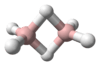Phosphine
Phosphine (IUPAC name: phosphane) is the compound with the chemical formula PH3. It is a colorless, flammable, toxic gas and is classed as a pnictogen hydride. Pure phosphine is odorless, but technical grade samples have a highly unpleasant odor like garlic or rotting fish, due to the presence of substituted phosphine and diphosphane (P2H4). With traces of P2H4 present, PH3 is spontaneously flammable in air (pyrophoric), burning with a luminous flame. Phosphines are also a group of organophosphorus compounds with the formula R3P (R = organic derivative). Organophosphines are important in catalysts where they complex to various metal ions; complexes derived from a chiral phosphine can catalyze reactions to give chiral, enantioenriched products.
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Phosphane | |||
| Other names
Phosphamine Phosphorus trihydride Phosphorated hydrogen | |||
| Identifiers | |||
CAS Number |
|||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.029.328 | ||
| EC Number |
| ||
Gmelin Reference |
287 | ||
PubChem CID |
|||
| RTECS number |
| ||
| UN number | 2199 | ||
CompTox Dashboard (EPA) |
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula |
PH3 | ||
| Molar mass | 33.99758 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Faint, fish-like or garlic-like[1] | ||
| Density | 1.379 g/l, gas (25 °C) | ||
| Melting point | −132.8 °C (−207.0 °F; 140.3 K) | ||
| Boiling point | −87.7 °C (−125.9 °F; 185.5 K) | ||
Solubility in water |
31.2 mg/100 ml (17 °C) | ||
| Solubility | Soluble in alcohol, ether, CS2 slightly soluble in benzene, chloroform, ethanol | ||
| Vapor pressure | 41.3 atm (20 °C)[1] | ||
| Conjugate acid | Phosphonium (chemical formula PH+ 4) | ||
Refractive index (nD) |
2.144 | ||
| Viscosity | 1.1×10−5 Pa⋅s | ||
| Structure | |||
Molecular shape |
Trigonal pyramidal | ||
Dipole moment |
0.58 D | ||
| Thermochemistry | |||
Heat capacity (C) |
37 J/mol⋅K | ||
Std molar entropy (S |
210 J/mol⋅K[2] | ||
Std enthalpy of formation (ΔfH⦵298) |
5 kJ/mol[2] | ||
Gibbs free energy (ΔfG˚) |
13 kJ/mol | ||
| Hazards | |||
| Safety data sheet | ICSC 0694 | ||
| GHS pictograms |     | ||
| NFPA 704 (fire diamond) | |||
| Flash point | Flammable gas | ||
Autoignition temperature |
38 °C (100 °F; 311 K) (see text) | ||
| Explosive limits | 1.79–98%[1] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
3.03 mg/kg (rat, oral) | ||
LC50 (median concentration) |
11 ppm (rat, 4 hr)[3] | ||
LCLo (lowest published) |
1000 ppm (mammal, 5 min) 270 ppm (mouse, 2 hr) 100 ppm (guinea pig, 4 hr) 50 ppm (cat, 2 hr) 2500 ppm (rabbit, 20 min) 1000 ppm (human, 5 min)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 0.3 ppm (0.4 mg/m3)[1] | ||
REL (Recommended) |
TWA 0.3 ppm (0.4 mg/m3), ST 1 ppm (1 mg/m3)[1] | ||
IDLH (Immediate danger) |
50 ppm[1] | ||
| Related compounds | |||
Other cations |
Ammonia Arsine Stibine Bismuthine | ||
Related compounds |
Trimethylphosphine Triphenylphosphine | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
History
Philippe Gengembre (1764–1838), a student of Lavoisier, first obtained phosphine in 1783 by heating phosphorus in an aqueous solution of potash (potassium carbonate).[4][5]
Perhaps because of its strong association with elemental phosphorus, phosphine was once regarded as a gaseous form of the element, but Lavoisier (1789) recognised it as a combination of phosphorus with hydrogen and described it as phosphure d'hydrogène (phosphide of hydrogen).[6]
In 1844, Paul Thénard, son of the French chemist Louis Jacques Thénard, used a cold trap to separate diphosphine from phosphine that had been generated from calcium phosphide, thereby demonstrating that P2H4 is responsible for spontaneous flammability associated with PH3, and also for the characteristic orange/brown color that can form on surfaces, which is a polymerisation product.[7] He considered diphosphine's formula to be PH2, and thus an intermediate between elemental phosphorus, the higher polymers, and phosphine. Calcium phosphide (nominally Ca3P2) produces more P2H4 than other phosphides because of the preponderance of P-P bonds in the starting material.
The name "phosphine" first appeared in combined form in 1857.[8] The gas PH3 was named "phosphine" by 1865 (or earlier).[9]
Structure and properties
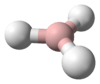
PH3 is a trigonal pyramidal molecule with C3v molecular symmetry. The length of the P-H bond is 1.42 Å, the H-P-H bond angles are 93.5°. The dipole moment is 0.58 D, which increases with substitution of methyl groups in the series: CH3PH2, 1.10 D; (CH3)2PH, 1.23 D; (CH3)3P, 1.19 D. In contrast, the dipole moments of amines decrease with substitution, starting with ammonia, which has a dipole moment of 1.47 D. The low dipole moment and almost orthogonal bond angles lead to the conclusion that in PH3 the P-H bonds are almost entirely pσ(P) – sσ(H) and phosphorus 3s orbital contributes little to the bonding between phosphorus and hydrogen in this molecule. For this reason, the lone pair on phosphorus may be regarded as predominantly formed by the 3s orbital of phosphorus. The upfield chemical shift of the phosphorus atom in the 31P NMR spectrum accords with the conclusion that the lone pair electrons occupy the 3s orbital (Fluck, 1973). This electronic structure leads to a lack of nucleophilicity in general and lack of basicity in particular (pKaH = –14),[10] as well as an ability to form only weak hydrogen bonds.[11]
The aqueous solubility of PH3 is slight; 0.22 mL of gas dissolve in 1 mL of water. Phosphine dissolves more readily in non-polar solvents than in water because of the non-polar P-H bonds. It is technically amphoteric in water, but acid and base activity is poor. Proton exchange proceeds via a phosphonium (PH4+) ion in acidic solutions and via PH2− at high pH, with equilibrium constants Kb = 4 × 10−28 and Ka = 41.6 × 10−29.
Phosphine burns producing a dense white cloud of phosphoric acid:
- PH3 + 2 O2 → H3PO4
Preparation and occurrence
Phosphine may be prepared in a variety of ways.[12] Industrially it can be made by the reaction of white phosphorus with sodium or potassium hydroxide, producing potassium or sodium hypophosphite as a by-product.
- 3 KOH + P4 + 3 H2O → 3 KH2PO2 + PH3
Alternatively the acid-catalyzed disproportioning of white phosphorus yields phosphoric acid and phosphine. Both routes have industrial significance; the acid route is preferred method if further reaction of the phosphine to substituted phosphines is needed. The acid route requires purification and pressurizing. It can also be made (as described above) by the hydrolysis of a metal phosphide, such as aluminium phosphide or calcium phosphide. Pure samples of phosphine, free from P2H4, may be prepared using the action of potassium hydroxide on phosphonium iodide (PH4I).
Laboratory routes
It is prepared in the laboratory by disproportionation of phosphorous acid[13]
- 4 H3PO3 → PH3 + 3 H3PO4
Phosphine evolution occurs around 200 °C. Alternative methods involve the hydrolysis of aluminium phosphide, calcium phosphide, and tris(trimethylsilyl)phosphine.
Occurrence
Phosphine is a constituent of the atmosphere at very low and highly variable concentrations.[14] It may contribute significantly to the global phosphorus biochemical cycle. The most likely source is reduction of phosphate in decaying organic matter, possibly via partial reductions and disproportionations, since environmental systems do not have known reducing agents of sufficient strength to directly convert phosphate to phosphine.[15]
Phosphines
Organophosphines are compounds with the formula PRnH3−n. These compounds are often classified according to the value of n: primary phosphines (n = 1), secondary phosphines (n = 2), tertiary phosphines (n = 3). All adopt pyramidal structures. Their reactivity is also similar – they can be oxidized to the phosphorus(V) level, they can be protonated and alkylated at phosphorus to give phosphonium salts, and, for primary and secondary derivatives, they can be deprotonated by strong bases to give organophosphide derivatives.
Primary phosphines
Primary phosphines are typically prepared by alkylation of phosphine. Simple alkyl derivatives such as methylphosphine (CH3PH2) are prepared by alkylation of alkali metal derivatives MPH2 (M = Li, Na, K). Another synthetic route involves treatment of the corresponding chlorophosphines with hydride reagents. For example, reduction of dichlorophenylphosphine with lithium aluminium hydride affords phenylphosphine (PhPH2).
Secondary phosphines
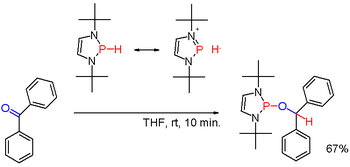
Secondary phosphines are prepared analogously to the primary phosphines. They are also obtained by alkali-metal reductive cleavage of triarylphosphines followed by hydrolysis of the resulting phosphide salt. The latter route is employed to prepare diphenylphosphine (Ph2PH). Diorganophosphinic acids, R2P(O)OH, can also be reduced with diisobutylaluminium hydride. Secondary phosphines are typically protic in character. When however modified with suitable substituents as in certain (rare) diazaphospholenes (scheme 3) the polarity of the P-H bond can be inverted (see: umpolung) and the resulting phosphine hydride can reduce a carbonyl group as in the example of benzophenone in yet another way.[16]
Tertiary phosphines
Tertiary phosphines are generally obtained by treatment of phosphorus trichloride or triphenylphosphite with organolithium reagents or Grignard reagents. They are commonly used as ligands in coordination chemistry. Tertiary phosphines of the type PRR'R" are "P-chiral" and optically stable.
Cyclic phosphines
Secondary and tertiary phosphines occur in cyclic forms. Three-membered rings are phosphiranes (unsaturated: phosphirenes), five-membered rings are phospholanes (unsaturated: phosphole), and six-membered rings are phosphinanes.
Applications
Organophosphorus chemistry
Phosphine is mainly consumed as an intermediate in organophosphorus chemistry. In an illustrative reaction, formaldehyde adds in the presence of hydrogen chloride to give tetrakis(hydroxymethyl)phosphonium chloride, which is used in textiles.
Microelectronics
Small amounts of phosphine are used as a dopant in the semiconductor industry, and a precursor for the deposition of compound semiconductors.[17]
Fumigant
For farm use, pellets of aluminium phosphide, calcium phosphide, or zinc phosphide release phosphine upon contact with atmospheric water or rodents' stomach acid. These pellets also contain agents to reduce the potential for ignition or explosion of the released phosphine. A more recent alternative is the use of phosphine gas itself which requires dilution with either CO2 or N2 or even air to bring it below the flammability point. Use of the gas avoids the issues related with the solid residues left by metal phosphide and results in faster, more efficient control of the target pests.
Because the previously popular fumigant methyl bromide has been phased out in some countries under the Montreal Protocol, phosphine is the only widely used, cost-effective, rapidly acting fumigant that does not leave residues on the stored product. Pests with high levels of resistance toward phosphine have become common in Asia, Australia and Brazil. High level resistance is also likely to occur in other regions, but has not been as closely monitored. Genetic variants that contribute to high level resistance to phosphine have been identified in the dihydrolipoamide dehydrogenase gene.[18] Identification of this gene now allows rapid molecular identification of resistant insects.
Safety
Phosphine gas is denser than air and hence may collect in low-lying areas. It can form explosive mixtures with air and also self-ignite.
Phosphine can be absorbed into the body by inhalation. Direct contact with phosphine liquid – although unlikely to occur – may cause frostbite, like other cryogenic liquids. The main target organ of phosphine gas is the respiratory tract.[19] According to the 2009 U.S. National Institute for Occupational Safety and Health (NIOSH) pocket guide, and U.S. Occupational Safety and Health Administration (OSHA) regulation, the 8 hour average respiratory exposure should not exceed 0.3 ppm. NIOSH recommends that the short term respiratory exposure to phosphine gas should not exceed 1 ppm. The Immediately Dangerous to Life or Health level is 50 ppm. Overexposure to phosphine gas causes nausea, vomiting, abdominal pain, diarrhea, thirst, chest tightness, dyspnea (breathing difficulty), muscle pain, chills, stupor or syncope, and pulmonary edema.[20][21] Phosphine has been reported to have the odor of decaying fish or garlic at concentrations below 0.3 ppm. The smell is normally restricted to laboratory areas or phosphine processing since the smell comes from the way the phosphine is extracted from the environment. However, it may occur elsewhere, such as in industrial waste landfills. Exposure to higher concentrations may cause olfactory fatigue.[22]
Toxicity
Deaths have resulted from accidental exposure to fumigation materials containing aluminum phosphide or phosphine.[23][24][25][26] It can be absorbed either by inhalation or transdermally.[23] As a respiratory poison, it affects the transport of oxygen or interferes with the utilization of oxygen by various cells in the body.[25] Exposure results in pulmonary edema (the lungs fill with fluid).[26] Phosphine gas is heavier than air so stays nearer the floor where children are likely to play.[27]
In popular culture
In the pilot episode of Breaking Bad, Walter White killed Krazy-8 and Emilio Koyama by generating phosphine gas.
See also
- Diphosphane, H2PPH2, simplified to H4P2
- Diphosphines, R2PPR2, R2P(CH2)nPR2
- Diphosphene, HP=PH
- Diphosphenes, R-P=P-R'
- Phosphine oxide, R3P=O
- Phosphorane, PR5, R3P=CR2
- Phosphinite, P(OR)R2
- Phosphonite, P(OR)2R
- Phosphite, P(OR)3
- Phosphinate, R2P(RO)O
- Phosphonate, RP(RO)2O
- Phosphate, P(RO)3O
References
- NIOSH Pocket Guide to Chemical Hazards. "#0505". National Institute for Occupational Safety and Health (NIOSH).
- Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A22. ISBN 0-618-94690-X.
- "Phosphine". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- Gengembre (1783) "Mémoire sur un nouveau gas obtenu, par l'action des substances alkalines, sur le phosphore de Kunckel" (Memoir on a new gas obtained by the action of alkaline substances on Kunckel's phosphorus), Mémoires de mathématique et de physique, 10 : 651–658.
- For further information about the early history of phosphine, see:
- The Encyclopædia Britannica (1911 edition), vol. 21, p. 480: Phosphorus: Phosphine.
- Thomas Thomson, A System of Chemistry, 6th ed. (London, England: Baldwin, Cradock, and Joy, 1820), vol. 1, p. 272.
- Note:
- On p. 222 of his Traité élémentaire de chimie, vol. 1, (Paris, France: Cuchet, 1789), Lavoisier calls the compound of phosphorus and hydrogen "phosphure d'hydrogène" (hydrogen phosphide). However, on p. 216, he calls the compound of hydrogen and phosphorus "Combinaison inconnue." (unknown combination), yet in a footnote, he says about the reactions of hydrogen with sulfur and with phosphorus: "Ces combinaisons ont lieu dans l'état de gaz & il en résulte du gaz hydrogène sulfurisé & phosphorisé." (These combinations occur in the gaseous state, and there results from them sulfurized and phosphorized hydrogen gas.)
- In Robert Kerr's 1790 English translation of Lavoisier's Traité élémentaire de chimie … — namely, Lavoisier with Robert Kerr, trans., Elements of Chemistry … (Edinburgh, Scotland: William Creech, 1790) — Kerr translates Lavoisier's "phosphure d'hydrogène" as "phosphuret of hydrogen" (p. 204), and whereas Lavoisier — on p. 216 of his Traité élémentaire de chimie … — gave no name to the combination of hydrogen and phosphorus, Kerr calls it "hydruret of phosphorus, or phosphuret of hydrogen" (p. 198). Lavoisier's note about this compound — "Combinaison inconnue." — is translated: "Hitherto unknown." Lavoisier's footnote is translated as: "These combinations take place in the state of gas, and form, respectively, sulphurated and phosphorated oxygen gas." The word "oxygen" in the translation is an error because the original text clearly reads "hydrogène" (hydrogen). (The error was corrected in subsequent editions.)
- Paul Thénard (1844) "Mémoire sur les combinaisons du phosphore avec l'hydrogène" (Memoir on the compounds of phosphorus with hydrogen), Comptes rendus, 18 : 652–655.
- In 1857, August Wilhelm von Hofmann announced the synthesis of organic compounds containing phosphorus, which he named "trimethylphosphine" and "triethylphosphine", in analogy with "amine" (organo-nitrogen compounds), "arsine" (organo-arsenic compounds), and "stibine" (organo-antimony compounds). See: A.W. Hofmann and Auguste Cahours (1857) "Researches on the phosphorus bases," Proceedings of the Royal Society of London, 8 : 523–527. From page 524: "The bases Me3P and E3P, the products of this reaction, which we propose to call respectively trimethylphosphine and triethylphosphine, … "
- William Odling, A Course of Practical Chemistry Arranged for the Use of Medical Students, 2nd ed. (London, England: Longmans, Green, and Co., 1865), pp. 227, 230.
- Streitwieser, Andrew; Heathcock, Clayton H.; Kosower, Edward M. (2017). Introduction to Organic Chemistry. New Delhi: Medtech (Scientific International, reprint of revised 4th edition, Macmillan, 1998). p. 828. ISBN 9789385998898.
- Sennikov, P. G. (1994). "Weak H-Bonding by Second-Row (PH3, H2S) and Third-Row (AsH3, H2Se) Hydrides". Journal of Physical Chemistry. 98 (19): 4973–4981. doi:10.1021/j100070a006.
- Toy, A. D. F. (1973). The Chemistry of Phosphorus. Oxford, UK: Pergamon Press.
- Gokhale, S. D.; Jolly, W. L., "Phosphine", Inorganic Syntheses 1967, volume 9, pp. 56–58. doi:10.1002/9780470132401.ch17
- Gassmann, G.; van Beusekom, J. E. E.; Glindemann, D. (1996). "Offshore atmospheric phosphine". Naturwissenschaften. 83 (3): 129–131. Bibcode:1996NW.....83..129G. doi:10.1007/BF01142178.
- Roels, J.; Verstraete, W. (2001). "Biological formation of volatile phosphorus compounds, a review paper". Bioresource Technology. 79 (3): 243–250. doi:10.1016/S0960-8524(01)00032-3. PMID 11499578.
- Burck, S.; Gudat, D.; Nieger, M.; Du Mont, W.-W. (2006). "P-Hydrogen-Substituted 1,3,2-Diazaphospholenes: Molecular Hydrides". Journal of the American Chemical Society. 128 (12): 3946–3955. doi:10.1021/ja057827j.
- Bettermann, G.; Krause, W.; Riess, G.; Hofmann, T. (2002). "Phosphorus Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_527. ISBN 3527306730.
- Schlipalius, D. I., Valmas, N., Tuck, A. G., Jagadeesan, R., Ma, L., Kaur, R. et al. (2012). "A Core Metabolic Enzyme Mediates Resistance to Phosphine Gas". Science. 338: 807–810. Bibcode:2012Sci...338..807S. doi:10.1126/science.1224951. PMID 23139334.CS1 maint: uses authors parameter (link)
- "NIOSH Emergency Response Card". CDC. Retrieved 6 April 2010.
- "NIOSH pocket guide". CDC. 3 February 2009. Retrieved 6 April 2010.
- "WHO (Data Sheets on Pesticides-No. 46): Phosphine". Inchem.org. Retrieved 6 April 2010.
- "NIOSH Alert: Preventing Phosphine Poisoning and Explosions during Fumigation". CDC. 10 July 1995. Retrieved 6 April 2010.
- "Two toddlers die after Jerusalem home sprayed for pests". Haaretz. 22 January 2014. Retrieved 23 January 2014.
- rtve.es: "La familia de Alcalá de Guadaíra murió tras inhalar fosfina de unos tapones", 2014-02-03
- "Deaths of Quebec women in Thailand may have been caused by pesticide". cbc.ca. 13 March 2014.
- "4 children killed after pesticide released toxic gas underneath their home, police say". Washington Post. Retrieved 6 January 2017.
- "Pesticide blamed in 8-month-old's death in Fort McMurray". CBC News. 23 February 2015. Retrieved 23 February 2015.
Further reading
- Fluck, E. (1973). "The Chemistry of Phosphine". Topics in Current Chemistry. Fortschritte der Chemischen Forschung. 35: 1–64. doi:10.1007/BFb0051358. ISBN 3-540-06080-4.
- WHO (World Health Organisation) (1988). Phosphine and Selected Metal Phosphides. Environmental Health Criteria. 73. Geneva: Joint sponsorship of UNEP, ILO and WHO.