Rizatriptan
Rizatriptan, sold under the brand name Maxalt among others, is a medication used for the treatment of migraine headaches.[1] It should be used as soon as the headache starts.[2] It is taken by mouth.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Maxalt, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601109 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Triptan |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 45% |
| Protein binding | 14% |
| Metabolism | by monoamine oxidase |
| Elimination half-life | 2–3 hours |
| Excretion | 82% urine; 12% faeces |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.243.719 |
| Chemical and physical data | |
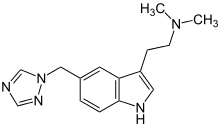
| Formula | C15H19N5 |
| Molar mass | 269.345 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Common side effects include chest pain, dizziness, dry mouth, and tingling.[1] Other side effects may include myocardial infarction, stroke, high blood pressure, serotonin syndrome, and anaphylaxis.[1] Excessive use may result in medication overuse headaches.[1] Use is not recommended during pregnancy and breastfeeding is not recommended within 24 hours after taking a dose.[2] Rizatriptan is in the triptan class and is believed to work by activating the 5-HT1 receptor.[1]
Rizatriptan was patented in 1991 and came into medical use in 1998.[3] It is available as a generic medication.[2] A dose in the United Kingdom costs the NHS about 3.10 £ as of 2019.[2] In the United States the wholesale cost of this amount is about US$0.73.[4] In 2016 it was the 232nd most prescribed medication in the United States with more than 2 million prescriptions.[5]
Medical uses
Rizatriptan is used to treat acute migraine attacks with or without aura. It does not prevent future migraine attacks.[6] Rizatriptan is also used off-label to treat cluster headaches.
Contraindications
Adverse effects
Severe:
- serotonin syndrome when used with MAOIs
- coronary artery vasospasm
- transient myocardial ischemia
- myocardial infarction
- ventricular tachycardia
- ventricular fibrillation
- hypertensive crisis
Atypical sensations:
Ear, nose, and throat:
Muscular:
Neurological:
- phonophobia
- photophobia
Respiratory:
Skin:
Miscellaneous:
Interactions
- Monoamine oxidase inhibitors
- Propranolol[7]
Mechanism of action
Rizatriptan acts as an agonist at serotonin 5-HT1B and 5-HT1D receptors.[8] Like the other triptans sumatriptan and zolmitriptan, rizatriptan induces vasoconstriction—possibly by inhibiting the release of calcitonin gene-related peptide from sensory neurons in the trigeminal nerve.[8]
Society and culture
It is typically by prescription except in Brazil.
Names
Brandnames include Bizaliv, Rizalt, and Maxalt.
References
- "Rizatriptan Benzoate Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 18 March 2019.
- British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 473. ISBN 9780857113382.
- Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 531. ISBN 9783527607495.
- "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Retrieved 3 March 2019.
- "The Top 300 of 2019". clincalc.com. Retrieved 22 December 2018.
- "Rizatriptan". MedlinePlus. U.S. National Library of Medicine.
- Millson, D; Tepper, S (2000). "Migraine pharmacotherapy with oral triptans: a rational approach to clinical management". Expert Opin Pharmacother. 1 (3): 391–404. doi:10.1517/14656566.1.3.391. PMID 11249525.
- Wellington, K; Plosker, G. L. (2002). "Rizatriptan: An update of its use in the management of migraine". Drugs. 62 (10): 1539–74. doi:10.2165/00003495-200262100-00007. PMID 12093318.