Quifenadine
Quifenadine (Russian: хифенадин, trade name: Phencarol, Фенкарол) is a 2nd generation antihistamine drug, marketed mainly in post-Soviet countries.[2][3] Chemically, it is a quinuclidine derivative.
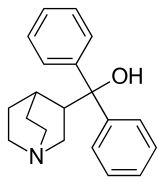 | |
| Clinical data | |
|---|---|
| Trade names | Fencarol |
| Other names | 3-Quinuclidinyldiphenylmethanol |
| Routes of administration | Oral (tablets), IM injection |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 45% (Tmax = 1 hour)[1] |
| Metabolism | Liver |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C20H23NO |
| Molar mass | 293.40 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Indications
- Allergic rhinitis
- Acute and chronic urticaria
- Angioedema
- Dermatitis
- Atopic dermatitis
- Pruritus[1]
References
- "Fencarol (quifenadine) Tablets, for Oral Use. Full Prescribing Information". State Register of Medicines (in Russian). Retrieved 4 January 2016.
- "Quifenadine". Drugs.com. Retrieved 28 January 2014.
- Makarov, L.; Balykova, L.; Soldatova, O.; Komolyatova, V.; Serebruany, V. (2010). "The antiarrhythmic properties of quifenadine, H1-histamine receptor blocker in children with premature beats: A randomized controlled pilot trial". American Journal of Therapeutics. 17 (4): 396–401. doi:10.1097/MJT.0b013e3181a86987. PMID 19487925.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.