1979, 1985a, 1985b], Weast et al. [1988], and Windholz et al [1983]. [return to table]
**Not available. [return to table]
***Depolymerizes to formaldehyde above 60°C. [return to table]
Appendix B
Production, Exposure Potential, and Standards for Acetaldehyde, Malonaldehyde, and Nine Related Aldehydes
| Standards and recommendations | |||||
| Aldehyde | Estimated amount produced (tons)* |
Number of workers potentially exposed |
NIOSH recommendation (ppm)** |
OSHA PEL (ppm)*** |
MSHA PEL (ppm)† |
| Acetaldehyde | 281,000 | 14,054†† | Ca ††† | 100 (180 mg/m3), TWA 150 (270 mg/m3), STEL |
100(180 mg/m3), TWA 150 (270 mg/m3), STEL |
| Acrolein | 25,000 | 7,300§ | 0.1 (0.25 mg/m3), TWA 0.3 (0.8 mg/m3), STEL |
0.1 (0.25 mg/m3), TWA 0.3 (0.8 mg/m3), STEL |
0.1 (0.25 mg/m3), TWA 0.3 (0.8 mg/m3), STEL |
| Butyraldehyde | 750,000 | 5,392†† | -- | -- | -- |
| Crotonaldehyde | 5,000 | 148†† | 2 (6 mg/m3), TWA | 2 (6 mg/m3), TWA | 2 (6 mg/m3), TWA |
| Glutaraldehyde | 1.5 | 353,905†† | 0.2 (0.8 mg/m3), CL | 0.2 (0.8 mg/m3), CL | 0.2 (0.8 mg/m3), CL |
| Glyoxal | -- | 44,937†† | -- | -- | -- |
| Malonaldehyde | -- | -- | Ca | -- | -- |
| Paraformaldehyde | -- | 125,645†† | -- | -- | -- |
| Propiolaldehyde | -- | -- | -- | -- | - |
| Propionaldehyde | 50,000 | 1,557†† | -- | -- | -- |
| Valeraldehyde | -- | -- | 50 (175 mg/m3), TWA | 50 (175 mg/m3), TWA | 50 (175 mg/m3), TWA |
Appendix C
Mutagenicity and Toxicity of Nine Low-Molecular-Weight Aldehydes
| Aldehyde | Health effects | Organism | Exposure route | References |
| Acrolein | Mutagenicity: Hyperplastic and metaplastic changes of the nasal cavity |
Hamsters | Inhalation | Feron and Kruysse [1977] |
| Adenomas and neoplastic nodules | Rats | Oral administration | Lijinsky and Reuber [1987] | |
| Mutagenic. cytotoxic response | Salmonella typhimurium TA104 | In vitro | Marnett at al. [1985] | |
| Mutagenic response | Salmonella typhimurium TA100 ±S9* | In vitro | Haworth et al. [1983] | |
| Mutagenic response | Salmonella typhimurium TA100 -S9 | In vitro | Lutz et al. [1982] | |
| Inhibited activity of DNA polymerase | Regenerating rat liver DNA polymerase | In vitro | Munsch et al. [1973] | |
| Developmental toxicity: | ||||
| Fetal resorption, malformation | Rats | Intra-amniotic injection | Slott and Hales [1985] | |
| Malformation | Chick embryos | Injection of air chamber | Korhonen et al. [1983] | |
| Reduced pregnancy | Mice | Intraperitoneal. injection | Epstein et al. [1972] | |
| Maternal death, malformation, fetal death | Rabbits, yolk sacs | Intravenous injection | Claussen et al. [1980] | |
| Arrested differentiation | Rats, fetal explants | Exposure in culture | Schmid et al. [1981] | |
| Acute toxicity: | ||||
| Changes in blood pressure and heart rate | Rats | Cannulation, inhalation | Egle and Hudgins [1974] | |
| Pulmonary edema, death | Rats | Inhalation | Kutzman et al. [1985] | |
| LD50,** 0.046 g/kg | Rats | Oral administration | Smyth et al. [1951] | |
| Ciliotoxicity | Tracheal mucosa (species not specified) | In vitro | Dalhamn and Rosengren [1971] | |
| Lacrimation, irritation, 0.805 ppm/10 min or 1.220 ppm/5 min | Humans | Inhalation | Sim and Pattle [1957] | |
| RD50,*** 6.0 ppm | Rats | Inhalation | Babiuk et al. [1985] | |
| RD50, 1.0 ppm | Mice | Inhalation | ||
| LD50, 0.05 g/kg | Rats | Subcutaneous injection | Skog [1950] | |
| LD50 0.03 g/kg | Mice | Subcutaneous injection | ||
| LD50, 0.30 mg/l | Rats | Inhalation | ||
| Cardiovascular effects: | ||||
| Inhibited guanethidine blockade | Rats | Cannulated femoral vein | Green and Egle [1983] | |
| Pressor and depressor effects on blood pressure, increased heart rate | Rats | Cannulated femoral vein | Egle and Hudgins [1974] | |
| Butyraldehyde | Developmental toxicity: | |||
| Sperm meiotic anomalies | Mice | Intraperitoneal injection | Moutschen-Dahmen et al. [1976] | |
| Acute toxicity: | ||||
| RD50, 5,572 ppm | Rats | Inhalation | Babiuk et al. [1985] | |
| RD50, 1,015 ppm | Mice | Inhalation | ||
| LD50, 0.8 g/kg | Rats | Intraperitoneal injection | Brabec [1981] | |
| LD50, 10.0 g/kg | Rats | Subcutaneous infection | ||
| LD50, 5.9 g/kg | Rats | Oral administration | ||
| LC50,† 60,000 ppm | Rats | Inhalation | ||
| Mean fatal dose: | ||||
| 26.7 x 105 mg-min/m3 | Mice | Inhalation | Salem and Cullumbine, [1960] | |
| 24.1 x 105 mg-min/m3 | Rabbits | Inhalation | ||
| LD50, 10.00 g/kg | Rats | Subcutaneous injection, | Skog [1950] | |
| LD50, 6.17 g/kg | Mice | Subcutaneous injection | ||
| LD50, 174 mg/l | Rats | Inhalation | ||
| LD50, 5.89 g/kg | Rats | Oral administration | Union Carbide Corporation [1965] | |
| LD50, 3.56 ml/kg | Rabbits | Skin | ||
| Serious irritation | Rabbits | Eye | ||
| Slight irritation | Rabbits | Skin | ||
| No irritation at 230 ppm for 30 min | Humans | Inhalation | Sim and Pattle [1957] | |
| Cardiovascular effects: | ||||
| Bradycardia, arterial hypertension, tachycardia | Dogs | Direct intranodal perfusion | James and Bear [1968] | |
| Increased blood pressure | Cats | Femoral vein infusion | Eade [1959] | |
| Crotonaldehyde | Mutagenicity: | |||
| Neoplastic liver lesions | Rats | Oral administration | Chung et al. [1986] | |
| Sex-linked recessive lethal mutation | Drosophila melanogaster | Injection | Woodruff et al. [1985] | |
| Mutagenic response | Salmonella typhimurium TA 104 | In vitro | Marnett et al. [1985] | |
| Nonmutagenic response | Salmonella typhimurium TA 100 | In vitro | Cooper et al. [1987] | |
| Mutagenic response | Salmonella typhimurium TA 100 ±S9 | In vitro | Neudecker et al. [1981] | |
| Mutagenic response | Salmonella typhimurium TA 100 ±S9 | In vitro | Haworth et al. [1983] | |
| Mutagenic response | Salmonella typhimurium TA 100 ±S9 | In vitro | Lutz et al. [1982] | |
| Developmental toxicity: | ||||
| Sperm meiotic anomalies | Mice | Intraperitoneal injection | Moutschen-Dahmen et al. [1976] | |
| Mice | Oral administration | |||
| Acute toxicity: | ||||
| Mean fatal dose: | ||||
| 1.1x105 mg-min/m3 | Mice | Inhalation | Salem and Cullumbine [1960] | |
| 2.0x105 mg-min/m3 | Guinea pigs | Inhalation | ||
| 1.9x105 mg-min/m3 | Rabbits | Inhalation | ||
| LD50, 0.14 g/kg | Rats | Subcutaneous injection | Skog [1950] | |
| LD50, 0.16 g/kg | Mice | Subcutaneous injection | ||
| LD50, 4.00 mg/l | Rats | Inhalation | ||
| LD50, 0.30 g/kg | Rats | Oral administration | Smyth and Carpenter [1944] | |
| LD50, 0.03 ml/kg | Guinea pigs | Skin | ||
| RD50, 23.2 ppm | Rats | Inhalation | Babiuk et al. [1985] | |
| RD50, 3.5 ppm | Mice | Inhalation | ||
| Ciliotoxicity | Tracheal mucosa (species unspecified) | In vitro | Dalhamn and Rosengren [1971] | |
| Irritation of upper respiratory tract, lacrimation at 4.1 ppm for 10 min | Humans | Inhalation | Sim and Pattle [1957] | |
| Irritation, lacrimation | Humans | Vapor exposure | Stokinger [1953] | |
| Glutaraldehyde | Mutagenicity: | |||
| Mutagenic response | Salmonella typhimurium TA100 ±S9 | In vitro | Haworth et al. [1983] | |
| Mutagenic response | Salmonella typhimurium TA102 | In vitro | Levin et al. [1982] | |
| Nonmutagenic response | Salmonella typhimurium TA98, TA100, TA1535, TA1537, TA1538 ±S9 | In vitro | Slesinski et al. [1983] | |
| Nonmutagenic response | CHO,†† HGPRT | In vitro | ||
| No increase in SCE§ | CHO | In vitro | ||
| No increase in UDS§§ | Rat hepatocytes | In vitro | ||
| Mutagenic response | Salmonella typhimurium TA104 | In vitro | Marnett et al. [1985] | |
| Blocked mitosis | Pleurodele egg cell culture | In vitro | Sentein [1975] | |
| Acute toxicity: | ||||
| LD50, 2,390 mg/kg | Rats | Subcutaneous injection | Uemitsu et al. [1976] | |
| LD50, 1,430 mg/kg | Mice | Subcutaneous injection | ||
| Occupational dermatitis | Humans | Skin | Hansen [1983a,b] | |
| Occupational dermatitis | Humans | Skin | Nethercott and Holness [1988] | |
| Occupational dermatitis, respiratory tract irritation, headache, nausea | Humans | Skin Inhalation | Norback [1988] | |
| Cardiovascular effects: | ||||
| Increased ventilation and heart rate, decreased alveolar CO2 | Humans | Intravenous infusion | Asmussen et al. [1948] | |
| Disorganized effects, depolarization | Dogs | Ventricular intranodal perfusion | James and Bear [1968] | |
| Glyoxal | Mutagenicity: | |||
| Mutagenic response | Salmonella typhimurium TA104 | In vitro | Marnett et al. [1985] | |
| Mutagenic response | Salmonella typhimurium TA100 ±S9 | In vitro | Sasaki and Endo [1978] | |
| Mutagenic response | Salmonella typhimurium TA100 ±S9 | In vitro | Yamaguchi and Nakagawa [1983] | |
| Mutagenic response | Salmonella typhimurium TA102 | In vitro | Levin et al. [1982] | |
| Increased DNA synthesis, UDS, IDC§§§ | Rats | Stomach intubation | Furihata et al. [1985] | |
| Acute toxicity: | ||||
| LD50, 7.07 ml/kg | Rats | Oral administration | Union Carbide Corporation [1965] | |
| LD50, 10.0 ml/kg | Rabbits | Skin | ||
| Severe injury | Rabbits | Cornea | Carpenter and Smyth [1946] | |
| Contact dermatitis | Humans | Skin | Hindson and Lawlor [1982] | |
| Paraformaldehyde | Mutagenicity: | |||
| Transformation | Rat embryo cells | In vitro | Traul et al. [1981] | |
| Acute toxicity: | ||||
| Paresthesis | Humans | Root canal | Grossman and Tatoian [1978] | |
| Necrosis of crestal bone | Humans | Root canal | Stabholz and Blush [1983] | |
| Propiolaldehyde | Mutagenicity: | |||
| Mutagenic response | Salmonella typhimurium hisD3052 | In vitro | Basu and Marnett [1984] | |
| No DNA cross-links | Species not given | In vitro | Basu [1985] | |
| Propionaldehyde | Mutagenicity: | |||
| DNA breaks | CHO | In vitro | Marinari et al. [1984] | |
| Acute toxicity: | ||||
| Mean fatal dose: 7.9 x 105 mg-min/m3 | Mice | Inhalation | Salem and Cullumbine [1960] | |
| 7.4x105 mg-min/m3 | Rabbits | Inhalation | ||
| RD50, 6,789 ppm | Rats | Inhalation | Babiuk et al. [1985] | |
| RD50, 2,052 ppm | Mice | Inhalation | ||
| Mild irritation at 134 ppm for 30 min | Humans | Inhalation | Sim and Pattle [1957] | |
| LD50, 0.82 g/kg | Rats | Subcutaneous injection | Skog [1950] | |
| LD50, 0.68 g/kg | Mice | Subcutaneous injection | ||
| LD50, 62.0 mg/l | Rats | Inhalation | ||
| LD50, 800-1,600 mg/kg | Rats | Oral | Eastman Kodak [1971] | |
| LD50, 800-1,600 mg/kg | Mice | Oral | ||
| Anesthesia | Mice | Inhalation | Axelsson et al. [1953] | |
| Cardiovascular effects: | ||||
| Bradycardia, arterial hypertension, tachycardia | Dogs | Direct intranodal perfusion | James and Bear [1968] | |
| Increased heart rate and blood pressure | Rats | Inhalation | Egle [1972] | |
| Increased heart rate | Rats | Intravenous injection | Egle and Hudgins [1974] | |
| Increased blood pressure | Cats | Femoral vein infusion | Eade [1959] | |
| Valeraldehyde | Mutagenicity: | |||
| DNA breaks | CHO | In vitro | Marinari et al. [1984] | |
| Acute toxicity: | ||||
| No deaths at 2,359 mg/m3 | Mice Guinea pigs Rabbits |
Inhalation Inhalation Inhalation |
Salem and Cullumbine [1960] | |
*±S9 = With or without exogenous metabolic activation. [return to table]
**LD50 = Lethal dose for 50% of exposed animals. [return to table]
***RD50 = Concentration eliciting a 50% decrease in respiratory rate. [return to table]
†LC50 = Lethal concentration for 50% of exposed animals. [return to table]
††CHO = Chinese hamster ovary. [return to table]
†††HGPRT = Hypoxanthine guanine phosphoribosyl transferase. [return to table]
§SCE = Sister chromatid exchange. [return to table]
§§UDS = Unscheduled DNA synthesis. [return to table]
§§§ODC = Ornithine decarboxylation activity. [return to table]
*Code of Federal Regulations. See CFR in references. [return to text]
**Federal Register. See Fed. Reg. in references. [return to text]
***"'Potential occupational carcinogen' means any substance, or combination or mixture of substances, which causes an increased incidence of benign and/or malignant neoplasms, or a substantial decrease in the latency period between exposure and onset of neoplasms in humans or in one or more experimental mammalian species as the result of any oral, respiratory or dermal exposure, or any odor exposure which results in the induction of tumors at a site other than the site of administration. This definition also includes any substance which is metabolized into one or more potential occupational carcinogens by mammals" [29 CFR 1990.103]. [return to text]
Abernethy DJ, Frazelle JH, Boreiko CJ [1982]. Effects of ethanol, acetaldehyde and acetic acid in the C3H/10T1/2 Cl 8 cell transformation system [Abstract Bf-1]. Environ Mutagen 4:331.
ACGIH [1986]. Industrial ventilation: a manual of recommended practice. 19th ed. Cincinnati, OH: American Conference of Governmental Industrial Hygienists.
American Cancer Society [1980]. Guidelines for the cancer-related check-up: recommendations and rationale. Cancer 30:4-50.
Ames BN, McCann J, Yamasaki E [1975]. Methods for detecting carcinogens and mutagens with the Salmonella /mammalian-microsome mutagenicity test. Mutat Res 31:347-363.
ANSI [1988]. American national standard fundamental governing the design and operation of local exhaust systems. New York, NY: American National Standards Institute, ANSI Z129.1-1988.
Asmussen E, Hald J, Larsen V [1948]. The pharmacological action of acetaldehyde on the human organism. Acta Pharmacol 4:311-320.
Auerbach C, Moutschen-Dahmen M Moutschen J [1977]. Genetic and cytogenetical effects of formaldehyde and related compounds. Mutat Res 39:317-362.
Axelsson B, Forssman S, Goldberg L, Skog E [1953]. Potentiating effect of hypoxia on the anesthetic action of organic volatile anesthetics. Arch Int Pharmacodyn XCV(3-4):333-350.
Babiuk C, Steinhagen WH, Barrow CS [1985]. Sensory irritation response to inhaled aldehydes after formaldehyde pretreatment. Toxicol Appl Pharmacol 79:143-149.
Barilyak IR, Kozachuk SY [1983]. Embryotoxic and mutagenic activity of ethanol and acetaldehyde after intra-amniotic injection. Tsitol Genet 17(5):60-63.
Barnett BM, Munoz ER [1969]. Mutation test with glyoxal in Drosophila melanogaster males. Dros Info Serv 44:119.
Basu AK, Marnett LJ [1983]. Unequivocal demonstration that malondialdehyde is a mutagen. Carcinogenesis 4(3):331-333.
Basu AK, Marnett LJ [1984]. Molecular requirements for the mutagenicity of malondialdehyde and related acroleins. Cancer Res 44:2848-2854. Basu AK, Marnett LJ, Romano LJ [1984]. Dissociation of malondialdehyde mutagenicity in Salmonella typhimurium from its ability to induce interstrand DNA cross-links. Mutat Res 129:39-46.
Basu AK [1985]. Studies on the mutagenicity of malondialdehyde and structurally related compounds. Dissertation Abstracts International 54(12):3798-B.
Baxter WF Jr. [1979]. Crotonaldehyde. In: Grayson M Eckroth D, Bushey GJ, Eastman CL, Klingsberg A, van Nes L, eds. Kirk-Othmer encyclopedia of chemical technology. 3rd ed. Vol. 7. New York, NY: John Wiley & Sons, Inc., pp. 207-218.
Bird RP, Draper HH [1980]. Effect of malonaldehyde and acetaldehyde on cultured mammalian cells: growth, morphology, and synthesis of macromolecules. J Toxicol Environ Health 6:811-823.
Bird RP, Draper HH, Basrur PK [1982]. Effect of malonaldehyde and acetaldehyde on cultured mammalian cells. Production of micronuclei and chromosomal aberrations. Mutat Res 101:237-246.
Bittersohl G [1975]. Epidemiological research on cancer risk by aldol and aliphatic aldehydes. Environ Quality Safety 4:235-238.
Brabec MJ [1981]. Aldehydes and acetals. Chapter 37. In: Clayton OD, Clayton FE, eds. Patty's industrial hygiene and toxicology. 3rd rev. ed. Vol. IIA, Toxicology. New York, NY: John Wiley& Sons, Inc., pp. 2629-2669.
Brambilla O, Sciabà L, Faggin P, Maura A, Marinari UM, Ferro M, Esterbauer H [1986]. Cytotoxicity, DNA fragmentation and sister-chromatid exchange in Chinese hamster ovary cells exposed to the lipid peroxidation product 4-hydroxynonenal and homologous aldehydes. Mutat Res 171:169-176.
Brooks BR, Klamerth OL [1968]. Interaction of DNA with bifunctional aldehydes. Eur J Biochem 5:178-182.
Canadian Task Force on the Periodic Health Examination [1979]. The periodic health examination. Can Med Assoc J 121:1-45.
Carpenter CP, Smyth HF Jr. [1946]. Chemical burns of the rabbit cornea. Am J Ophthalmol 29:1363-1372.
29 CFR 1910.134. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1910.1048. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1910.1200. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1915.98. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1917.28. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1918.90. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1926.59. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
29 CFR 1990. Code of Federal regulations. Washington, DC: U.S. Government Printing Office, Office of the Federal Register.
Chio KS, Tappel AL [1969]. Inactivation of ribonuclease and other enzymes by peroxidizing lipids and by malonaldehyde. Biochemistry 8(7):2827-2832.
Chopra VL [1966]. Lethal and mutagenic effects of glyoxal. Microbial Genet Bull 25(4):25.
Chung F-L, Tanaka T, Hecht SS [1986]. Induction of liver tumors in F344 rats by crotonaldehyde. Cancer Res 46:1285-1289.
Claussen U, Hellmann W, Pache G [1980]. The embryotoxicity of the cyclophosphamide metabolite acrolein in rabbits tested in vivo by i.v. injection and by the yolk-sac method. Drug Res 30 (II)(12):2080-2083.
Collins MA, Bigdeli MG [1975]. Tetrahydroisoquinolines in vivo. I. Rat brain formation of salsolinol, a condensation product of dopamine and acetaldehyde, under certain conditions during ethanol intoxication. Life Sci 16(4):585-602.
Cooper KO, Witz G, Witmer CM [1987]. Mutagenicity and toxicity studies of several a, ß-unsaturated aldehydes in the Salmonella typhimurium mutagenicity assay. Environ Mutagen 9:289-295.
CRCS [1984]. Information review: glyoxal. IR-424. Rockville, MD: CRCS, Inc. EPA contract no. 68-01-6650 for TSCA Interagency Testing Committee.
Dalhamn T, Rosengren A [1971]. Effect of different aldehydes on tracheal mucosa. Arch Otolaryngol 93:496-500.
Draper HH, McGirr LG, Hadley M [1986]. The metabolism of malondialdehyde. Lipids 21(4):305-307.
Dreosti IE, Ballard FJ, Belling GB, Record IR, Manuel SJ, Hetzel BS [1981]. The effect of ethanol and acetaldehyde on DNA synthesis in growing cells and on fetal development in the rat. Alcoholism: Clin Exp Res 5(3):357-362.
Eade NR [1959]. Mechanism of sympathomimetic action of aldehydes. J Pharmacol Exp Ther 127:29-34.
Eastman Kodak [1971]. Toxicity data on some commercially important compounds. Rochester, NY: Eastman Kodak Company.
Egle JL Jr. [1972]. Effects of inhaled acetaldehyde and propionaldehyde on blood pressure and heart rate. Toxicol Appl Pharmacol 23:131-135.
Egle JL Jr., Hudgins PM [1974]. Dose-dependent sympathomimetic and cardioinhibitory effects of acrolein and formaldehyde in the anesthetized rat. Toxicol Appl Pharmacol. 28:358-366.
Ellingboe J [1978]. Effects of alcohol on neurochemical processes. In: Lipton MA, DiMascio, A, Killam KF, eds. Psychopharmacology: a generation of progress. New York, NY: Raven Press, pp. 1653-1664.
Epstein SS, Arnold E, Andrea J, Bass W, Bishop Y [1972]. Detection of chemical mutagens, by the dominant lethal assay in the mouse. Toxicol Appl Pharmacol 23:288-325.
Fassett DW [1963]. Aldehydes and acetals. Chapter XLIII. la: Patty FA, ed. Patty's industrial hygiene and toxicology. 2nd rev. ed. Vol. IIA, Toxicology. New York, NY: John Wiley & Sons, Inc., pp. 1959-1989.
Fechner RE [1989]. Upper respiratory tract and ear. Chapter 22. In: Anderson WAD, ed. Anderson's Pathology. 6th ed. St Louis, MO: The CV Mosby Company, pp. 1077-1094.
54 Fed. Reg. 2445 [1989]. Occupational Safety and Health Administration: air contaminants; final rule. (To be codified at 29 CFR 1910.)
Feron VJ, Kruysse A [1977]. Effects of exposure to acrolein vapor in hamsters simultaneously treated with benzo[a]pyrene or diethylnitrosamine. J Toxicol Environ Health 3:379-394.
Feron VJ, Kruysse A, Woutersen RA [1982]. Respiratory tract tumours in hamsters exposed to acetaldehyde vapour alone or simultaneously to benzo(a)pyrene or diethylnitrosamine. Eur J Cancer Clin Oncol 18(l):13-31.
Fishbein L [1979]. Potential industrial carcinogens and mutagens. Studies in environmental science. Vol. 4. New York, NY: Elsevier Scientific Publishing; Company, pp. 147-150.
Furihata C, Yoshida S, Matsushima T [1985]. Potential initiating and promoting activities of diacetyl and glyoxal in rat stomach mucosa. Jpn J Cancer Res 76(9):809-814.
Goldstein BD, Mukai FH, Hamburger SJ, McDonagh EM [1975]. Malonaldehyde, a mutagenic agent resulting from lipid peroxidation [Abstract]. Clin Res 23(5):595A.
Grant WM [1974]. Toxicology of the eye: drugs, chemicals, plants, venoms. 2nd ed. Springfield, IL: Charles C. Thomas Publisher, p. 76.
Green MA, Egle JL Jr. [1983]. The effects of acetaldehyde and acrolein on blood pressure in guanethidine-pretreated hypertensive rats. Toxicol Appl Pharmacol 69:29-36.
Grossman LI, Tatoian J [1978]. Paresthesia from N2. Oral surgery, oral medicine, oral pathology 46(5):700-701.
Hagemeyer HJ [1978]. Acetaldehyde. In. Grayson M, Eckroth D, Bushey GJ, Klingsberg A., eds. Kirk-Othmer encyclopedia of chemical technology. 3rd ed. Vol. 1. New York, NY: John Wiley& Sons, Inc., pp. 97-112.
Hagopian JH Bastress EK [1976]. Recommended industrial ventilation guidelines. Cincinnati, OH: U.S. Department of Health Education, and Welfare, Public Health Service, Center for Disease Control National Institute for Occupational Safety and Health, DHEW (NIOSH) Publication No. 76-162.
Hansen KS [1983a]. Occupational dermatoses in hospital cleaning women. Contact Dermatitis 9:343-351.
Hansen KS [1983b]. Glutaraldehyde occupational dermatitis. Contact Dermatitis 9:81-82.
Hawley GG, ed. [1977]. The condensed chemical dictionary. 9th ed. New York, NY: Van Nostrand Reinhold Company, pp. 3, 13, 142, 239, 416, 419, 725, 908.
Haworth S, Lawlor T, Mortelmans K, Speck W, Zeiger E [1983]. Salmonella mutagenicity test results for 250 chemicals. Environ Mutagen (Suppl) 1:3-142.
He S-M Lambert B [1985]. Induction and persistence of SCE-inducing damage in human lymphocytes exposed to vinyl acetate and acetaldehyde in vitro. Mutat Res 158:201-208.
Hemminki K, Suni R [1984]. Sites of reaction of glutaraldehyde and acetaldehyde with nucleosides. Arch Toxicol 55:186-190.
Henson EV [1959]. The toxicology of some aliphatic aldehydes. J Occup Med 1(8):457-462.
Hem LG, Kurtz AN, Stanton DB [1978]. Acrolein and derivatives. In: Grayson K Eckroth D, Bushey GJ, Klingsberg A, eds. Kirk-Othmer encyclopedia of chemical technology. 3rd ed. Vol 1. New York, NY: John Wiley & Sons, Inc., pp. 277-297.
Hindson C, Lawlor F [1982]. Allergy to glyoxal in a polyvinyl resin emulsion. Contact Dermatitis 8(3):213.
Hoff A [1982]. Production, processing and degradation products of polyethylene and polypropylene processing. In: Jàrvisalo, J, Pfàffli P, Vainio H, eds. Industrial hazards of plastics and synthetic elastomers. Proceedings of the International Symposium on Occupational Hazards Related to Plastics and Synthetic Elastomers, Espoo, Finland, November 22-27,1982. New York, NY: Alan R. Liss, Inc., p. 306.
Huttel R [1941]. Malonaldehyde, Report I (translated). Ber 74B(12):1825-1829.
IARC [1979]. IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans: some monomers, plastics and synthetic elastomers, and acrolein. Vol. 19. Lyon, France: World Health Organization, International, Agency for Research on Cancer, pp. 479-492.
IARC [1982]. IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans: some industrial chemicals and dyestuffs. Vol. 29. Lyon, France: World Health Organization, International Agency for Research on Cancer, pp. 347-349.
IARC [1985a]. IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans: allyl compounds aldehydes epoxides and peroxides. Vol. 36. Lyon, France: World Health Organization, International Agency for Research on Cancer, pp. 101-132.
IARC [1985b]. IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans: allyl compounds, aldehydes, epoxides and peroxides. Vol. 36. Lyon, France: World Health Organization, International Agency for Research on Cancer, pp. 163-177.
IARC [1987]. IARC monographs on the evaluation of carcinogenic risks to humans: overall evaluations of carcinogenicity; an updating of IARC monographs. Vols. 1 to 42, Suppl. 7. Lyon, France: World Health Organization, International Agency for Research on Cancer, pp. 77-78.
James TN, Bear ES [1968]. Cardiac effects of some simple aliphatic aldehydes. J Pharmacol Exp Ther 163(2):300-308.
Jansson T [1982]. The frequency of sister chromatid exchanges in human lymphocytes treated with ethanol and acetaldehyde. Hereditas 97:301-303.
Kenney WC [1982]. Acetaldehyde adducts of phospholipids. Alcoholism: Clin Exp Res 6(3):412-416.
Kenney WC [1984]. Formation of Schiff base adduct between acetaldehyde and rat liver microsomal phosphatidylethanolamine. Alcoholism: Clin Exp Res 8(6):551-555.
Knadle S [1985]. Synergistic interaction between hydroquinone and acetaldehyde in the induction of sister chromatid exchange in human lymphocytes in vitro. Cancer Res 45:4853-4857.
Korhonen A, Hemminki K, Vainio H [1983]. Embryotoxic effects of acrolein, methacrylates, guanidines and resorcinol on three day chicken embryos. Acta Pharmacol Toxicol 52:95-99.
Korte A, Obe G [1981]. Influence of chronic ethanol uptake and acute acetaldehyde treatment on the chromosomes of bone-marrow cells and peripheral lymphocytes of chinese hamsters. Mutat Res 88:389-395.
Kutzman RS, Popenoe EA, Schmaeler M, Drew RT [1985]. Changes in rat lung structure and composition as a result of subchronic exposure to acrolein. Toxicology 34:139-151.
Lam C-W, Casanova M, Heck HD'A [1986]. Decreased extractability of DNA from proteins in the rat nasal mucosa after acetaldehyde exposure. Fund Appl Toxicol 6:541-550.
Lambert B, Chen Y, He S-M, Sten M [1985]. DNA cross-links in human leucocytes treated with vinyl acetate and acetaldehyde in vitro. Mutat Res 146:301-303.
Leidel NA, Busch KA, Lynch JR [1977]. Occupational exposure sampling strategy manual. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, DHEW (NIOSH) Publication No. 77-173, NTIS No. PB-274-792/A07.
Lemen RA [1987]. Letter of February 9, 1987, to Tom Hall, U.S. Department of Labor, regarding NIOSH additional material pertaining to the proposed revision of the regulation for occupational exposure to formaldehyde. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, Division of Standards Development and Technology Transfer.
Levin DE, Hollstein M, Christman NF, Schwiers EA, Ames BN [1982]. A new Salmonella tester strain (TA102) with AT base pairs at the site of mutation detects oxidative mutagens. Proc Natl Acad Sci USA 79:7445-7449.
Lijinsky W, Reuber MD [1987]. Chronic carcinogenesis studies of acrolein. and related compounds. Toxicol Ind Health 3(3):337-345.
Lubin M, Westerfeld WW [1945]. The metabolism of acetaldehyde. J Biol Chem 161:503-512.
Lutz WK [1979]. In vivo covalent binding of organic chemicals to DNA as a quantitative indicator in the process of chemical carcinogenesis. Mutat Res 65:289-356.
Lutz D, Eder E, Neudecker T, Henschler D [1982]. Structure-mutagenicity relationship in à, -saturated carbonylic compounds and their corresponding allylic alcohols. Mutat Res 93:305-315.
Mackison FW, Stricoff RS, Partridge LJ Jr., eds. [1981]. NIOSH/OSHA occupational health guidelines for chemical hazards. Occupational safety and health guidelines for acetaldehyde. Cincinnati, OH: Centers for Disease Control, National Institute for Occupational Safety and Health, DHEW NOSH) Publication No. 81-123.
Majumdar APN, Haiman MJ, Zylbert BA, Billy HT, Vesenka GD, Geokas MC [1986]. Acetaldehyde inhibition of protein synthesis in isolated rat pancreatic acini. Toxicol Appl Pharmacol 83:86-94.
Marinari UM, Ferro M, Sciab... L, Finollo R, Bassi AM, Brambilla G [1984]. DNA-damaging activity of biotic and xenobiotic aldehydes in chinese hamster ovary cells. Cell Biochem Function 2:243-248.
Marnett LJ, Bienkowski MJ, Raban M, Tuttle MA [1979]. Studies of the hydrolysis of 14C-labeled tetraethoxypropane to malondialdehyde. Anal Biochem 99:458-463.
Marnett LJ, Hurd HK, Hollstein MC, Levin DE, Esterbauer H, Ames BN [1985]. Naturally occurring carbonyl compounds are mutagens in Salmonella tester strain TA104. Mutat Res 148:25-34.
Marnett LJ, Tuttle MA [1980]. Comparison of the mutagenicities of malondialdehyde and the side products formed during its chemical synthesis. Cancer Res 40:276-282.
MCA, Inc. [1952]. Chemical safety data sheet SD-43: properties and essential information for safe handling and use of acetaldehyde. Washington, DC: Manufacturing Chemists' Association, Inc.
McLaughlin RS [1946]. Chemical burns of the human cornea. Am J Ophthalmol 29(11):1355-1362.
Mickelsen RL, Hall RC [1987]. A breakthrough time comparison of nitrile and neoprene glove materials produced by different glove manufacturers. Am Ind Hyg Assoc J 48(11):941-947.
Moutschen-Dahmen J, Moutschen-Dahmen M, Houbrechts N, Colizzi A [1976]. Cytotoxicity and mutagenicity of two aldehydes: crotonaldehyde and butyraldehyde in the mouse. Bull Soc R Sci Liege 45(1,2):58-72.
MSHA [1989]. Air quality, chemical substances, and respiratory protection standards. Arlington, VA: U.S. Department of Labor, Mine Safety and Health Administration, Office of Standards, Report No. RIN 1219-AA48.
Mukai FH, Goldstein BD [1976]. Mutagenicity of malonaldehyde, a decomposition product of peroxidized polyunsaturated fatty acids. Science 191:868-869.
Munsch N, de Recondo A-M, Frayssinet C [1973]. Effects of acrolein on DNA synthesis in vitro. FEBS Lett 30(3):286-290.
NCI [1987]. Working guidelines for early cancer detection: rationale and supporting evidence to decrease mortality. Bethesda, MD: National Cancer Institute.
Nethercott JR, Holness DL [1988]. Contact dermatitis in funeral service workers. Contact Dermatitis 18:263-267.
Neudecker T, Lutz D, Eder E, Henschler D [1981]. Crotonaldehyde is mutagenic in a modified Salmonella typhimurium mutagenicity testing system. Mutat Res 91:27-31.
NIOSH [1974]. National occupational hazard survey (NOHS) data base, 1972-74. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, Division of Surveillance, Hazard Evaluations, and Field Studies, Surveillance Branch, Hazard Section. Unpublished data base.
NIOSH [1976a]. Criteria for a recommended standard: occupational exposure to formaldehyde. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, DHEW NOSH) Publication No. 77-126.
NIOSH [1976b]. Health hazard evaluation determination report: Rock Hill Printing and Finishing Company, Rock Hill, SC. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. 75-89-344.
NIOSH [1977]. Hazard evaluation and technical assistance report: Xomed Company, Cincinnati, OH. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. TA-77-11, NTIS No. PB82-216359.
NIOSH [1980a]. Health hazard evaluation determination report: Continental Plastic Containers, Springdale, OH. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. HE 80-2-727.
NIOSH [1980b]. Industrial hygiene characterization of urea formaldehyde and polyurethane foam insulation. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NTIS Report No. PB82-153347.
NIOSH [1980c]. Industrial hygiene survey report on urea formaldehyde foam insulation manufacturing at C.P. Chemical Company, Inc., 25 Home Street, White Plains, NY. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NTIS Report No. PB81-241812.
NIOSH [1981]. Health hazard evaluation report: Lab-Crest Scientific Glass Company, Subsidiary of Fischer and Porter Company, Warminster, PA. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. HETA 81-098-941.
NIOSH [1982]. Health hazard evaluation report: Sandoz Colors and Chemicals, East Hanover, NJ. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. HETA 81-102-1244.
NIOSH [1983a]. National occupational exposure survey (NOES), 1981-83: estimated total and female employees, field observation data and survey data on acetaldehyde and propandiel, and the nine related aldehydes. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, Division of Surveillance, Hazard Evaluations, and Field Studies, Surveillance Branch, Hazard Section. Unpublished data base; provisional data as of 01/01/90.
NIOSH [1983b]. Health hazard evaluation report: Regional Transportation District, Denver, CO. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. HETA 82-137-1264.
NIOSH [1986a]. Registry of toxic effects of chemical substances. Vol. II. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, Division of Standards Development and Technology Transfer, p. 1322.
NIOSH [1986b]. Method Nos. 3507, 2501, and 2532. In: Eller PK ed. NIOSH manual of analytical methods. 3rd ed, Vol. 3. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 84-100.
NIOSH [1987a]. NIOSH certified equipment list as of October 1, 1987. Cincinnati OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 88-107.
NIOSH [1987b]. NIOSH guide to industrial respiratory protection. Morgantown, WV: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 87-116.
NIOSH [1987c]. NIOSH respirator decision logic. Cincinnati OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) publication No. 87-108.
NIOSH [1988a]. Testimony of the National Institute for Occupational Safety and Health on the Occupational Safety and Health Administration's proposed rule on air contaminants, 29 CFR Part 1910, Docket No. H-020. Presented at the OSHA informal public hearing, August 1, 1988. NIOSH policy statements. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health.
NIOSH [1988b]. Health hazard evaluation report: U.S. Department of the Treasury, Bureau of Engraving and Printing, Washington, DC. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. HETA 87-266-1860.
NIOSH [1989]. Method Nos. 2531, 2536, 2538, and 2539 (supplement issued 05/15/89). In: Eller PM, ed. NIOSH manual of analytical methods. 3rd ed. Vol. 1. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 84-100.
NIOSH/OSHA [1980]. Current Intelligence Bulletin 34: formaldehyde; evidence of carcinogenicity. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, U.S. Department of Labor, Occupational Safety and Health, DHHS (NIOSH) Publication No. 81-111.
NIOSH/OSHA [1981]. Acetaldehyde. In: NIOSH/OSHA occupational health guidelines for chemical hazards. Cincinnati, OH: U.S. Department of Health and Human Services, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 81-123.
Norback D [1988]. Skin and respiratory symptoms from exposure to alkaline glutaraldehyde in medical services. Scand J Work Environ Health 14:366-371.
Norppa H, Tursi F, Pfaffli P, Maki-Paakkanen J, Jarventaus H [1985]. Chromosome damage induced by vinyl acetate through in vitro formation of acetaldehyde in human lymphocytes and chinese hamster ovary cells. Cancer Res 45:4816-4821.
NTP [1988]. Toxicology and carcinogenesis studies of malonaldehyde, sodium salt (3-hydroxy-2-propenal, sodium salt) in F344/N rats and B6C3F1 mice. Research Triangle Park, NC National Toxicology Program. U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, Technical Report Series No. 331.
NTP [1989]. Fifth annual report on carcinogens: summary 1989. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, National Institute of Environmental Health Sciences, National Toxicology Program, NTP 89-239.
NTP [1990]. Chemicals considered or being evaluated for short term toxicity and for long term toxicology and carcinogenesis effects. Chemical Status Report. National Toxicology Program, Research Triangle Park, NC.
NTP [1991]. Chemicals considered or being evaluated for short term toxicity and for long term toxicology and carcinogenesis effects. Chemical Status Report. National Toxicology Program, Research Triangle Park, NC.
Obe G, Beek B [1979]. Mutagenic activity of aldehydes. Drug Alcohol Depend 4:91-94.
Obe G, Natarajan AT, Meyers K Hertog AD [1979]. Induction of chromosomal aberrations in peripheral lymphocytes of human blood in vitro, and of SCEs in bone-marrow cells of mice in vivo by ethanol and its metabolite acetaldehyde. Mutat Res 68:291-294.
O'Donnell JP [1982]. The reaction of amines with carbonyls: its significance in the nonenzymatic metabolism of xenobiotics. Drug Metab Rev 13(1):123-159.
Parrilla R, Ohkawa K, Lindros KO, Zimmerman U-JP, Ashi KK, Williamson JR [1974]. Functional compartmentation of acetaldehyde oxidation in rat liver. J Biol Chem 249(15):4926-4933.
Patty FA, ed. [1981]. Aldehydes and acetals. In: Patty's industrial hygiene and toxicology. 3rd rev. ed. Vol. IIA, Toxicology. New York, NY: John Wiley & Sons Inc., pp. 1959-1989.
Protopopova TV, Skoldinov AP [1958] Acyloxy-acroleins. J Gen Chem, USSR 28:241-243.
Ristow H, Obe G [1978]. Acetaldehyde induces cross-links in DNA and causes sister-chromatid exchanges in human cells. Mutat Res 58:115-119.
Rosenkranz [1977]. Mutagenicity of halogenated alkanes and their derivatives. Environ Health Perspect 21:79-84.
Saladino AJ, Willey JC, Lechner JF, Grafstrom RC, LaVeck M, Harris CC [1985]. Effects of acetaldehyde, benzoyl peroxide, and hydrogen peroxide on cultured normal human bronchial epithelial cells. Cancer Res 45:2522-2526.
Salem H, Cullumbine H [1960]. Inhalation toxicities of some aldehydes. Toxicol Appl Pharmacol 2:183-187.
Sasaki Y, Endo R [1978]. Mutagenicity of aldehydes in Salmonella. Mutat Res 54:251-252.
Sax NI [1979]. General chemicals. Section 12. In: Sax NI, ed. Dangerous properties of industrial materials. 5th ed. New York, NY: Van Nostrand Reinhold Company, pp. 332, 705.
Schmid BP, Goulding E, Kitchin K, Sanyal MK [1981]. Assessment of the teratogenic potential of acrolein and cyclophosphamide in a rat embryo culture system. Toxicology 22:235-243.
Schwope AD, Costas PP, Jackson JO, Stull JO, Weitzman DJ [1985]. Guidelines for the selection of chemical protective clothing. 3rd ed. Vol I, Technical and reference manual. Cincinnati, OH: American Conference of Governmental Industrial Hygienists, Publication No. 0460.
Sentein P [1975]. Action of glutaraldehyde and formaldehyde on segmentation mitoses. Exp Cell Res 95:233-246.
Shamberger RJ, Andreone TL, Willis CE [1974]. Antioxidants and cancer. IV. Initiating activity of malonaldehyde as a carcinogen. J Natl Cancer Inst 53(6):1771-1773.
Shamberger RJ, Tytko SA, Willis CE [1975]. Malonaldehyde is a carcinogen [Abstract No. 3431]. Fed Proc 34(3):827.
Shamberger RJ [1980]. Evidence for the antimutagenicity and the mutagenicity of selenium. Biol Trace Element Res 2:81-87.
Silverman L, Schulte HF, First MW [1946]. Further studies on sensory response to certain industrial solvent vapors. J Ind Hyg Toxicol 28(6):262-266.
Sim VM, Pattle RE [1957]. Effect of possible smog irritants on human subjects. JAMA 163(13): 1908-1913.
Siu GK Draper HH [1982]. Metabolism of malonaldehyde in vivo and in vitro. Lipids 17(5):349-355.
Skog E [1950]. A toxicological investigation of lower aliphatic aldehydes. I. Toxicity of formaldehyde, acetaldehyde, propionaldehyde and butyraldehyde; as well as of acrolein and crotonaldehyde. Acta Pharmacol Toxicol 6:299-318.
Slesinski RS, Hengler WC, Guzzie PJ, Wagner KJ [1983]. Mutagenicity evaluation of glutaraldehyde in a battery of in vitro bacterial and mammalian test systems. Food Chem Toxicol, 21(5):621-629.
Slott VL, Hales BF [1985]. Teratogenicity and embryolethality of acrolein and structurally related compounds in rats. Teratology 32:65-72.
Smyth HP Jr., Carpenter CP [1944]. The place of the range finding test in the industrial toxicology laboratory. J Ind Hyg Toxicol 26(8):269-273.
Smyth HP Jr., Carpenter CP, Weil CS [1951]. Range-finding toxicity data: list IV. Arch Ind Hyg Occup Med 4(2):119-122.
SRI [1979]. Acetaldehyde. In: Chemical economics handbook manual of current indicators supplemental data. Menlo Park, CA: Stanford Research Institute, pp. 601.5020A-601.5020M.
SRI [1985]. Acetaldehyde. In: 1985 Directory of chemical producers, United States of America. Menlo Park, CA: Stanford Research Institute International, p. 389.
Stabholz A, Blush MS [1983]. Necrosis of the crestal bone caused by the use of Toxavit. J Endodontics 9(3):110-113.
Stayner LT, Elliott L, Blade L, Keenlyside R, Halperin W [1988]. A retrospective cohort mortality study of workers exposed to formaldehyde in the garment industry. Am J Ind Med 13:667-681.
Stayner LT [1989]. Human studies of formaldehyde exposure and cancers of the respiratory tract. In: Feron VJ, Bosland MC, eds. Proceedings of the TNO-CIVO/NYU Nose Symposium: nasal carcinogenesis in rodents; relevance to human health risk. Wageningen, Netherlands: Pudoc, pp. 98-109.
Stokinger HE [1953]. Toxicity of crotonaldehyde. Occup Health 13(4):55-56.
Szabad J, Soos I, Polgar G, Hejja G [1983]. Testing the mutagenicity of malondialdehyde and formaldehyde by the Drosophila mosaic and the sex-linked recessive lethal tests. Mutat Res 113:117-133.
Traul KA, Takayama K, Kachevsky V, Hink RJ, Wolff JS [1981]. A rapid in vitro assay for carcinogenicity of chemical substances in mammalian cells utilizing an attachment-independence endpoint. J Appl Toxicol 1(3):190-195.
TSCA [1977]. Toxic Substances Control Act inventory list: total production reported for a specific chemical Washington, DC: U.S. Environmental Protection Agency.
Tuma DJ, Sorrell NF [1985]. Covalent binding of acetaldehyde to hepatic proteins: role in alcoholic liver injury. In: Aldehyde adducts in alcoholism. Alan R. Liss, Inc., pp. 3-17.
Uemitsu N, Kawasaki H, Furuhashi T, Miyoshi K, Ohtaka T, Nomura A, et al. [1976]. Acute and subacute toxicity studies and local irritation study of glutaraldehyde. Oyo Yakuri 12(l):11-32.
Union Carbide Corporation [1965]. Summary of single exposure studies in animals: butyraldehyde and glyoxal. Industrial Medicine and Toxicology Department. Unpublished data.
Ura S, Horiuchi H, Maeda Y, Tokugawa Y [1983]. Corrugated board. Jap Pat Kokai 91:892.
USITC (U.S. International Trade Commission) [1986]. Synthetic organic chemicals: United States production and sales, 1986. Washington, DC: US. Government Printing Office, USITC Publication 2009, p. 210.
U.S. Preventive Services Task Force [1989]. Guide to clinical preventive services: an assessment for the effectiveness of 169 interventions. Baltimore, MD: Williams & Wilkins, p. 294.
Veghelyi PV, Osztovics M [1977]. The alcohol syndromes: the intrarecombigenic effect of acetaldehyde. Experientia 34(2):195-196.
Weast RC, Astle MJ, Beyer WH, eds. [1988]. CRC handbook of chemistry and physics. Boca Raton, FL: CRC Press, Inc., pp. C-42, 57, 179, 220, 288, 292, 449, 543.
Weiss G [1980]. Acetaldehyde. In: Hazardous chemicals data book. Park Ridge, NJ: Noyes Data Corporation, p. 42.
Windholz M, Budavari S, Blumetti RF, Otterbein ES, eds. [1983]. The Merck Index: an encyclopedia of chemicals, drugs, and biologicals. 10th ed. Rahway, NJ: Merck and Company, Inc., pp. 6, 19, 220, 372, 641, 647, 1008, 1127, 1416.
Woodruff RC, Mason JM, Valencia R, Zimmering S [1985]. Chemical metagenesis testing in Drosophila. V. Results of 53 coded compounds tested for the National Toxicology Program. Environ Mutagen 7:677-702.
Woutersen RA, Appelman LM, Van Garderen-Hoetmer A, Feron VJ [1986]. Inhalation toxicity of acetaldehyde in rats. III. Carcinogenicity study. Toxicology 41:213-231.
Yamaguchi T, Nakagawa K [1983]. Mutagenicity of and formation of oxygen radicals by trioses and glyoxal derivatives. Agric Biol Chem 47(11):2461-2465.
Yau TM [1979]. Mutagenicity and cytotoxicity of malonaldehyde in mammalian cells. Mech Ageing Dev 11:137-144.
Yonei S, Furui H [1981]. Lethal and mutagenic effects of malondialdehyde, a decomposition product of peroxidized lipids, on Escherichia coli with different DNA-repair capacities. Mutat Res 88:23-32.
- Page last reviewed: June 6, 2014
- Page last updated: June 6, 2014
- Content source:
- National Institute for Occupational Safety and Health Education and Information Division


 ShareCompartir
ShareCompartir