 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Prevention of Hepatitis A Through Active or Passive Immunization: Recommendations of the Advisory Committee on Immunization Practices (ACIP)Advisory Committee on Immunization Practices CHAIRMAN John F. Modlin, M.D. EXECUTIVE SECRETARY Dixie E. Snider, Jr., M.D., M.P.H. MEMBERS Richard D. Clover, M.D. David W. Fleming, M.D. Mary P. Glode, M.D. Marie R. Griffin, M.D., M.P.H. Fernando A. Guerra, M.D., M.P.H. Charles M. Helms, M.D., Ph.D. David R. Johnson, M.D., M.P.H. Chinh T. Le, M.D. Paul A. Offit, M.D. Jessie L. Sherrod, M.D. Bonnie M. Word, M.D. EX OFFICIO MEMBERS Robert F. Breiman, M.D. William Egan, Ph.D. Geoffrey S. Evans, M.D. T. Randolph Graydon Regina Rabinovich, M.D. Kristin Lee Nichol, M.D., M.P.H. David H. Trump, M.D., M.P.H. LIAISON REPRESENTATIVES American Academy of Family Physicians American Academy of Pediatrics American Association of Health Plans American College of Obstetricians and Gynecologists American College of Physicians American Hospital Association American Medical Association Association of Teachers of Preventive Medicine Biotechnology Industry Organization Canadian National Advisory Committee on Immunization Hospital Infection Control Practices Advisory Committee Infectious Diseases Society of America National Immunization Council and Child Health Program, Mexico National Medical Association National Vaccine Advisory Committee Pharmaceutical Research and Manufacturers of America Members of the Hepatitis A Working Group Mary P. Glode, M.D., Chairman Natalie Smith, M.D., M.P.H. Robert W. England, Jr., M.D., M.P.H. Francisco M. Averhoff, M.D., M.P.H. Beth P. Bell, M.D., M.P.H. Martin I. Meltzer, Ph.D.
The following CDC staff members prepared this report: Beth P. Bell, M.D., M.P.H.
Prevention of Hepatitis A Through Active or Passive Immunization: Recommendations of the Advisory Committee on Immunization Practices (ACIP) Summary Routine vaccination of children is the most effective way to reduce hepatitis A incidence nationwide over time. Since licensure of hepatitis A vaccine in 1995, this strategy has been implemented incrementally, starting with the recommendation of the Advisory Committee on Immunization Practices (ACIP) in 1996 to vaccinate children living in communities with the highest rates of infection and disease. These updated recommendations represent the next phase of this hepatitis A immunization strategy. Vaccination of children living in states and communities with consistently elevated rates of hepatitis A will provide protection from disease and is expected to reduce the overall incidence of hepatitis A. This report updates the ACIP's 1996 recommendations on the prevention of hepatitis A through immunization (MMWR 1996;45:[No. RR-15]) and includes a) new data about the epidemiology of hepatitis A; b) recent findings about the effectiveness of community-based hepatitis A vaccination programs; and c) recommendations for the routine vaccination of children in states, counties, and communities with rates that are twice the 1987-1997 national average or greater (i.e., greater than or equal to 20 cases per 100,000 population) and consideration of routine vaccination of children in states, counties, and communities with rates exceeding the 1987-1997 national average (i.e., greater than or equal to 10 but less than 20 cases per 100,000 population). Unchanged in this report are previous recommendations regarding the vaccination of persons in groups at increased risk for hepatitis A or its adverse consequences and recommendations regarding the use of immune globulin for protection against hepatitis A. INTRODUCTION Hepatitis A continues to be one of the most frequently reported vaccine-preventable diseases in the United States, despite the licensure of hepatitis A vaccine in 1995 (1). Widespread vaccination of appropriate susceptible populations would substantially lower disease incidence and potentially eliminate indigenous transmission of hepatitis A virus (HAV) infection. The Advisory Committee on Immunization Practices (ACIP) 1996 recommendations on the prevention of hepatitis A through immunization focused primarily on vaccinating persons in groups shown to be at high risk for infection (e.g., travelers to countries with high or intermediate disease endemicity, men who have sex with men, injecting-drug users, persons with clotting-factor disorders), persons with chronic liver disease because they are at increased risk for acute liver failure from hepatitis A, and children living in communities with high rates of disease (2). However, a review of available national epidemiologic data and results from community-based hepatitis A vaccination programs indicate that continued implementation of these recommendations would not result in vaccination of most populations with consistently elevated rates of disease and therefore would have a limited impact on the overall incidence of disease in the United States. To achieve a sustained reduction in hepatitis A rates, a shift is needed from the present immunization strategy to one that achieves widespread routine vaccination of children to prevent infection in these age groups and eventually among older persons. Primary Changes in the Statement This statement includes the following information that was not included in the 1996 statement:
Unchanged in this report are previous recommendations regarding the vaccination of persons in groups at increased risk for hepatitis A or its adverse consequences and recommendations regarding the use of immune globulin (IG) for protection against hepatitis A. FEATURES OF HEPATITIS A Clinical Illness HAV, a 27-nm RNA agent classified as a picornavirus, can produce either asymptomatic or symptomatic infection in humans after an average incubation period of 28 days (range, 15-50 days) (3). The illness caused by HAV infection typically has an abrupt onset of symptoms that can include fever, malaise, anorexia, nausea, abdominal discomfort, dark urine, and jaundice. The likelihood of having symptoms with HAV infection is related to the person's age. In children less than 6 years of age, most (70%) infections are asymptomatic; if illness does occur, it is not usually accompanied by jaundice (4). Among older children and adults, infection is usually symptomatic, with jaundice occurring in greater than 70% of patients (5). Signs and symptoms usually last less than 2 months, although 10%-15% of symptomatic persons have prolonged or relapsing disease lasting up to 6 months (6). In infected persons, HAV replicates in the liver, is excreted in bile, and is shed in the stool. Peak infectivity of infected persons occurs during the 2-week period before onset of jaundice or elevation of liver enzymes, when the concentration of virus in stool is highest (7,8). The concentration of virus in stool declines after jaundice appears (7,8). Children and infants can shed HAV for longer periods than adults, up to several months after the onset of clinical illness (9). Chronic shedding of HAV in feces does not occur; however, shedding can occur in persons who have relapsing illness (10). Viremia occurs soon after infection and persists through the period of liver enzyme elevation (11,12). Diagnosis Hepatitis A cannot be differentiated from other types of viral hepatitis on the basis of clinical or epidemiologic features alone. Serologic testing to detect immunoglobulin M (IgM) antibody to the capsid proteins of HAV (IgM anti-HAV) is required to confirm a diagnosis of acute HAV infection. In most persons, IgM anti-HAV becomes detectable 5-10 days before the onset of symptoms and can persist for up to 6 months after infection (12,13). Immunoglobulin G (IgG) anti-HAV, which appears early in the course of infection, remains detectable for the person's lifetime and confers lifelong protection against the disease (14). Commercial diagnostic tests are available for the detection of IgM and total (IgM and IgG) anti-HAV in serum. HAV RNA can be detected in the blood and stool of most persons during the acute phase of infection by using nucleic acid amplification methods, and nucleic acid sequencing has been used to determine the relatedness of HAV isolates (15). However, these methods, available in only a limited number of research laboratories, generally are not used for diagnostic purposes. EPIDEMIOLOGY AND PREVENTION OF HEPATITIS A VIRUS INFECTION Transmission Routes of Transmission HAV infection is acquired primarily by the fecal-oral route by either person-to-person contact or ingestion of contaminated food or water. On rare occasions, HAV infection has been transmitted by transfusion of blood or blood products collected from donors during the viremic phase of their infection (11,16). In experimentally infected nonhuman primates, HAV has been detected in saliva during the incubation period; however, transmission by saliva has not been demonstrated (17). Depending on conditions, HAV can be stable in the environment for months (18). Heating foods at temperatures greater than 185 F (85 C) for 1 minute or disinfecting surfaces with a 1:100 dilution of sodium hypochlorite (i.e., household bleach) in tap water is necessary to inactivate HAV (19). Role of Children in Hepatitis A Virus Transmission Because most children have asymptomatic or unrecognized infections, they play an important role in HAV transmission and serve as a source of infection for others (20,21). In one study of adults without an identified source of infection, 52% of their households included a child less than 6 years old, and the presence of a young child was associated with HAV transmission within the household (20). In studies where serologic testing of the household contacts of adults without an identified source of infection was performed, 25%-40% of the contacts less than 6 years old had serologic evidence of acute HAV infection (IgM anti-HAV) (20) (CDC, unpublished data, 1994). Burden of Hepatitis A Each year in the United States, an estimated 100 persons die as a result of acute liver failure due to hepatitis A. Although the case-fatality rate for fulminant hepatitis A among persons of all ages with acute hepatitis A reported to CDC is approximately 0.3%, the rate is 1.8% among adults greater than 50 years of age; persons with chronic liver disease are at increased risk for fulminant hepatitis A (22-26). The costs associated with hepatitis A are substantial. Between 11% and 22% of persons who have hepatitis A are hospitalized (27). Adults who become ill lose an average of 27 days of work (Table 1). Health departments incur substantial costs in providing postexposure prophylaxis to an average of 11 contacts per case (Table 1). Average costs (direct and indirect) of hepatitis A range from $1,817 to $2,459 per case for adults and from $433 to $1,492 per case for children less than 18 years of age (Table 1). In a recent common-source outbreak involving 43 persons, the estimated total cost was approximately $800,000 (28). In 1989, the estimated annual direct and indirect costs of hepatitis A in the United States were more than $200 million, equivalent to more than $300 million in 1997 dollars (29) (CDC, unpublished data, 1999). Surveillance and Seroprevalence Data National Trends In the United States, cyclic increases in the incidence of hepatitis A have occurred approximately every decade; the last nationwide increase occurred in 1995 (1,30). Between epidemics, hepatitis A continues to occur at relatively high rates. In 1997, 30,021 hepatitis A cases were reported to the National Notifiable Diseases Surveillance System (NNDSS) (1). After the NNDSS data were adjusted for disease under-reporting and asymptomatic infections, the number of cases reported in 1997 represented an estimated 90,000 persons with symptomatic hepatitis A and 180,000 persons with HAV infection (CDC, unpublished data, 1998). Sources of Infection Most U.S. cases of hepatitis A result from person-to-person transmission during communitywide outbreaks (31) The most frequently reported source of infection (12%-26%) is either household or sexual contact with a person with hepatitis A (27,31). Approximately 11%-16% of reported cases occur among children or employees in day care centers or among their contacts; however, this estimate might be too high because hepatitis A cases are attributed to day care center-related contact without requiring that the contact have hepatitis A or that a case of hepatitis A be identified in the day care center. An additional 4%-6% of reported cases occur among international travelers. Children account for approximately 36% of these cases, with Mexico being the most frequent (84%) destination (CDC, unpublished data 1998). Another 2%-3% of cases are associated with recognized food or waterborne disease outbreaks (27,30,31). In addition, cyclic outbreaks have occurred among users of injecting and noninjecting drugs and among men who have sex with men (31). During outbreak years, up to 10% of nationally reported cases occur among persons reporting these behaviors (CDC unpublished data, 1999). Approximately 50% of persons with hepatitis A do not have a source identified for their infection (27,31). Variations by Age, Race/Ethnicity, and Socioeconomic Status The reported incidence of hepatitis A is highest among children 5-14 years of age (Figure 1), with approximately one-third of reported cases involving children less than 15 years of age (27). Many more children have unrecognized, asymptomatic infection and can be the source of infection for others. Hepatitis A incidence varies by race/ethnicity, with highest rates among American Indians/Alaskan Natives and lowest rates among Asians; rates among Hispanics are higher than among non-Hispanics (Figure 2). Racial/ethnic differences in rates most likely reflect differences in the risk for infection related to factors such as differences in socioeconomic levels and resultant living conditions (e.g., crowding) and more frequent contact with persons from countries where hepatitis A is endemic (e.g., Mexico and Central America). About a third of the U.S. population has serologic evidence of prior HAV infection, according to data from the Third National Health and Nutrition Examination Survey (NHANES-III) conducted during 1988-1994 (CDC, unpublished data, 1998). Anti-HAV prevalence varies directly with age: among persons 6-11 years of age, the prevalence is 9%; 20-29 years of age, 19%; 40-49 years of age, 33%; and greater than 70 years of age, 75%. Age-adjusted anti-HAV prevalence is highest among Mexican-Americans (70%), compared with non-Hispanic blacks (39%) and non-Hispanic whites (23%). Anti-HAV prevalence is inversely related to income and household size. Variations by Region Over the past several decades, the highest rates of hepatitis A have occurred in a limited number of states and counties in the United States (27) (Figure 3), with rates being substantially higher in the western United States than in other U.S. regions. Although yearly rates in states with the highest disease rates can fluctuate, they consistently remain above the U.S. national average (Figure 4). During 1987-1997, an average of 50% of reported hepatitis A cases each year was from states with average disease rates greater than twice the national average of approximately 10 cases per 100,000, yet the total population of these states represented approximately 22% of the U.S. population (Table 2). An additional 18% of cases were from states with average annual disease rates above the national average during this time but less than twice the national average (Table 3). Disease Patterns in Communities Communities in the United States can be considered to have high, intermediate, or low rates of hepatitis A on the basis of epidemiologic characteristics such as age-specific rates of infection and temporal patterns of disease incidence. Most cases of hepatitis A result from person-to-person transmission during communitywide outbreaks in areas with high and intermediate rates of hepatitis A (31,32). Surveillance data demonstrate that communities with high and intermediate rates are concentrated in states with consistently elevated disease rates (Figures 3 and 4). Communities With High Rates of Hepatitis A Communities with high rates of hepatitis A typically have epidemics every 5-10 years that can last for several years. The peak incidence during these epidemics is generally greater than 700 cases per 100,000 population, and few cases occur among persons aged greater than 15 years. Seroprevalence data indicate that 30%-40% of children in these communities acquire infection before 5 years of age and almost all persons become infected before reaching young adulthood (33-35). These communities often are relatively well defined, either geographically or culturally, and include American Indian, Alaskan Native, and selected Hispanic, migrant, and religious communities (33-41). Widespread postexposure prophylaxis with IG has been used in efforts to control outbreaks in these communities but with little success (33). Since hepatitis A vaccine has become available, routine preexposure vaccination of children has been shown to be feasible in these communities through a number of venues, including public health clinics, physician offices, and schools. Moreover, when relatively high (65%-80%) first-dose vaccination coverage of preschool and school-age children is achieved and routine vaccination of young children is sustained, ongoing outbreaks of hepatitis A have been effectively interrupted, a sustained reduction in disease incidence has been observed, and subsequent outbreaks have been prevented (42-45). Communities With Intermediate Rates of Hepatitis A In communities with intermediate rates of hepatitis A, most disease occurs over a wider range of ages (i.e., children, adolescents, and young adults) than in communities with high rates of hepatitis A. Communities with intermediate rates often are large and include metropolitan areas and counties (46-48). Epidemics often occur at regular intervals and persist for several years. However, some communities with intermediate rates of hepatitis A do not have periodic epidemics but instead have sustained elevated rates of disease for many years. Overall disease rates during epidemic periods typically range from 50 to 200 cases per 100,000 population per year; however, cases are often concentrated in specific census tracts or neighborhoods where disease rates can be as high as those in communities with high rates of hepatitis A. During epidemic periods, hepatitis A rates generally increase among all age groups, indicating widespread disease within the community (31). During some communitywide outbreaks, the number of cases might increase among injecting-drug users, among men who have sex with men, or among children and employees in day care centers (4,31,48-52). Although persons with these exposures might be at increased risk for infection, they generally do not account for the majority of cases in a community, and the specific risk groups vary from community to community and outbreak to outbreak (4,50-52). Children with asymptomatic HAV infection can be a substantial source of infection for older persons (20) (CDC unpublished data, 1994). The feasibility and effectiveness of hepatitis A vaccination to control outbreaks in areas with intermediate rates of hepatitis A have been variable. In vaccination programs targeting children, generally first-dose coverage of preschool and school-age children has been low (20-45%) (53,54), and the impact of vaccination has been modest and often limited to reducing reported disease rates in the targeted age groups, which might not represent the majority of cases (54). In addition, once the outbreak has subsided, routine vaccination usually has not been sustained to prevent future outbreaks. In communities where outbreaks occurred among adults in particular risk groups (i.e., men who have sex with men or injecting-drug users), vaccination programs have been difficult to implement. Identified barriers have included the cost of the program and problems accessing the at-risk population (50,55) (CDC, unpublished data, 1999). Communities With Low Rates of Hepatitis A In communities with low rates of hepatitis A, most cases are reported among school-age children, adolescents, and young adults; rates reflect little year-to-year variation, and communitywide outbreaks are unusual (31). Although the most frequently reported source of infection is close contact with a person who has hepatitis A, cases attributed to international travel might account for 10%-12% of reported cases because of fewer sources of transmission within the community (31). Approximately half of reported cases do not have a recognized source. Groups at Increased Risk for Hepatitis A or Severe Outcomes Travelers Persons from developed countries who travel to developing countries are at substantial risk for acquiring hepatitis A (56). Such persons include tourists, military personnel, missionaries, and others who work or study abroad in countries that have high or intermediate endemicity of hepatitis A (Figure 5). Data from prospective studies indicate that the risk among travelers who do not receive preexposure prophylaxis with IG is 3/1,000-5/1,000 per month of stay; among some travelers, the risk is higher (57). The risk varies according to region visited and the length of stay and is increased even among travelers who report that they observe measures to protect themselves against enteric infection or stay only in urban areas, in luxury hotels, or in both (CDC, unpublished data, 1986). In the United States, children account for approximately one third of reported travel-related cases (CDC, unpublished data, 1998). Men Who Have Sex with Men Hepatitis A outbreaks among men who have sex with men have been reported frequently. Cyclic outbreaks have occurred in urban areas in the United States, Canada, Europe, and Australia (52,55,58-60). In serologic surveys, anti-HAV-positive persons reported more frequent oral-anal contact, longer duration of homosexual activity, and a larger number of sexual partners than persons without serologic evidence of HAV infection (61-64). Users of Injecting and Noninjecting Drugs Outbreaks have been reported among users of injecting and noninjecting drugs in the United States and in Europe (48-50,65). In the late 1980s, 10%-19% of reported hepatitis A cases occurred among persons who reported a history of injecting-drug use. More recently, outbreaks involving users of injected and noninjected methamphetamine have been reported in many communities in the midwestern and western United States, accounting for up to 30% of reported cases in these areas (50,66). Cross-sectional serologic surveys have demonstrated that injecting-drug users have higher anti-HAV seropositivity than the general U.S. population (63) (CDC, unpublished data, 1998). Transmission among injecting-drug users likely occurs through percutaneous and fecal-oral routes (e.g., sharing needles, sharing contaminated "works," and having household or other close personal contact) (67). Persons Who Have Clotting-Factor Disorders During 1992-1993, several outbreaks of hepatitis A were reported in Europe among persons with clotting-factor disorders who had been administered solvent-detergent-treated factor VIII concentrates that presumably had been contaminated from plasma donors incubating hepatitis A (68). In the United States, data from one serologic study suggested that hemophilic patients might be at increased risk for HAV infection (69). During 1995-1996, several patients who had clotting-factor disorders developed hepatitis A after having been administered solvent-detergent-treated factor VIII and factor IX concentrates (16). Persons Working with Nonhuman Primates Outbreaks of hepatitis A have been reported among persons working with non-human primates that are susceptible to HAV infection, including several Old World and New World species (70,71). Primates that were infected were those that had been born in the wild, not those that had been born and raised in captivity. Persons With Chronic Liver Disease Although not at increased risk for HAV infection, persons who have chronic liver disease are at increased risk for fulminant hepatitis A (23,25,26). Death certificate data indicate a higher prevalence of chronic liver disease among persons who died of fulminant hepatitis A compared with persons who died of other causes (22). Risk for Hepatitis A In Other Groups and Settings Food-Service Establishments/Food Handlers Recognized foodborne hepatitis A outbreaks are relatively uncommon in the United States. Nevertheless, when such outbreaks occur, intensive public health efforts are required for their control. These outbreaks are usually associated with contamination of food during preparation by an HAV-infected food handler (72). However, outbreaks associated with food (e.g., shellfish, raw produce) that has been contaminated before reaching the food-service establishment have been recognized increasingly in recent years (15,73-75) (CDC, unpublished data, 1999). Although persons who work as food handlers have a critical role in common-source foodborne HAV transmission, they are not at increased risk for hepatitis A because of their occupation. In a study of hepatitis A cases in Washington State during 1987-1988, rates of hepatitis A among food handlers were found to be similar to rates among the general population in the state (Trueman Sharp, University of Washington, unpublished data, 1989). Day Care Centers Outbreaks among children attending day care centers and persons employed at these centers have been recognized since the 1970s (4,51,76). Because infection among children is usually mild or asymptomatic, outbreaks often are recognized only when adult contacts (usually parents) become ill (4). Poor hygiene among children who wear diapers and the handling and changing of diapers by staff contribute to the spread of HAV infection; outbreaks rarely occur in day care centers in which care is provided only to children who are toilet trained. Despite the occurrence of outbreaks when HAV is introduced into day care centers, the results of serologic surveys do not indicate a substantially increased prevalence of HAV infection among staff at day care centers compared with the prevalence among control populations (77,78). Furthermore, NHANES-III data did not indicate an increased prevalence of HAV infection among children and adolescents who previously attended day care centers (CDC, unpublished data, 1995). Although day care centers can be the source of outbreaks of hepatitis A within some communities, disease within day care centers more commonly reflects extended transmission in the community. Health-Care Institutions Nosocomial HAV transmission is rare. Outbreaks have occasionally been observed in neonatal intensive-care units because of infants acquiring infection from transfused blood and subsequently transmitting hepatitis A to other infants and staff (9,79,80). Outbreaks of hepatitis A caused by transmission from adult patients to health-care workers are usually associated with fecal incontinence, although most hospitalized patients who have hepatitis A are admitted after onset of jaundice when they are beyond the point of peak infectivity (81,82). Data from serologic surveys of many types of health-care workers have not indicated an increased prevalence of HAV infection in these groups compared with that in control populations (83,84). Institutions for Persons Who Have Developmental Disabilities Historically, HAV infection was highly endemic in institutions for persons with developmental disabilities (85). As fewer children have been institutionalized and conditions within institutions have improved, the incidence and prevalence of HAV infection have decreased, although sporadic outbreaks can occur in these settings (73). Schools In the United States, the occurrence of hepatitis A cases within elementary or secondary schools usually reflects disease acquisition within the community. Child-to-child disease transmission within the school setting is uncommon; thus, if multiple cases occur among children at a school, the possibility of a common source of infection should be investigated (15,73). Workers Exposed to Sewage Data from serologic studies among Scandinavian and English workers who had been exposed to sewage indicated a possible elevated risk for HAV infection; however, in these studies, the data were not controlled for other risk factors (e.g., socioeconomic status) (86,87). Recently, two serologic surveys were conducted in the United States comparing the prevalence of anti-HAV among sewage workers to that among other municipal workers. Neither survey found a substantial increase in prevalence among sewage workers, although in one study the odds ratio of 2 was at the limit of statistical significance (CDC, unpublished data, 1998). No work-related instances of HAV transmission have been reported among sewage workers in the United States. Other Settings Waterborne outbreaks of hepatitis A are infrequent in developed countries with well-maintained sanitation and water supplies. Most outbreaks are associated with sewage-contaminated or inadequately treated water (88-90). SURVEILLANCE FOR HEPATITIS A Hepatitis A is a reportable disease in all states. The goals of hepatitis A surveillance at the national, state, and local levels include a) identifying contacts of case-patients who might require postexposure prophylaxis; b) detecting outbreaks; c) determining the effectiveness of hepatitis A vaccination; d) monitoring disease incidence by identifying acute, symptomatic infections in all age groups; e) determining the epidemiologic characteristics of infected persons, including the source of infection; and f) determining missed opportunities for vaccination. Cases of hepatitis A should be reported to local or state health departments (according to specific state requirements) so that appropriate control measures can be implemented, if indicated. Cases meeting specified criteria are reported by state health departments to CDC (91). Hepatitis A surveillance must be maintained at the local level so that the various recommended immunization strategies can be implemented and their outcome at the local, state, and national levels can be assessed. Laws requiring laboratories to promptly report all IgM anti-HAV positive results are likely to improve the completeness and timeliness of reporting. RATIONALE FOR PREVENTION OF HEPATITIS A THROUGH ROUTINE ACTIVE IMMUNIZATION The overall incidence of hepatitis A has declined in the United States over the past several decades primarily as a result of better hygienic and sanitary conditions (e.g., improved water supplies, sewage disposal, and food sanitation and less crowded living conditions). However, hepatitis A continues to be one of the most frequently reported vaccine preventable diseases, and the continued occurrence of extensive communitywide outbreaks indicates that hepatitis A remains a major public health problem. The availability of hepatitis A vaccine provides the opportunity to substantially lower disease incidence and potentially eliminate infection. The similarities between the epidemiology of hepatitis A and poliomyelitis indicate that a reduction in disease incidence can be achieved once persons in age groups that have the highest rates of HAV infection and who serve as a reservoir of infection are immunized (92). Producing a highly immune population reduces the incidence of hepatitis A and decreases transmission by preventing fecal shedding of HAV. The goals of hepatitis A immunization are to a) protect persons from infection; b) reduce disease incidence by preventing transmission; and c) ultimately eliminate transmission (93). Because of their high disease incidence and critical role in HAV transmission, children should be a primary focus of immunization strategies. Routine childhood vaccination would a) prevent infection in age groups that account for at least one third of cases; b) eliminate a major source of infection for other children and for some adults; and c) eventually prevent infection in all older persons as vaccinated children become adults, because immunity appears to be long-lasting. To achieve these goals, hepatitis A immunization strategies have been developed and implemented incrementally, on the basis of the characteristics of hepatitis A epidemiology and the feasibility and effectiveness of hepatitis A vaccination. Initial recommendations involved vaccination of persons in populations at increased risk for hepatitis A and of children living in communities with the highest rates of infection and disease. Vaccination of persons in groups at increased risk for HAV infection (e.g., travelers) or its adverse outcomes (e.g., persons with chronic liver disease) will provide personal protection to these persons but will have little effect on national disease rates, because most cases do not occur among persons in these groups. Routine vaccination of children living in communities with the highest rates of disease (i.e., high rate communities) has been effective in interrupting ongoing outbreaks and preventing subsequent outbreaks in these communities (see Communities with High Rates of Hepatitis A on page 9). Vaccination limited to these areas might have some impact on overall disease incidence. However, only a small proportion of nationally reported hepatitis A cases occur in these communities. To achieve a sustained reduction in national incidence of hepatitis A, more widespread routine vaccination of children is needed. Surveillance data have been used to identify states, counties, and communities that have had consistently elevated rates of hepatitis A and that contribute the majority of cases to the current national disease burden. The 11 states in which the average annual incidence of hepatitis A was greater than or equal to 20 cases per 100,000 during 1987-1997, representing 22% of the U.S. population, accounted for an average of 50% of reported cases each year (Table 2). Reducing hepatitis A incidence in these states, counties, and communities through sustained routine vaccination of children should substantially reduce national disease incidence. Assuming that a linear decline in hepatitis A cases would occur during a 30-year period in which successive cohorts of children living in areas with consistently elevated rates are vaccinated, the direct medical costs per case prevented would be expected to be of the same magnitude as those for other recently recommended vaccines (94,95) (CDC, unpublished data, 1999). Because a vaccine formulation or schedule for vaccinating children during the first 2 years of life is not currently available, routine vaccination of older children is required to achieve effective prevention and control of this disease. The feasibility of vaccinating older children and adolescents has been shown in a number of communities (53,96). Once hepatitis A vaccine can be used for infant and early childhood immunization, elimination of HAV transmission in the United States will become an attainable goal. PROPHYLAXIS AGAINST HEPATITIS A VIRUS INFECTION Immune Globulin IG is a sterile preparation of concentrated antibodies (immunoglobulins) made from pooled human plasma processed by cold ethanol fractionation (97). In the United States, only plasma that has tested negative for a) hepatitis B surface antigen (HBsAg), b) antibody to human immunodeficiency virus (HIV), and c) antibody to hepatitis C virus (HCV) is used to produce IG. In addition, the U.S. Food and Drug Administration (FDA) requires that the process used to produce IG include a viral inactivation step or that final products test negative for HCV RNA by polymerase chain reaction. No transmission of hepatitis B virus, HIV, HCV, or other viruses has been reported from IG for intramuscular administration (IGIM) (98,99). Anti-HAV titers differ between IG lots, and slightly lower titers have been observed over the past several decades, probably because of the decreasing prevalence of previous HAV infection among plasma donors (100). However, no clinical or epidemiologic evidence of decreased protection has been observed. IG provides protection against hepatitis A through passive transfer of antibody. Both IGIM and IG for intravenous administration (IGIV) contain anti-HAV, but IGIM is the product used for the prevention of HAV infection. The concentrations of anti-HAV achieved following administration of IGIM are below the level of detection of commercially available diagnostic tests (101). When used for preexposure prophylaxis, a dose of 0.02 mL/kg of IG administered intramuscularly (IM) confers protection for less than 3 months, and a dose of 0.06 mL/kg IG administered IM confers protection for less than or equal to 5 months (Table 4). When administered within 2 weeks following an exposure to HAV (0.02 mL/kg IM), IG is greater than 85% effective in preventing hepatitis A (102-104). Efficacy is greatest when IG is administered early in the incubation period; when administered later in the incubation period, IG often only attenuates the clinical expression of HAV infection (102). IGIM is available in single-use (2-mL) and multidose (10-mL) vials. Some preparations are formulated without a preservative; other preparations include thimerosal as a preservative in a concentration of 100 mg/L. When administration of IGIM is indicated for infants or pregnant women, preparations that do not contain thimerosal should be used. For administration of IGIM, an appropriate muscle mass (i.e., the deltoid or gluteal muscle) should be chosen into which a large volume can be injected by using a needle length appropriate for the person's age and size (105). If a gluteal muscle is used, the central region of the buttock should be avoided: only the upper outer quadrant should be used, and the needle should be directed anteriorly to minimize the possibility of injury to the sciatic nerve (105). Serious adverse events from IGIM are rare. Anaphylaxis has been reported after repeated administration to persons who have known immunoglobulin A (IgA) deficiency; thus, IGIM should not be administered to these persons (106). Pregnancy or lactation is not a contraindication to IG administration. IG does not interfere in general with the immune response to inactivated vaccines or to oral poliovirus vaccine or yellow fever vaccine. However, IG can interfere with the response to other live, attenuated vaccines (e.g., measles, mumps, rubella vaccine [MMR] and varicella vaccine) when administered as either individual or combination vaccines. Administration of MMR should be delayed for at least 3 months, and varicella vaccine should be delayed for at least 5 months after administration of IG for hepatitis A prophylaxis. IG should not be administered within 2 weeks after the administration of MMR or within 3 weeks after varicella vaccine unless the benefits of IG administration exceed the benefits of vaccination (105,107). If IG is administered within 2 weeks after administration of MMR or within 3 weeks after administration of varicella vaccine, the person should be revaccinated, but not sooner than 3 months after the IG administration for MMR or 5 months for varicella vaccine (105). Hepatitis A Vaccine Several inactivated and attenuated hepatitis A vaccines have been developed and evaluated in human clinical trials and in nonhuman primate models of HAV infection (108); however, only inactivated vaccines have been evaluated for efficacy in controlled clinical trials (36,109). The vaccines currently licensed in the United States are HAVRIX® (manufactured by SmithKline Beecham Biologicals) and VAQTA® (manufactured by Merck & Co., Inc.). Both are inactivated vaccines. Preparation Inactivated hepatitis A vaccine is prepared by methods similar to those used for inactivated poliovirus vaccine (110,111). Cell-culture-adapted virus is propagated in human fibroblasts, purified from cell lysates by ultrafiltration and exclusion gel chromatography or other methods, formalin inactivated, and adsorbed to an aluminum hydroxide adjuvant; 2-phenoxyethanol is used as a preservative for HAVRIX®, and VAQTA® is formulated without a preservative. For HAVRIX, the antigen content of the final aqueous preparation is determined by reactivity in a quantitative immunoassay for HAV antigen, and final vaccine potency (per dose) is expressed as enzyme-linked immunosorbent assay (ELISA) units (EL.U.). For VAQTA®, the antigen content is expressed as units (U) of HAV antigen. Vaccine Storage and Shipment Hepatitis A vaccine should be stored and shipped at temperatures ranging from 35.6 F (2 C) to 46.4 F (8 C) and should not be frozen. However, the reactogenicity and immunogenicity of HAVRIX® after storage at 98.6 F (37 C) for 1 week and the stability profile of VAQTA® when stored at this temperature for greater than 12 months do not differ from those of vaccines stored at the recommended temperature (112) (Merck & Co., Inc., unpublished data, 1996). Route of Administration, Vaccination Schedule, and Dosage The vaccine should be administered intramuscularly into the deltoid muscle. A needle length appropriate for the person's age and size should be used (105). HAVRIX® is available in two formulations, and the formulation differs according to the person's age: for persons 2-18 years of age, 720 EL.U. per dose in a two-dose schedule; and for persons greater than 18 years of age, 1,440 EL.U. per dose in a two-dose schedule (Table 5). A pediatric formulation of 360 EL. U. per dose administered in a three-dose schedule is no longer available. VAQTA® is licensed in two formulations, and the formulation differs according to the person's age: for persons 2-17 years of age, 25 U in a two-dose schedule; for persons greater than 17 years of age, 50 U per dose in a two-dose schedule (Table 6). Vaccine Performance Detection of anti-HAV after vaccination. Concentrations of antibody achieved after passive transfer by IG or active induction by vaccination are 10-100-fold lower than those produced after natural infection and are often below the detection level of commercially available diagnostic assays (101). To measure lower levels of antibody, more sensitive immunoassays have been developed that correlate more closely with neutralizing antibody assays (101). However, these assays have not been reviewed by the FDA and are not approved for any clinical indication. Anti-HAV concentrations are measured in comparison with a World Health Organization reference immunoglobulin reagent and are expressed as milli-International Units per milliliter (mIU/mL). The lower limits of detection are approximately 100 mIU/mL by unmodified, commercially available assays and 10 mIU/mL by more sensitive assays. Thus, a positive anti-HAV result by a standard assay indicates protection. However, after vaccination, persons who are anti-HAV negative by standard assays might still have protective levels of antibody. The absolute lower limit of antibody required to prevent HAV infection has not been defined. In vitro studies using cell-culture-derived virus indicate that low levels of antibody (e.g., less than 20 mIU/mL) can be neutralizing (113). Clinical studies have yielded few data from which a minimum protective antibody level can be derived because vaccine-induced levels of antibody have been high and few infections have been detected among vaccinated persons. Experimental studies in chimpanzees indicate that low levels of passively transferred antibody (less than 10 mIU/mL) obtained from immunized persons do not protect against infection but do prevent clinical hepatitis and virus shedding (114). To define a protective antibody response, clinical studies conducted with HAVRIX® have used levels greater than 20 mIU/mL (or greater than 33 mIU/mL in more recent studies) as measured with modified enzyme immunoassays, and studies conducted with VAQTA® have used levels greater than 10mIU/mL as measured with a modified radioimmunoassay (115,116). Immunogenicity in adults. Both licensed vaccines are highly immunogenic in persons aged greater than or equal to 18 years when administered according to the recommended schedules (116-118). Protective antibody levels developed in 94%-100% of adults 1 month after the first dose. After the second dose, all persons had protective levels of antibody, with high geometric mean antibody concentrations (GMCs). Few data are available regarding the timing of the appearance of neutralizing antibody. Among a sample of vaccinated persons, 54%-62% were positive for neutralizing antibody 14 days after the first dose, and 94%-100% were positive at 1 month (116) (SmithKline Beecham Biologicals, unpublished data, 1994). Immunogenicity in children and adolescents. Both vaccines are highly immunogenic when administered to children and adolescents according to a variety of schedules. From 97% to 100% of persons aged 2-18 years had protective levels of antibody 1 month after receiving the first dose, and 100% had protective levels 1 month after the second dose, with high GMCs (116-121). Immunogenicity in infants. Available data indicate that hepatitis A vaccine is immunogenic in children aged less than 2 years who do not have passively acquired maternal antibody. All such infants administered hepatitis A vaccine developed protective antibody levels, with the final GMCs varying depending on the dosage and schedule (122-125). Infants with passively acquired maternal antibody have reduced GMCs after vaccination (see Factors Associated with Reduced Immunogenicity on page 22). IgM anti-HAV after vaccination. Hepatitis A vaccination can induce IgM anti-HAV that is detectable by standard assays, particularly if the test is conducted soon after vaccination. IgM anti-HAV has been detected 2-3 weeks after administration of one dose of vaccine in 8%-20% of adults (126) (CDC, unpublished data, 1995). However, when tested 1 month after vaccination, only 1% of 311 adults had detectable IgM anti-HAV (127). Efficacy. The efficacy of HAVRIX® was evaluated in a double-blind, controlled, randomized clinical trial conducted in Thailand among approximately 40,000 children 1-16 years of age living in villages that had high rates of hepatitis A (109). After two doses of vaccine (360 EL.U. per dose) administered 1 month apart, the efficacy of vaccine in protecting against clinical hepatitis A was 94% (95% confidence interval, 79%-99%). A double-blind, placebo-controlled, randomized clinical trial using VAQTA® was conducted among approximately 1,000 children 2-16 years of age living in a New York community that had a high rate of hepatitis A. The protective efficacy against clinical hepatitis A was 100% (lower bound of the 95% confidence interval, 87%) after administration of one dose (25 U) of vaccine (36). Studies of chimpanzees indicate that hepatitis A vaccine can prevent HAV infection if administered shortly after exposure (128). Because the incubation period of hepatitis A can be 50 days, the fact that during a clinical efficacy trial, no cases of hepatitis A occurred in vaccine recipients beginning 17 days after vaccination also suggests a possible postexposure effect (36,45). In a small randomized trial, investigators found that hepatitis A vaccine was 79% efficacious in preventing IgM anti-HAV positivity after household exposure to hepatitis A when compared with no treatment (129). However, the confidence interval was extremely wide (7%-95%) and investigators did not assess the efficacy of the vaccine compared with IG (130). Results of an appropriately designed clinical trial comparing the postexposure efficacy of vaccine with that of IG are needed to determine if hepatitis A vaccine without IG could be recommended to prevent hepatitis A after exposure. Effectiveness in outbreak settings. Several studies have examined the effectiveness of hepatitis A vaccine in controlling outbreaks in communities that have high rates of hepatitis A. Administration of hepatitis A vaccine to children aged 2-16 years during a clinical trial evaluating vaccine efficacy resulted in a substantial decrease in community hepatitis A rates, and ongoing vaccination of young children has prevented expected communitywide outbreaks in subsequent years (36,45). In several Alaskan villages in which hepatitis A outbreaks were occurring, vaccination of children and adolescents and of susceptible adults with one dose of hepatitis A vaccine resulted in a rapid decrease in the number of new cases (42). In addition, in several American Indian communities experiencing outbreaks, early and rapid implementation of childhood hepatitis A vaccination programs stopped the outbreaks (43,44). Ongoing vaccination of young children has continued in these areas, and no further cases have been reported in subsequent years (CDC unpublished data, 1999). In these settings, vaccination was carried out in small, well-defined communities, and in most circumstances, an estimated 70% or more of persons in the target population were vaccinated. Hepatitis A vaccine has been used in several communities that have intermediate rates of hepatitis A and were experiencing outbreaks. In Butte County, California, hepatitis A incidence decreased concurrently with the implementation of a program in which approximately 37% of children aged 2-12 years were administered one dose of hepatitis A vaccine (53). In Memphis, Tennessee, following a targeted vaccination program in which one dose of vaccine was administered to 52% of eligible children aged 2-9 years, hepatitis A rates decreased in this target population (54). In two villages in Slovakia, a communitywide outbreak ended 2 months after approximately two thirds of school-age children were administered two doses of vaccine (131). Few data are available regarding the use of hepatitis A vaccine without IG to control outbreaks in day care centers. In one Italian study, hepatitis A vaccine was administered to children and staff of a nursery school and some of their household contacts to attempt to interrupt a 6-week long outbreak (132). Clinical cases among vaccinated children occurred up to 10 days after vaccination, but cases among unvaccinated adult household contacts continued to occur for 2 months. Further study is needed to determine if hepatitis A vaccine can be used alone during day care center outbreaks. Long-term protection. Among adults who received three doses of HAVRIX® (720 EL.U. per dose at 0-, 1-, and 6-month intervals), 100% of those persons had anti-HAV levels greater than 20 mIU/mL 8 years after the initial dose (P. Van Damme, University of Antwerp, Belgium, unpublished data, 1999). Six years after vaccination, all but one of 313 adults administered two doses of 1,440 EL. U. of HAVRIX® had anti-HAV levels greater than 20 mIU/mL (133). Protective levels of anti-HAV were still observed in 99% of 549 children evaluated 5-6 years after receiving VAQTA® (134). Estimates of antibody persistence derived from kinetic models of antibody decline indicate that protective levels of anti-HAV could be present for greater than or equal to 20 years (133,135-137). Whether other mechanisms (e.g., cellular memory) also contribute to long-term protection is unknown. As has been done for other vaccines, surveillance data and population-based studies are being conducted to monitor the long-term protective efficacy of hepatitis A vaccine and to determine the possible need for a booster dose. In the longest such follow-up reported to date, no cases of hepatitis A have been detected among children followed for 7 years after vaccination (45) (Merck & Co., Inc. unpublished data, 1999). Vaccination schedules. According to the licensed schedule, 18-year-old persons should receive the adult dosage of VAQTA® (Table 6). However, the seroconversion rate among 18-year-old persons administered 25U of VAQTA® was similar to the rate for those vaccinated with the dosage licensed for this age (i.e., 50 U) (Merck & Co., Inc., unpublished data, 1999). Results of a number of studies indicate that among adults administered hepatitis A vaccine according to a schedule that mixed the two currently licensed vaccines, the proportion who developed protective antibody levels did not differ from that of adults vaccinated according to the licensed schedules, and final GMCs were high (138,139). Although using the vaccines according to the licensed schedule is preferable, given the similar immunogenicity of both vaccines in adults and children, these data indicate that the two brands of hepatitis A vaccine can be considered interchangeable. Limited data are available regarding response to a delayed second vaccine dose. In one study, 97% of 87 persons aged greater than 18 years old who had received one dose of VAQTA® (50 U) had anti-HAV levels greater than 10 mIU/mL 18 months later. None reported a history of hepatitis A and all responded to a second dose. There was no difference in final GMCs compared with persons vaccinated according to the licensed schedule (Table 6) (Merck & Co., Inc., unpublished data, 1999). In another study, 82% of 51 persons aged 5 months to 39 years who had responded to one dose of HAVRIX® (720 EL.U. for children aged less than 18 years; 1440 EL.U. for adults) had anti-HAV levels greater than 20 mIU/mL a mean of 27 months later. None of these persons reported a history of hepatitis A. All of these persons responded to a second dose, with a large boost in GMCs (B. McMahon, Viral Hepatitis Program, Alaska Native Medical Center, Anchorage, unpublished data, 1999). In a third study, 79% of 124 persons had antibody titers greater than 20 mIU/mL a median of 35 months (range, 24-66 months) after receiving the first vaccine dose. All of these persons responded to a second dose; no difference was observed in final GMCs among these persons compared with persons vaccinated according to the recommended schedule (140). Factors associated with reduced immunogenicity. The presence of anti-HAV at the time of vaccination appears to blunt the immune response. Administration of IG concurrently with the first dose of hepatitis A vaccine did not decrease the proportion of adults who developed protective levels of antibody compared with adults who had been administered hepatitis A vaccine alone (141,142), but the GMCs of adults who received IG were substantially lower 1 month after completion of the vaccination series than the GMCs of adults who had been administered hepatitis A vaccine alone. However, their antibody levels were at least 100-fold higher than levels considered to be protective. Therefore, the reduced immunogenicity of hepatitis A vaccine that occurs with concurrent administration of IG does not appear to be clinically significant. Reduced vaccine immunogenicity also has been observed in infants who had passively acquired antibody because of prior maternal HAV infection and were administered hepatitis A vaccine according to a number of different schedules (122-124). In most studies, all infants developed protective levels of antibody, but the final GMCs were approximately one third to one tenth those of infants born to anti-HAV-negative mothers and vaccinated according to the same schedule. In some studies, administration of hepatitis A vaccine to persons with HIV infection resulted in lower seroprotection rates and antibody concentrations (143,144). In one study, 77% of HIV-infected persons had protective antibody levels after completing the vaccine series, and their final GMCs were considerably lower than those for HIV-negative persons (143). Among HIV-infected men, those who responded to hepatitis A vaccination had significantly more CD4+ T-lymphocytes at baseline (540/uL) compared with those who did not respond (280/uL) (144). Vaccination of adults with chronic liver disease of viral or nonviral etiology produced seroprotection rates similar to those observed in healthy adults (145,146). However final antibody levels were substantially lower for each group of chronic liver disease patients than for healthy adults. In one small study, none of the eight patients who had received a liver transplant responded to hepatitis A vaccination (147). Limited data indicate that age might reduce the immunogenicity of hepatitis A vaccine. In several studies, the proportion of persons aged greater than 40 years who had protective antibody levels was similar to that of persons aged less than or equal to 40 years, but final antibody levels were lower in the older age group (118,148-150). Additional factors associated with decreased immunogenicity to other vaccines (e.g., smoking, obesity) have not been evaluated for the currently licensed formulations of hepatitis A vaccine. No data are available pertaining to response rates to revaccination among persons who do not respond to the primary vaccination series. Simultaneous administration with other vaccines. Limited data from studies conducted among adults indicate that simultaneous administration of hepatitis A vaccine with diphtheria, poliovirus (oral and inactivated), tetanus, typhoid (both oral and IM), cholera, Japanese encephalitis, rabies, or yellow fever vaccines does not decrease the immune response to either vaccine or increase the frequency of reported adverse events (151-153). Studies indicate that hepatitis B vaccine can be administered simultaneously with hepatitis A vaccine without affecting either vaccine's immunogenicity or increasing the frequency of adverse events (154). Several studies are being conducted among infants and young children to evaluate whether simultaneous administration of hepatitis A vaccine with diphtheria-tetanus-pertussis (DTP), diphtheria-tetanus-acellular pertussis (DTaP), Haemophilus influenzae type b (Hib), hepatitis B, MMR, and oral and inactivated poliovirus vaccines affects the immunogenicity and reactogenicity of these vaccines. Side Effects and Adverse Events Data concerning adverse events are derived from prelicensure clinical studies worldwide, reports following licensure of HAVRIX® in Europe and Asia, other post-licensure studies, and reports to the national Vaccine Adverse Events Reporting System (VAERS) following licensure of HAVRIX® and VAQTA® in the United States. Local reactions. Approximately 50,000 persons were administered HAVRIX® in prelicensure clinical studies (155). No serious adverse events were attributed definitively to hepatitis A vaccine. Among adults, the most frequently reported side effects occurring within 3 days after the 1,440 EL.U. dose were soreness at the injection site (56%), headache (14%), and malaise (7%). In clinical studies among children, the most frequently reported side effects were soreness at the injection site (15%), feeding problems (8%), headache (4%), and injection-site induration (4%). Approximately 9,200 persons were administered VAQTA® in prelicensure clinical studies, with no serious adverse events reported among participants (156). Among adults, the most frequent side effects that occurred within 5 days following vaccination included tenderness (53%), pain (51%), and warmth (17%) at the injection site and headache (16%). Among children, the most common side effects reported were pain (19%), tenderness (17%), and warmth (9%) at the injection site. Serious adverse events. An estimated 1.3 million persons in Europe and Asia were vaccinated with HAVRIX® before the vaccine's licensure in the United States in 1995. Reports of serious adverse events, without regard to causality, received by the vaccine manufacturer included anaphylaxis, Guillain-Barre syndrome, brachial plexus neuropathy, transverse myelitis, multiple sclerosis, encephalopathy, and erythema multiforme (SmithKline Beecham Biologicals, unpublished data, 1995). Most of these events occurred among adults, and approximately one third occurred among persons receiving other vaccines concurrently. For serious adverse events for which background incidence data were known (e.g., Guillain-Barre syndrome and brachial plexus neuropathy), the rates for vaccine recipients were not higher than would be expected for an unvaccinated population (CDC, unpublished data, 1995). No serious adverse events were reported for approximately 40,000 children who were administered the 360 EL.U. dose of HAVRIX® in the protective efficacy study (109). In a post-licensure study of 11,417 children and 25,023 adults administered VAQTA®, no serious adverse events considered to be associated with administration of the vaccine occurred (157) (Merck & Co., Inc., unpublished data, 1999). From the time hepatitis A vaccine was first licensed in the United States in 1995 through December 1998, greater than 6.5 million doses were administered to the U.S. civilian population, including greater than 2.3 million pediatric doses (SmithKline Beecham Biologicals, unpublished data, 1999, and Merck & Co., Inc., unpublished data, 1999). During this 4-year period, VAERS received 247 reports of unexplained adverse events within 6 weeks after hepatitis A vaccination, including 80 among children less than 19 years old and 167 among adults (CDC, unpublished data, 1999). Approximately one third of events involved the concurrent use of other vaccines with hepatitis A vaccine. Thirteen of the events among children (0.6/100,000 vaccine doses distributed) and 85 of the events among adults (1.4/100,000 vaccine doses distributed) were considered serious. These events, without regard to causality, included neurologic, hematologic, and autoimmune syndromes. No reported serious events could be definitively attributed to hepatitis A vaccination. For serious adverse events for which incidence data are available, VAERS reporting rates were not higher than reported background rates (CDC, unpublished data, 1999). For example, published background incidence rates for Guillain-Barre syndrome have ranged from 0.5 to 2.4 cases per 100,000 person-years (158,159). The five Guillain-Barre cases among adult recipients of hepatitis A vaccine represent an estimated incidence of 0.2 cases per 100,000 person-years. In total, greater than 65 million doses of hepatitis A vaccine have been administered worldwide (personal communications, SmithKline Beecham Biologicals, 1999, and Merck & Co., Inc., 1999). Reviews of data from multiple sources for greater than 5 years regarding adverse events did not identify any serious adverse events among children or adults that could be definitively attributed to hepatitis A vaccine or an increase in serious adverse events among vaccinated persons above baseline rates (109,157,160). The safety of the vaccine will continue to be assessed through ongoing monitoring of data from VAERS and other surveillance systems. Any adverse event suspected to be associated with hepatitis A vaccination should be reported to VAERS. VAERS forms can be obtained by calling (800) 822-7967, and information on how to report adverse events can be obtained from the VAERS home page at http://www.fda.gov/cber/vaers/vaers.htm. Contraindications and Precautions Hepatitis A vaccine should not be administered to persons with a history of a severe reaction to a prior dose of hepatitis A vaccine or to a vaccine component (e.g., alum, 2-phenoxyethanol [in the case of HAVRIX®]). The safety of hepatitis A vaccination during pregnancy has not been determined; however, because hepatitis A vaccine is produced from inactivated HAV, the theoretical risk to the developing fetus is expected to be low. The risk associated with vaccination should be weighed against the risk for hepatitis A in women who might be at high risk for exposure to HAV. Because hepatitis A vaccine is inactivated, no special precautions need to be taken when vaccinating immunocompromised persons. Prevaccination Serologic Testing for Susceptibility Antibody production in response to HAV infection results in lifelong immunity to hepatitis A and, presumably, to HAV infection. Vaccination of a person who is immune because of prior infection does not increase the risk for adverse events. In populations that have expected high rates of prior HAV infection, prevaccination testing may be considered to reduce costs by not vaccinating persons who have prior immunity. Testing of children is not indicated because of their expected low prevalence of infection. For adults, the decision to test should be based on a) the expected prevalence of immunity; b) the cost of vaccination compared with the cost of serologic testing (including the cost of an additional visit); and c) the likelihood that testing will not interfere with initiating vaccination. For example, if the cost of screening (including laboratory and office visits) is one third the cost of the vaccine series, then screening potential recipients in populations where the prevalence of infection is likely to be greater than 33% should be cost-effective (161). Persons for whom prevaccination testing will likely be most cost-effective include adults who were either born in or lived for extensive periods in geographic areas that have a high endemicity of HAV infection (Figure 5); older adolescents and adults in certain population groups (i.e., American Indians, Alaskan Natives, and Hispanics); and adults in certain groups that have a high prevalence of infection (e.g., injecting drug users). In addition, the prevalence might be high enough among all older adults to warrant prevaccination testing. For example, the anti-HAV prevalence among persons greater than 40 years of age, determined by NHANES-III testing, is generally greater than 33% (regardless of race/ethnicity or income level). Thus, if the cost of screening is one third the cost of the vaccination series, prevaccination testing of any person greater than 40 years of age would likely be cost-effective. Commercially available tests for total anti-HAV should be used for prevaccination testing. Postvaccination Testing for Serologic Response Postvaccination testing is not indicated because of the high rate of vaccine response among adults and children. In addition, testing methods that have the sensitivity to detect low anti-HAV concentrations after vaccination are not approved for routine diagnostic use in the United States. RECOMMENDATIONS FOR USE OF HEPATITIS A VACCINE AND IMMUNE GLOBULIN Preexposure Protection Against Hepatitis A Virus Infection Hepatitis A vaccination provides preexposure protection from HAV infection in children and adults. Hepatitis A vaccination is recommended for persons who are at increased risk for infection and for any person wishing to obtain immunity. Children Who Should Be Routinely Vaccinated or Considered for Vaccination Children living in areas where rates of hepatitis A are at least twice the national average should be routinely vaccinated. These children include
Children living in areas where rates of hepatitis A are greater than the national average but lower than twice the national average should be considered for routine vaccination. These children include
For children living in states with rates of hepatitis A that are greater than or equal to twice the national average for 1987-1997, routine vaccination statewide is recommended. For children living in states where disease incidence is lower than twice the national average, the decision of whether to adopt a statewide or community-based (e.g., county, city, or town) vaccination strategy should include considerations such as feasibility of implementation, the extent to which areas with elevated rates cluster, and whether the strategy is likely to lower overall disease incidence in the state. Determination of age groups recommended for vaccination should take into consideration community disease patterns. In communities with high rates of hepatitis A (e.g., American Indian reservations, Alaskan Native villages), routine vaccination of children beginning at greater than or equal to 2 years of age and catch-up vaccination of preschool children should receive highest priority, as previously recommended (2) (See Communities With High Rates of Hepatitis A on page 9). In addition, to more effectively prevent epidemics of hepatitis A in these communities, vaccination of previously unvaccinated older children (e.g., up to 10-15 years of age) continues to be recommended (2). Prevaccination serologic testing is not indicated for vaccination of previously unvaccinated children in this setting. In other areas where routine childhood vaccination is recommended, possible strategies include vaccinating one or more single-age cohorts of children or adolescents (e.g., children at the age of entry into preschool, elementary school, and/or middle school), vaccination of children and adolescents in selected settings (e.g., day care) or vaccination of children and adolescents over a wide range of ages in a variety of settings, such as when they seek health care for other purposes (see Children Who Should Be Routinely Vaccinated or Considered for Vaccination on page 25). Persons at Increased Risk for Hepatitis A Virus Infection Who Should be Routinely Vaccinated Persons traveling to or working in countries that have high or intermediate endemicity of infection. All susceptible persons traveling to or working in countries that have high or intermediate HAV endemicity (Figure 5) should be vaccinated or receive IG before departure (Tables 4-6). Hepatitis A vaccination at the age-appropriate dose is preferred (Tables 5 and 6), particularly for children, adolescents, and adults who plan frequent travel or who reside for long periods in a high-risk area. IG is recommended for travelers less than 2 years of age because the vaccine is currently not licensed for use in this age group. Prevaccination testing should be considered for older travelers or for younger persons in certain population groups (see Prevaccination Serologic Testing for Susceptibility on page 24). Travelers to Canada, western Europe, Japan, Australia, or New Zealand are at no greater risk for infection than in the United States. Data are not available regarding the risk for hepatitis A for persons traveling to developed areas of the Caribbean, although vaccine or IG should be considered if travel to areas that have questionable sanitation is anticipated. Travelers who are administered vaccine can be assumed to be protected by 4 weeks after receiving the first vaccine dose and therefore should receive this dose at least 4 weeks before departure. Although according to both vaccines' licensure information, the first dose can be given at least 2 weeks before departure, available data suggest that 40%-45% of vaccinated persons might lack neutralizing antibody at 14 days after receiving the first dose (see Immunogenicity in Adults on page 19). No data are currently available regarding the risk for hepatitis A among persons vaccinated 2-4 weeks before departure. Because protection might not be complete until 4 weeks after vaccination, persons traveling to a high-risk area less than 4 weeks after the initial dose also should be administered IG (0.02 mL/kg), but at a different anatomic injection site. A second vaccine dose administered according to the recommended schedule (Tables 5 and 6) is necessary for long-term protection. Travelers who are allergic to a vaccine component or who elect not to receive vaccine should receive a single dose of IG (0.02 mL/kg), which provides effective protection against hepatitis A for up to 3 months (Table 4). Travelers whose travel period exceeds 2 months should be administered IG at 0.06 mL/kg; administration must be repeated if the travel period exceeds 5 months (Table 4). Men who have sex with men. Sexually active men who have sex with men (both adolescents and adults) should be vaccinated. Prevaccination testing is not indicated for the vaccination of adolescents and young adults in this group but might be warranted for older adults. Illegal-drug users. Vaccination is recommended for users of injecting and non-injecting illegal drugs. Prevaccination testing is not indicated for the vaccination of adolescent users of illegal drugs but might be warranted for adults. Persons who have occupational risk for infection. Persons who work with HAV-infected primates or with HAV in a research laboratory setting should be vaccinated. Studies conducted to date among U.S. workers exposed to raw sewage do not indicate a significantly increased risk for HAV infection. No other groups have been shown to be at increased risk for HAV infection because of occupational exposure. Persons who have clotting-factor disorders. Susceptible persons who are administered clotting-factor concentrates, especially solvent-detergent-treated preparations, should be administered hepatitis A vaccine. Vaccination of Persons Who Have Chronic Liver Disease Susceptible persons who have chronic liver disease should be vaccinated. Available data do not indicate a need for routine vaccination of persons with chronic hepatitis B virus or hepatitis C virus infections without evidence of chronic liver disease. Susceptible persons who either are awaiting or have received liver transplants also should be vaccinated. Hepatitis A Vaccination During Outbreaks Vaccination for outbreak-control should take into consideration the characteristics of hepatitis A epidemiology in the community and existing hepatitis A vaccination programs. Outbreaks in communities with high rates of hepatitis A. If routine vaccination programs have not achieved at least 70% vaccination coverage of preschool and school-age children, intensified vaccination efforts among preschool children and an accelerated vaccination program for school-age children should be implemented to achieve these coverage levels. The upper age for vaccination of older, previously unvaccinated children should be determined by using age-specific rates of hepatitis A (or seroprevalence data, if available) but will usually be 10-15 years of age. To prevent future outbreaks, ongoing vaccination of young children should be maintained once the outbreak has subsided (see Children Who Should Be Routinely Vaccinated or Considered for Vaccination on page 25). Outbreaks in communities with intermediate rates of hepatitis A. Routine vaccination of children is recommended for most of these communities and implementation of these programs will eventually prevent outbreaks. If routine childhood vaccination has not been implemented, this should be initiated as recommended (see Children Who Should Be Routinely Vaccinated or Considered for Vaccination on page 25) Accelerated vaccination can be considered as an additional measure to control these outbreaks. These communities often are located in large cities or counties; thus, widespread vaccination might not be feasible. Because outbreaks in these communities often involve both children and adults and might include adults in groups at increased risk for infection (e.g., illegal-drug users, men who have sex with men), local surveillance and epidemiologic data should be used to define populations (e.g., age groups or risk groups) or areas (e.g., census tracts) within the community that have the highest rates of disease. Factors to consider in deciding whether to initiate an outbreak-control vaccination program include the feasibility of rapidly vaccinating the target population of children, adolescents, or young adults, and program cost. Vaccination programs to control outbreaks occurring primarily among adults in identified risk groups (i.e., men who have sex with men, injecting-drug users) have been difficult to implement. Therefore, efforts to control ongoing hepatitis A outbreaks among adults in these groups also should involve initiating and sustaining routine vaccination of these persons to prevent future outbreaks. Because the results of vaccination programs to control hepatitis A outbreaks in communities that have intermediate rates of disease have been variable, evaluation of the effectiveness should be an essential element of programs in these settings. In all such communities, ongoing vaccination of children should be sustained to maintain high levels of immunity and prevent future epidemics. Outbreaks in communities with low rates of hepatitis A. Communitywide outbreaks are uncommon in communities with low rates of hepatitis A. If outbreaks occur, the response should be based on an examination of the epidemiologic characteristics of the outbreak. Vaccination programs to control outbreaks occurring primarily among adults in identified risk groups (i.e., men who have sex with men, injecting-drug users) have been difficult to implement. Therefore, efforts to control and prevent hepatitis A outbreaks among adults in these groups primarily should be focused on initiating and sustaining routine vaccination of these persons. If outbreaks involving children occur, implementing programs similar to those recommended for intermediate rate communities can be considered, including ongoing routine vaccination of children. Outbreaks in other settings. The frequency of outbreaks in day care centers, hospitals, institutions (e.g., institutions for the developmentally disabled, prisons), and schools is not high enough to warrant routine hepatitis A vaccination of persons specifically because they are in these settings, and few data exist regarding the role of hepatitis A vaccine in controlling outbreaks in these settings (see Risk for Hepatitis A In Other Groups and Settings on page 12 and Effectiveness in Outbreak Settings on page 20). When outbreaks are recognized in day care centers, aggressive use of IG is effective in limiting transmission to employees and families of attendees (see Postexposure Prophylaxis with Immune Globulin below). When outbreaks occur in hospitals, institutions, and schools, use of IG for persons in close contact with infected patients or students is recommended (see Postexposure Prophylaxis with Immune Globulin below). In areas where routine childhood vaccination is recommended, previously unvaccinated children receiving postexposure prophylaxis with IG could also receive hepatitis A vaccine. Persons who work as food handlers can contract hepatitis A and potentially transmit HAV to others. To decrease the frequency of evaluations of food handlers with hepatitis A and the need for postexposure prophylaxis of patrons, consideration may be given to vaccination of employees who work in areas where state and local health authorities or private employers determine that such vaccination is cost-effective. Postexposure Prophylaxis With Immune Globulin Persons who have been recently exposed to HAV and who have not previously been administered hepatitis A vaccine should be administered a single IM dose of IG (0.02 mL/kg) as soon as possible, but not greater than 2 weeks after the last exposure. Persons who have been administered one dose of hepatitis A vaccine at least 1 month before exposure to HAV do not need IG. Because hepatitis A cannot be reliably diagnosed on clinical presentation alone, serologic confirmation of HAV infection in index patients by IgM anti-HAV testing is recommended before postexposure treatment of contacts. Screening of contacts for immunity before giving IG is not recommended because screening is likely to be more costly than IG and would delay its administration. IG should be administered to previously unvaccinated persons in the following situations. If hepatitis A vaccine is recommended for a person being given IG, it can be administered simultaneously with IG at a separate anatomic injection site. The use of hepatitis A vaccine alone is not recommended for postexposure prophylaxis. Close personal contact. IG should be administered to all previously unvaccinated household and sexual contacts of persons who have serologically confirmed hepatitis A. In addition, persons who have shared illegal drugs with a person who has serologically confirmed hepatitis A should receive IG and hepatitis A vaccine. Consideration should also be given to providing IG to persons with other types of ongoing, close personal contact (e.g., regular babysitting). Day care centers. IG should be administered to all previously unvaccinated staff and attendees of day care centers or homes if a) one or more cases of hepatitis A are recognized in children or employees or b) cases are recognized in two or more households of center attendees. In centers that do not provide care to children who wear diapers, IG need be given only to classroom contacts of an index case-patient. When an outbreak occurs (i.e., hepatitis cases in three or more families), IG also should be considered for members of households that have children (center attendees) in diapers. In those communities where routine vaccination is recommended, hepatitis A vaccine can be administered at the same time as IG for children receiving postexposure prophylaxis in day care centers. Common-source exposure. If a food handler is diagnosed with hepatitis A, IG should be administered to other food handlers at the same establishment. Because common-source transmission to patrons is unlikely, IG administration to patrons is usually not recommended but can be considered if a) during the time when the food handler was likely to be infectious, the food handler both directly handled uncooked foods or foods after cooking and had diarrhea or poor hygienic practices; and b) patrons can be identified and treated within 2 weeks after the exposure. In settings where repeated exposures to HAV might have occurred (e.g., institutional cafeterias), stronger consideration of IG use might be warranted. In the event of a common-source outbreak, IG should not be administered to exposed persons after cases have begun to occur because the 2-week period during which IG is effective will have been exceeded. Schools, hospitals, and work settings. IG is not routinely indicated when a single case occurs in an elementary or secondary school, an office, or in other work settings, and the source of infection is outside the school or work setting. Similarly, when a person who has hepatitis A is admitted to a hospital, staff should not routinely be administered IG; instead, careful hygienic practices should be emphasized. IG should be administered to persons who have close contact with index patients if an epidemiologic investigation indicates HAV transmission has occurred among students in a school or among patients or between patients and staff in a hospital. FUTURE CONSIDERATIONS Implementation of these recommendations should significantly lower the incidence of hepatitis A in the United States. When this occurs, the opportunity will be present to eliminate HAV transmission (93). However, to achieve this goal, children throughout the United States will need to be vaccinated against hepatitis A. This effort would be facilitated by the availability of a vaccine formulation or schedule for use in infants or children in the second year of life and combination vaccines that include hepatitis A vaccine. In the interim, a number of issues should be addressed through clinical trials and other studies:
References
Table 1 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 1. Morbidity associated with hepatitis A, by source of data and year -- United States
* TR Eng, CDC, unpublished data, 1991; based upon interviews of 144 patients; cost data based upon a subset of 74 patients with complete cost information. Return to top. Figure 1 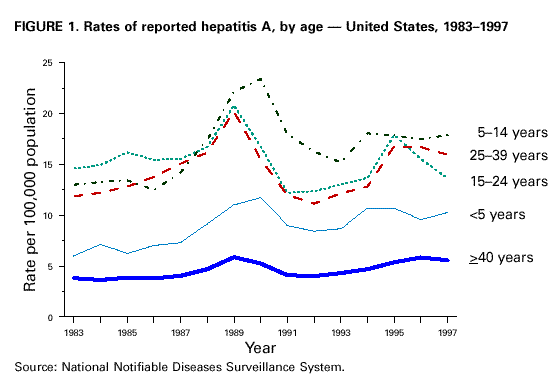 Return to top. Table 2 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 2. Burden of hepatitis A in states with average reported incidence of >=20 cases per 100,000 population -- 1987-1997*
* United States reported disease incidence during 1987-1997 was 10.8 cases per 100,000 population. Reported hepatitis A cases from these 11 states accounted for an average of 50% of reported cases each year, yet the total population of these states represents 22% of the U.S. population. Source: National Notifiable Diseases Surveillance System. Return to top. Figure 2 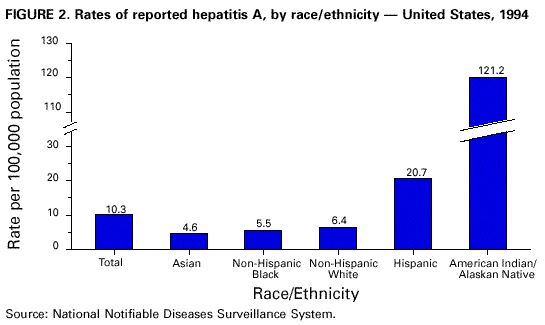 Return to top. Table 3 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 3. Burden of hepatitis A in states with average reported incidence of >=10 cases per 100,000 but <20 per 100,000 population -- 1987-1997*
* United States reported disease incidence during 1987-1997 was 10.8 cases per 100,000 population. Source: National Notifiable Diseases Surveillance System. Return to top. Figure 3 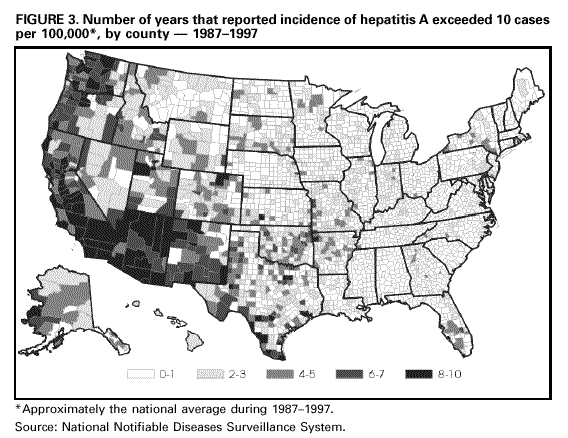 Return to top. Table 4 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 4. Recommended doses of immune globulin (IG) for hepatitis A preexposure and postexposure prophylaxis*
* Infants and pregnant women should receive a preparation that does not include thimerosal. Return to top. Figure 4 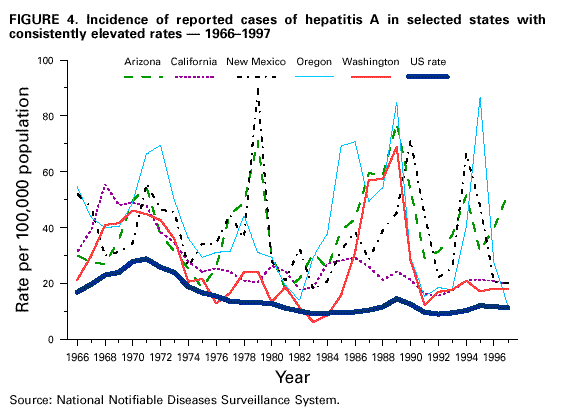 Return to top. Table 5 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 5. Recommended dosages of HAVRIX®*
* Hepatitis A vaccine, inactivated, SmithKline Beecham Biologicals. Return to top. Figure 5 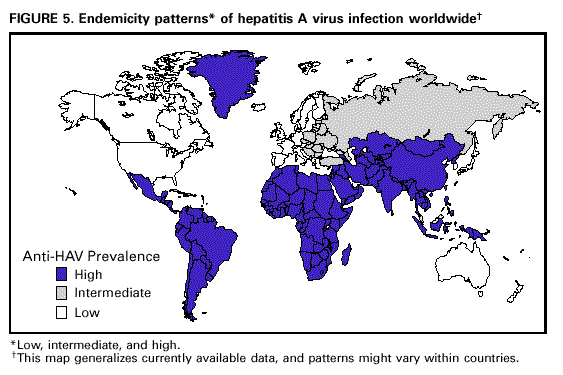 Return to top. Table 6 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 6. Recommended dosages of VAQTA®*
* Hepatitis A vaccine, inactivated, Merck ¶ Co., Inc. Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 9/30/1999 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
This page last reviewed 5/2/01
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||