Pulmonary alveolus
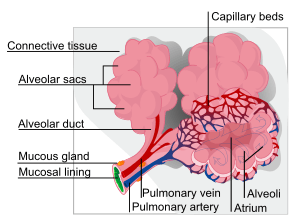
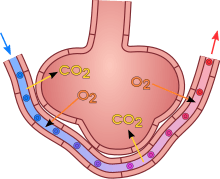
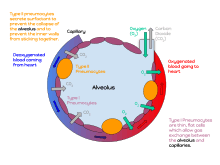
A pulmonary alveolus (plural: alveoli, from Latin alveolus, "little cavity") is a hollow cup-shaped cavity found in the lung parenchyma where gas exchange takes place. Lung alveoli are found in the acini at the beginning of the respiratory zone. They are located sparsely on the respiratory bronchioles, line the walls of the alveolar ducts, and are more numerous in the blind-ended alveolar sacs. The acini are the basic units of respiration, with gas exchange taking place in all the alveoli present.[1] The alveolar membrane is the gas exchange surface, surrounded by a network of capillaries. Across the membrane oxygen is diffused into the capillaries and carbon dioxide released from the capillaries into the alveoli to be breathed out.[2][3]
| Pulmonary alveolus | |
|---|---|
 The alveoli | |
| Details | |
| System | Respiratory system |
| Location | Lung |
| Identifiers | |
| Latin | alveolus pulmonis |
| MeSH | D011650 |
| TH | H3.05.02.0.00026 |
| Anatomical terminology | |
Alveoli are particular to mammalian lungs. Different structures are involved in gas exchange in other vertebrates.[4]
Structure
_(14577681128).jpg)
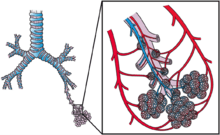
The alveoli are located in the alveolar sacs of the lungs in the pulmonary lobules of the respiratory zone, representing the smallest functional units in the respiratory tract. They are also present in the respiratory bronchioles as scattered outpockets, extending from their lumens. The respiratory bronchioles lead into alveolar ducts which are deeply lined with alveoli. Each respiratory bronchiole gives rise to between two and eleven alveolar ducts. Each duct opens into five or six alveolar sacs into which clusters of alveoli open. New alveoli continue to form until the age of eight years.[5]
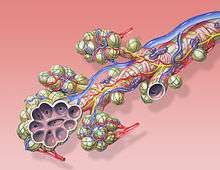
A typical pair of human lungs contain about 300 million alveoli, producing 70 m2 (750 sq ft) of surface area.[6] Each alveolus is wrapped in a fine mesh of capillaries covering about 70% of its area. The diameter of an alveolus is between 200 and 500 µm.[6]
Microanatomy
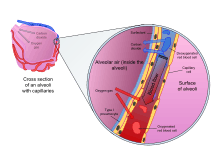
The alveoli consist of an epithelial layer of simple squamous epithelium,[7] and an extracellular matrix surrounded by capillaries. The epithelial lining is part of the alveolar membrane, also known as the respiratory membrane, that allows the exchange of gases. The membrane has several layers – a layer of lining fluid that contains surfactant, the epithelial layer and its basement membrane; a thin interstitial space between the epithelial lining and the capillary membrane; a capillary basement membrane that often fuses with the alveolar basement membrane, and the capillary endothelial membrane. The whole membrane however is only between 0.2 µm at its thinnest part and 0.6 µm at its thickest.[8]
In the alveolar walls there are interconnecting air passages between the alveoli known as the pores of Kohn. The alveoli contain some collagen fibers and elastic fibers. The elastic fibres allow the alveoli to stretch when they fill with air during inhalation. They then spring back during exhalation in order to expel the carbon dioxide-rich air.
There are three major types of alveolar cell. Two types are pneumocytes known as type I and type II cells found in the alveolar wall, and a large phagocytic cell known as an alveolar macrophage that moves about in the lumens of the alveoli, and in the connective tissue between them.[6] Type I cells are squamous, thin and flat and form the structure of the alveoli. Type II cells release pulmonary surfactant to lower surface tension. Type II cells can also differentiate to replace damaged type I cells.
Function
Type I cells
Type I cells are thin and flat epithelial lining cells, that form the structure of the alveoli. They are squamous (giving more surface area to each cell) and their long cytoplasmic extensions line more than 95% of the alveolar surface.[6][9]
Type I cells are involved in the process of gas exchange between the alveoli and blood. These cells are extremely thin sometimes only 25 nm – the electron microscope was needed to prove that all alveoli are lined with epithelium. This thin lining enables a fast diffusion of gas exchange between the air in the alveoli and the blood in the surrounding capillaries.
The nucleus of a type I cell occupies a large area of free cytoplasm and its organelles are clustered around it reducing the thickness of the cell. This also keeps the thickness of the blood-air barrier reduced to a minimum.
The cytoplasm in the thin portion contains pinocytotic vesicles which may play a role in the removal of small particulate contaminants from the outer surface. In addition to desmosomes, all type I alveolar cells have occluding junctions that prevent the leakage of tissue fluid into the alveolar air space.
The relatively low solubility (and hence rate of diffusion) of oxygen, necessitates the large internal surface area (about 80 square m [96 square yards]) and very thin walls of the alveoli. Weaving between the capillaries and helping to support them is an extracellular matrix, a meshlike fabric of elastic and collagenous fibres. The collagen fibres, being more rigid, give the wall firmness, while the elastic fibres permit expansion and contraction of the walls during breathing.
Type I pneumocytes are unable to replicate and are susceptible to toxic insults. In the event of damage, type II cells can proliferate and differentiate into type I cells to compensate.
Type II cells
Type II cells are the most numerous cells in the alveoli, yet do not cover as much surface area as the squamous type I cells. Type II cells in the alveolar wall contain secretory granular organelles known as lamellar bodies that fuse with the cell membranes and secrete pulmonary surfactant. This surfactant is a film of fatty substances, a group of phospholipids that reduce alveolar surface tension. The phospholipids are stored in the lamellar bodies. Without this coating, the alveoli would collapse. The surfactant is continuously released by exocytosis. Reinflation of the alveoli following exhalation is made easier by the surfactant, that reduces surface tension in the thin fluid coating of the alveoli. The fluid coating is produced by the body in order to facilitate the transfer of gases between blood and alveolar air, and the type II cells are typically found at the blood-air barrier. [10][11]
Type II cells start to develop at about 26 weeks of gestation, secreting small amounts of surfactant. However, adequate amounts of surfactant are not secreted until about 35 weeks of gestation – this is the main reason for increased rates of infant respiratory distress syndrome, which drastically reduces at ages above 35 weeks gestation.
Type II cells are also capable of cellular division, giving rise to more type I and II alveolar cells when the lung tissue is damaged.[12]
MUC1, a human gene associated with type II pneumocytes, has been identified as a marker in lung cancer.[13]
Macrophages
The alveolar macrophages reside on the internal lumenal surfaces of the alveoli, the alveolar ducts, and the bronchioles. They are mobile scavengers that serve to engulf foreign particles in the lungs, such as dust, bacteria, carbon particles, and blood cells from injuries.[14] They are also called dust cells.
Clinical significance
Diseases
Surfactant
Insufficient surfactant in the alveoli is one of the causes that can contribute to atelectasis (collapse of part or all of the lung). Without pulmonary surfactant, atelectasis is a certainty.[15] Insufficient surfactant in the lungs of preterm infants causes infant respiratory distress syndrome (IRDS). Impaired surfactant regulation can cause an accumulation of surfactant proteins to build up in the alveoli in a condition called pulmonary alveolar proteinosis. This results in impaired gas exchange.
Inflammation
Pneumonia is an inflammatory condition of the lung parenchyma, which can be caused by both viruses and bacteria. Cytokines and fluids are released into the alveolar cavity, interstitium, or both, in response to infection, causing the effective surface area of gas exchange to be reduced. In severe cases where cellular respiration cannot be maintained, supplemental oxygen may be required.[16][17]
- Diffuse alveolar damage can be a cause of acute respiratory distress syndrome(ARDS) a severe inflammatory disease of the lung. [18]:187
- In asthma, the bronchioles, or the "bottle-necks" into the sac are restricted, causing the amount of air flow into the lungs to be greatly reduced. It can be triggered by irritants in the air, photochemical smog for example, as well as substances that a person is allergic to.
- Chronic bronchitis occurs when an abundance of mucus is produced by the lungs. The production of this substance occurs naturally when the lung tissue is exposed to irritants. In chronic bronchitis, the air passages into the alveoli, the respiratory bronchioles, become clogged with mucus. This causes increased coughing in order to remove the mucus, and is often a result of extended periods of exposure to cigarette smoke.
- Hypersensitivity pneumonitis
Structural
Almost any type of lung tumor or lung cancer can compress the alveoli and reduce gas exchange capacity. In some cases the tumor will fill the alveoli.[19]
- Cavitary pneumonia is a process in which the alveoli are destroyed and produce a cavity. As the alveoli are destroyed, the surface area for gas exchange to occur becomes reduced. Further changes in blood flow can lead to decline in lung function.
- Emphysema is another disease of the lungs, whereby the elastin in the walls of the alveoli is broken down by an imbalance between the production of neutrophil elastase (elevated by cigarette smoke) and alpha-1 antitrypsin (the activity varies due to genetics or reaction of a critical methionine residue with toxins including cigarette smoke). The resulting loss of elasticity in the lungs leads to prolonged times for exhalation, which occurs through passive recoil of the expanded lung. This leads to a smaller volume of gas exchanged per breath.
- Pulmonary alveolar microlithiasis is a rare lung disorder of small stone formation in the alveoli.
Fluid
A pulmonary contusion is a bruise of the lung tissue caused by trauma.[20] Damaged capillaries can cause blood and other fluids to accumulate in the tissue of the lung, impairing gas exchange .
Pulmonary edema is the build up of fluid in the parenchyma and alveoli usually caused by left ventricular heart failure, or by damage to the lung or its vasculature.
See also
- Interstitial lung disease
- A549 cells
References
- Hansen, J. E.; Ampaya, E. P.; Bryant, G. H. & Navin, J. J. (1975). "The Branching Pattern of Airways and Air Spaces of a Single Human Terminal Bronchiole". Journal of Applied Physiology. 38 (6): 983–989. PMID 1141138.
- C. Michael Hogan. 2011. "Respiration". Encyclopedia of Earth. Eds. Mark McGinley & C. J. Cleveland. National council for Science and the Environment. Washington, D.C.
- Steve, Paxton,; Michelle, Peckham,; Adele, Knibbs (2003). "The Leeds Histology Guide".CS1 maint: extra punctuation (link)
- Daniels, Christopher B. & Orgeig, Sandra (2003). "Pulmonary Surfactant: The Key to the Evolution of Air Breathing". News in Physiological Sciences. 18 (4): 151–157. PMID 12869615.
- Moore, Keith (2018). Clinically oriented anatomy. Wolters Kluwer. p. 336. ISBN 9781496347213.
- Saladin, K (2011). Human anatomy (3rd ed.). McGraw-Hill. pp. 641–643. ISBN 9780071222075.
- "Bronchi, Bronchial Tree & Lungs". nih.gov. Retrieved 17 September 2019.
- Hall, John (2011). Guyton and Hall Textbook of Medical Physiology. Saunders Elsevier. pp. 489–491. ISBN 9781416045748.
- Weinberger, Steven; Cockrill, Barbara; Mandell, Jess (2019). Principles of pulmonary medicine (Seventh ed.). Elsevier. pp. 126–129. ISBN 9780323523714.
- "Histology, A Text and Atlas, Sixth Edition," 2011, by Ross, Michael H, and Pawlina, Wojciech
- Fehrenbach H. 2001. "Alveolar epithelial type II cell: defender of the alveolus revisited." Respir Res.2(1):33–46.
- "Lung – Regeneration – Nonneoplastic Lesion Atlas". ntp.niehs.nih.gov. Retrieved 2018-05-18.
- Jarrard JA, Linnoila RI, Lee H, Steinberg SM, Witschi H, Szabo E (December 1998). "MUC1 is a novel marker for the type II pneumocyte lineage during lung carcinogenesis". Cancer Res. 58 (23): 5582–9. PMID 9850098.
- By: the editors of Encyclopædia Britannica
- Saladin, Kenneth S. (2007). Anatomy and Physiology: the unity of form and function. New York: McGraw Hill. ISBN 0-07-322804-4.
- "Pneumonia – Symptoms and causes". Mayo Clinic. Retrieved 2019-06-10.
- "Pneumonia Symptoms and Diagnosis". American Lung Association. Retrieved 2019-06-10.
- Britton, the editors Nicki R. Colledge, Brian R. Walker, Stuart H. Ralston; illustrated by Robert (2010). Davidson's principles and practice of medicine (21st ed.). Edinburgh: Churchill Livingstone/Elsevier. ISBN 978-0-7020-3085-7.
- Hasleton, Philip (1996). Spencer’s Pathology of the Lung. McGraw-Hill. p. 1076. ISBN 0071054480.
- "Pulmonary Contusion – Injuries and Poisoning". Merck Manuals Consumer Version. Retrieved 2019-06-10.
External links
| Wikimedia Commons has media related to Pulmonary alveoli. |
- Pulmonary+Alveoli at the US National Library of Medicine Medical Subject Headings (MeSH)