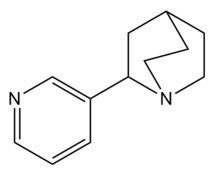
RJR-2429
RJR-2429 is a drug that acts as an agonist at neural nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes. RJR-2429 is stronger than nicotine but weaker than epibatidine in most assays, and with high affinity for both α3β4 and α4β2 subtypes, as well as the less studied α1βγδ subtype.[1][2][3]
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C12H16N2 |
| Molar mass | 188.27 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
References
- Bencherif, M.; Schmitt, J. D.; Bhatti, B. S.; Crooks, P.; Caldwell, W. S.; Lovette, M. E.; Fowler, K.; Reeves, L.; Lippiello, P. M. (1998). "The heterocyclic substituted pyridine derivative (+/-)-2-(-3-pyridinyl)-1-azabicyclo2.2.2octane (RJR-2429): A selective ligand at nicotinic acetylcholine receptors". The Journal of Pharmacology and Experimental Therapeutics. 284 (3): 886–894. PMID 9495846.
- Yokotani, K.; Okada, S.; Nakamura, K. (2002). "Characterization of functional nicotinic acetylcholine receptors involved in catecholamine release from the isolated rat adrenal gland". European Journal of Pharmacology. 446 (1–3): 83–87. doi:10.1016/s0014-2999(02)01819-8. PMID 12098588.
- Bhatti, B.; Strachan, J.; Breining, S.; Miller, C.; Tahiri, P.; Crooks, P.; Deo, N.; Day, C.; Caldwell, W. (2008). "Synthesis of 2-(Pyridin-3-yl)-1-azabicyclo[3.2.2]nonane, 2-(Pyridin-3-yl)-1-azabicyclo[2.2.2]octane, and 2-(Pyridin-3-yl)-1-azabicyclo[3.2.1]octane, a Class of Potent Nicotinic Acetylcholine Receptor-Ligands". The Journal of Organic Chemistry. 73 (9): 3497–507. doi:10.1021/jo800028q. PMID 18363376.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.