Original antigenic sin
Original antigenic sin, also known as the Hoskins effect,[1] refers to the propensity of the body's immune system to preferentially utilize immunological memory based on a previous infection when a second slightly different version of that foreign entity (e.g. a virus or bacterium) is encountered. This leaves the immune system "trapped" by the first response it has made to each antigen, and unable to mount potentially more effective responses during subsequent infections. The phenomenon of original antigenic sin has been described in relation to influenza virus, dengue fever, human immunodeficiency virus (HIV), and to several other viruses.[2]

This phenomenon was first described in 1960 by Thomas Francis, Jr. in the article "On the Doctrine of Original Antigenic Sin".[3][4] It is named by analogy to the theological concept of original sin. According to Thomas Francis, who originally described the idea,[3] and cited by Richard Krause:[4]
"The antibody of childhood is largely a response to dominant antigen of the virus causing the first type A influenza infection of the lifetime. [...] The imprint established by the original virus infection governs the antibody response thereafter. This we have called the Doctrine of the Original Antigenic Sin."
In B cells
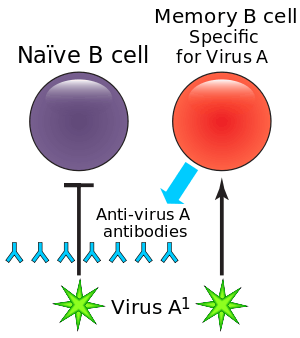
During a primary infection, long-lived memory B cells are generated, which remain in the body, and provide protection from subsequent infections. These memory B cells respond to specific epitopes on the surface of viral proteins in order to produce antigen-specific antibodies, and are able to respond to infection much faster than B cells are able to respond to novel antigens. This effect shortens the amount of time required to clear subsequent infections.
Between primary and secondary infections, or following vaccination, a virus may undergo antigenic drift, in which the viral surface proteins (the epitopes) are altered through natural mutation, allowing the virus to escape the immune system. When this happens, the altered virus preferentially reactivates previously activated high-affinity memory B cells and spur antibody production. However, the antibodies produced by these B cells generally ineffectively bind to the altered epitopes. In addition, these antibodies inhibit the activation of higher-affinity naive B cells that would be able to make more effective antibodies to the second virus. This leads to a less effective immune response and recurrent infections may take longer to clear.[5]
Original antigenic sin is of particular importance in the application of vaccines.[6] In dengue fever, the effect of original antigenic sin has important implications for vaccine development. Once a response against a dengue virus serotype has been established, it is unlikely that vaccination against a second will be effective, implying that balanced responses against all four virus serotypes have to be established with the first vaccine dose.[7] However, in 2015, a new class of highly potent, neutralizing antibodies that is effective against all four virus serotypes has been isolated, bringing hope for the development of a universal dengue vaccine.[8]
The specificity and the quality of the immune response against novel strains of influenza is often diminished in individuals who are repeatedly immunized (by vaccination or recurrent infections).[9] However, the impact of antigenic sin on protection has not been well established, and appears to differ with each infectious agent vaccine, geographic location, and age.[5] Researchers found reduced antibody responses to the 2009 pandemic H1N1 influenza vaccine in individuals who had been vaccinated against the seasonal A/Brisbane/59/2007 (H1N1) within the previous three months.[6]
In cytotoxic T cells
A similar phenomenon has been described in cytotoxic T cells (CTL).[10] It has been demonstrated that during the secondary infection by a different strain of dengue virus, the CTLs prefer to release cytokines instead of causing lysis of cell. As a result, the production of these cytokines are thought to increase vascular permeability and exacerbate the damage of endothelial cells.[11]
Several groups have attempted to design vaccines for HIV and hepatitis C based on induction of cytotoxic (CTL) responses. The finding that CTL may be biased by original antigenic sin, may help to explain the limited effectiveness of these vaccines. Viruses like HIV are highly variable and undergo mutation frequently, and thus, due to original antigenic sin, HIV infection induced by viruses that express slightly different epitopes (than those in a viral vaccine) would fail to be controlled by the vaccine. In fact, the vaccine might make the infection even worse, by "trapping" the immune response into the first, ineffective, response it made against the virus.[10]
See also
- Antibody-dependent enhancement
- Cell mediated immunity
- Humoral immunity
- Polyclonal response
References
- FDA Center for Biologics Evaluation and Research Vaccines and Related Biological Products: Advisory Committee(RTF)
- Deem, Michael W.The Adaptive Immune Response Rice University
- Thomas Francis Jr (1960). "On the doctrine of original antigenic sin". Proceedings of the American Philosophical Society. 104 (6): 572–578. JSTOR 985534.
- Krause R (2006). "The swine flu episode and the fog of epidemics". Emerg Infect Dis. 12 (1): 40–43. doi:10.3201/eid1201.051132. PMC 3291407. PMID 16494715.
- Lambert PH, Liu M, Siegrist CA (2005). "Can successful vaccines teach us how to induce efficient protective immune responses?". Nat Med 11(4 Suppl): S54-62,doi:10.1038/nm1216 PMID 15812491
- Choi, Yoon Seok; Baek, Yun Hee; Kang, Wonseok; et al. (September 2011). "Reduced Antibody Responses to the Pandemic (H1N1) 2009 Vaccine after Recent Seasonal Influenza Vaccination". Clinical and Vaccine Immunology. 18 (9): 1519–1523. doi:10.1128/CVI.05053-11. PMC 3165229. PMID 21813667.
- Midgley, Claire M.; Bajwa-Joseph, Martha; Vasanawathana, Sirijitt; et al. (January 2011). "An In-Depth Analysis of Original Antigenic Sin in Dengue Virus Infection". Journal of Virology. 85 (1): 410–421. doi:10.1128/JVI.01826-10. PMC 3014204. PMID 20980526.
- Wanwisa Dejnirattisai; Juthathip Mongkolsapaya; Wiyada Wongwiwat; Sunpetchuda Supasa; Xiaokang Zhang; Xinghong Dai; Alexander Rouvinski; Amonrat Jumnainsong; Carolyn Edwards; Nguyen Than Ha Quyen; Thaneeya Duangchinda; Jonathan M Grimes; Wen-Yang Tsai; Chih-Yun Lai; Wei-Kung Wang; Prida Malasit; Jeremy Farrar; Cameron P Simmons; Z Hong Zhou; Felix A Rey; Gavin R Screaton (2015). "A new class of highly potent, broadly neutralizing antibodies isolated from viremic patients infected with dengue virus". Nature Immunology. 16 (2): 170–177. doi:10.1038/ni.3058. hdl:10044/1/34691. PMC 4445969. PMID 25501631.
- Kim, J.H.; Skountzou, I.; Compans, R.; Jacob, J. (1 September 2009). "Original antigenic sin responses to influenza viruses". Journal of Immunology. 183 (5): 3294–301. doi:10.4049/jimmunol.0900398. PMC 4460008. PMID 19648276.
- McMichael AJ (1998). "The original sin of killer T cells". Nature. 394 (6692): 421–422. doi:10.1038/28738. PMID 9697760.
- Juthathip Mongkolsapaya (2006). "T Cell Responses in Dengue Hemorrhagic Fever: Are Cross-Reactive T Cells Suboptimal?". J. Immunol. 176 (6): 3821–3829. doi:10.4049/jimmunol.176.6.3821. PMID 16517753.