Leptospirosis
Leptospirosis is an infection caused by corkscrew-shaped bacteria called Leptospira.[5] Signs and symptoms can range from none to mild such as headaches, muscle pains, and fevers to severe with bleeding from the lungs or meningitis.[5][6] If the infection causes the person to turn yellow, have kidney failure and bleeding, it is then known as Weil's disease.[6] If it also causes bleeding into the lungs then it is known as severe pulmonary haemorrhage syndrome.[6]
| Leptospirosis | |
|---|---|
| Other names | Rat fever,[1] field fever,[2] rat catcher's yellows,[3] pretibial fever[4] |
 | |
| Leptospira magnified 200-fold with a dark-field microscope | |
| Specialty | Infectious disease |
| Symptoms | None, headaches, muscle pains, fevers[5] |
| Complications | Bleeding from the lungs, meningitis, kidney failure[5][6] |
| Usual onset | 7–12 days[7] |
| Causes | Leptospira typically spread by rodents[5][8] |
| Risk factors | Exposure to infected animals or contaminated water[9] |
| Diagnostic method | Testing blood for antibodies against the bacterium or its DNA[10] |
| Differential diagnosis | Malaria, enteric fever, rickettsiosis, dengue[11] |
| Treatment | Doxycycline, penicillin, ceftriaxone[5] |
| Frequency | ~8.5 million people per year[12] |
| Deaths | 58,900 per year[13] |
Up to ten different genetic types of Leptospira may cause disease in humans.[14] It is transmitted by both wild and domestic animals.[6] The most common animals that spread the disease are rodents.[8] It is often transmitted by animal urine or by water or soil containing animal urine coming into contact with breaks in the skin, eyes, mouth, or nose.[5][15] In the developing world the disease most commonly occurs in farmers and low-income people who live in cities.[6] In the developed world it most commonly occurs in those involved in outdoor activities in warm and wet areas of the world.[5] Diagnosis is typically by looking for antibodies against the bacterium or finding its DNA in the blood.[10]
Efforts to prevent the disease include protective equipment to prevent contact when working with potentially infected animals, washing after this contact, and reducing rodents in areas where people live and work.[5] The antibiotic doxycycline, when used in an effort to prevent infection among travelers, is of unclear benefit.[5] Vaccines for animals exist for certain type of Leptospira which may decrease the risk of spread to humans.[5] Treatment if infected is with antibiotics such as: doxycycline, penicillin, or ceftriaxone.[5] Weil's disease and severe pulmonary haemorrhage syndrome result in death rates greater than 10% and 50%, respectively, even with treatment.[6]
It is estimated that seven to ten million people are infected by leptospirosis per year.[12] This results in about 58,900 deaths per year.[13] The disease is most common in tropical areas of the world but may occur anywhere.[5] Outbreaks may occur in slums of the developing world.[6] The disease was first described by physician Adolf Weil in 1886 in Germany.[5][16] Animals which are infected may have no symptoms, mild symptoms, or severe symptoms.[17] Symptoms may vary by the type of animal.[17] In some animals Leptospira live in the reproductive tract, leading to transmission during mating.[18]
Signs and symptoms
The symptoms appear after an incubation period of 7–12 days.[7] However, the incubation period can vary from 6 days to 29 days.[19] Leptospirosis is a biphasic disease.[20] The first phase (acute or septic phase) ends after 3–7 days of illness.[7] The second phase (immune phase) starts with the resolution of symptoms and appearance of antibodies.[21] Ninety percent of cases of the disease are mild leptospirosis. The rest experience severe disease, which develops during the second stage or occurs as a single progressive illness.[22]
Leptospiral infection in humans causes a range of symptoms, and some infected persons may have no symptoms at all. The disease begins suddenly with fever accompanied by chills, intense headache, severe muscle aches, abdominal pain, and occasionally a skin rash.[20] The headache in leptospirosis is characteristically located at the bilateral temporal or frontal regions with throbbing pain. The person could also have pain behind the eyes and sensitivity to light.[19] Muscle pain usually involves the calf muscle and the lower back.[19] The most characteristic feature of leptospirosis is the conjunctival suffusion (conjunctivitis without exudate) which is not commonly found in other febrile illnesses. Other characteristic findings on the eye include subconjunctival bleeding and jaundice.[19] Rash is rarely found in leptospirosis. When rash is found, other alternative diagnoses such as Dengue fever and Chikungunya fever should be considered.[19] Dry cough is observed in 20% to 57% of people with leptospirosis. Thus, this clinical feature can mislead a doctor to diagnose the disease as respiratory illness. Additionally, gastrointestinal symptoms such as nausea, vomiting, abdominal pain, and diarrhea frequently occurs. Vomiting and diarrhea may contribute to dehydration in conjunction with kidney failure due to excessive urine output.[19] The abdominal pain can be due to acalculous cholecystitis or inflammation of the pancreas.[19] Rarely, the lymph nodes, liver, and spleen may be enlarged and palpable.[21]
The disappearance of symptoms coincides with the appearance of antibodies against Leptospira and the disappearance of bacteria from the bloodstream. The patient is asymptomatic for 3–4 days until the second phase begins with another episode of fever.[20] The immune phase can last from 4 to 30 days. It can be anything from brain to kidney complications.[23] The hallmark of the second phase is meningitis (inflammation of the membranes covering the brain).[24] Signs and symptoms of meningitis include severe headache and neck stiffness. In 5% to 10% of those infected with jaundice, the disease can be rapidly progressive to multiorgan failure.[25]
Severe leptospirosis can cause liver, kidney, lungs, and brain damage. For those with signs of inflammation of membranes covering the brain and the brain itself, altered level of consciousness can occur. A variety of neurological complications can occur such as paralysis of half of the body, complete inflammation of whole section of spinal cord, and muscle weakness due to immune damage of the nerves supplying the muscles.[19] Signs of bleeding such as non-traumatic bruises at 1 mm, non-traumatic bruises more than 1 cm, nose bleeding, blackish stools due to bleeding in stomach, vomiting blood and bleeding from the lungs can also be found. Prolongation of prothrombin time in coagulation testing is associated with severe bleeding manifestation. However, low platelet count is not associated with severe bleeding.[19] In less than 5% of those infected, pulmonary haemorrhage can occur at the 4th to 6th day of the illness and can be rapidly fatal.[25] Leptospira causes alveolar haemorrhage (bleeding into the alveoli of the lungs) and massive coughing up blood. This causes acute respiratory distress syndrome. This feature increases the risk of death to more than 50%.[19] Rarely, inflammation of heart muscles, inflammation of membranes covering the heart, abnormalities in pacemaker of the heart and abnormal heart rhythms may occur.[21] The classic form of severe leptospirosis is known as Weil's disease, which is characterized by liver damage (causing jaundice), kidney failure, and bleeding.[26]
Cause
Bacteria
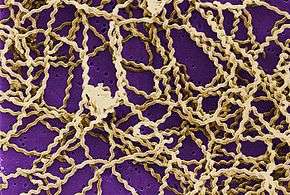
Leptospirosis belongs to the genus Leptospira which are aerobic,[21] right-handed helical[14] bacteria; sized at 6 to 20 micrometers. Hooked ends of this bacterium give it a "question mark" shape.[27] Like Gram-negative bacteria, Leptospira have an outer membrane studded with lipopolysaccharide (LPS) on the surface, an inner membrane, and a layer of peptidoglycan. However, unlike Gram-negative bacteria, the peptidoglycan layer in Leptospira lies closer to the inner membrane than the outer membrane. This results in a fluid outer membrane loosely associated with the cell wall.[28] In addition, Leptospira have two flagella located in the periplasm.[27] Chemoreceptors at the poles of the bacteria sense various substrates and change the direction of the bacteria's movement.[14] The bacteria are traditionally visualised using dark-field microscopy with silver staining or immunofluorescence staining.[29]
A total of 22 species of Leptospira have been identified.[14] The species can be divided into three clades based on their 16S rRNA sequence. The first is the "pathogens" clade comprising the eight species that can cause severe disease in humans: L. alexanderi, L. borgpetersenii, L. interrogans, L. kirschneri, L. mayottensis, L. noguchii, L. santarosai, and L. weilii, along with two related species that are not known to cause disease in any animal.[14] The second clade comprises five "intermediate" species that may cause mild disease in humans.[14] The remaining seven species are in the "saprophytes" clade which are known to consume decaying matter (saprotrophic nutrition).[14] Pathogenic Leptospira do not multiply in the environment. Leptospira require high humidity for survival but can remain alive in environments such as stagnant water or contaminated soil. The bacteria can be killed by temperatures of 50 °C (122 °F) and can be inactivated by 70% ethanol, 1% sodium hypochloride, formaldehyde, detergents, and acids.[25]
Leptospira are also classified based on their serovar. The diverse sugar composition of the lipopolysaccharide on the surface of the spirochete is responsible for the antigenic difference between serovars.[30] About 300 pathogenic serovars of Leptospira are recognized.[14] Antigenically related serovars (belonging to the same serogroup) may belong to different species because of horizontal gene transfer of LPS biosynthetic genes between different species.[14] Currently, the cross agglutination absorption test and DNA-DNA hybridisation are used to classify Leptospira species, but are time consuming. Therefore, total genomic sequencing could potentially replace these two methods as the new gold standard of classifying Leptospira species.[14]
Transmission
The bacteria can be found generally in various water-logged environments such as muddy riverbanks, ditches, gullies, and muddy livestock rearing areas where there is a regular passage of wild or farm mammals.[31] The L. interrogans species can survive in low nutrient environments such as moist soil and fresh water for prolonged periods.[27] However, L. borgpetersenii is devoid of critical genes necessary for survival in the environment.[27] Pathogenic Leptospira have been found in the form of aquatic biofilms, which may aid survival in the environment.[32]
The incidence of leptospirosis correlates directly with the amount of rainfall, making the disease seasonal in temperate climates and year-round in tropical climates.[31] The risk of contracting leptospirosis depends upon the risk of disease carriage in the community and the frequency of exposure.[19] In rural areas, farming and animal husbandry are the major risk factors for contracting leptospirosis.[27] Poor housing and inadequate sanitation also increase the risk of infection.[19] In tropical and semi-tropical areas, like South and South-east Asia, the disease often becomes widespread after heavy rains, sometimes after flooding. Periods of heavy rain followed by days of little or no rain seemed to be the setting for leptospirosis epidemics in these parts of the world.[33]
The bacteria is carried by various animals. Although Leptospira have been detected in reptiles and birds, only mammals are able to transmit the bacteria to humans and other animals.[34] Rats, mice, and moles are important primary hosts, but a wide range of other mammals including dogs, deer, rabbits, hedgehogs, cows, sheep, swine, raccoons, opossums, skunks, and certain marine mammals carry and transmit the disease as secondary hosts. In Africa, a number of wildlife hosts have been identified as carriers, including the banded mongoose, Egyptian fox, Rusa deer, and shrews.[35] There are various machanisms where the animals can infect each other. Dogs may lick the urine of an infected animal off the grass or soil, or drink from an infected puddle. House-bound domestic dogs have contracted leptospirosis, apparently from licking the urine of infected mice in the house.[19] Leptospirosis can also be transmitted via the semen of infected animals.[31] The duration of bacteria being consistently present in animal urine are long. In some animals such as cows, the bacterial shedding can persist for one year.[25]
Leptospirosis is transmitted by the urine of an infected animal and is contagious as long as the urine is still moist.[34] Humans become infected through contact with water, food, or soil that contains urine from these infected animals. This may happen by swallowing contaminated food or water or through skin contact.[36] The presence of wounds or fissures on the legs are a risk factor for acquiring the infection.[27] Occupations at risk of contracting leptospirosis include veterinarians, slaughterhouse workers, countryside rangers, farmers, sailors on rivers, sewer maintenance workers, waste disposal facility workers, and people who work on derelict buildings.[37] Slaughterhouse workers can contract the disease through contact with infected blood or body fluids.[9] The disease was once mostly work-related but is now often also related to adventure tourism and recreational activities.[6] Leptospirosis is common among water-sports enthusiasts in specific areas, as prolonged immersion in water promotes the entry of the bacterium. Surfers and whitewater paddlers[36] such as rowers, kayakers and canoeists[9] However, Leptospira is unlikely to penetrate intact skin.[21] The disease is not known to spread between humans, and bacterial dissemination in recovery period is extremely rare in humans.[21] Once humans are infected, bacterial shedding from the kidneys usually persists for up to 60 days.[25]
Rarely, leptospirosis can be transmitted through organ transplant.[38] Infection through the placenta during pregnancy is also possible.[39][40][41] Leptospirosis can cause miscarriage and infection in infants.[42]
Pathogenesis
When animals ingest the bacteria, the bacteria circulate in the blood stream, then lodge themselves into the kidneys through the glomerulular or peritubular capillaries. The bacteria then pass into the lumens of the renal tubules and colonise the brush border and proximal convoluted tubule. This causes the continuous shedding of bacteria in the urine without the animal experiencing significant ill effects. This relationship between the animal and the bacteria is known as commensal relationship and the animal is known as a reservoir host.[19]
Humans are the accidental host for leptospirosis.[27] The bacteria enters the human body through breaches in the skin or through the mucous membrane, then into the bloodstream. The bacteria later attach to the endothelial cells of the blood vessels and extracellular matrix (complex network of proteins and carbohydrates that present between the cells). The flagella of the bacteria help it move between cell layers. Leptospira binds to a variety of cells such as fibroblasts, macrophages, endothelial cells, and kidney epithelial cells.[14] Additionally, surface leptospiral immunoglobulin-like (Lig) proteins such as LigB, whose gene is found in all pathogenic Leptospira species, help Leptospira bind to several human proteins such as complement proteins, thrombin, fibrinogen, and plasminogen. Other bacterial surface proteins that helps to bind to human proteins are LipL32 and Len A.[14]
Through innate immune system, endothelial cells of the capillaries in the human body are activated by the presence of these bacteria. The endothelial cells produce cytokines and antimicrobial peptides against the bacteria. These products regulate the coagulation cascade and movements of white blood cells.[14] Macrophages presented in humans are able to engulf Leptospira. However, Leptospira are able to reside and proliferate in the cytoplasmic matrix after being ingested by human macrophages, which later results in the death of the macrophages.[27] Those with severe leptospirosis can experience a high level of cytokines such as Interleukin 6, Tumor necrosis factor alpha (TNF alpha), and Interleukin 10. The high level of cytokines causes sepsis-like symptoms which is life-threatening instead of helping to fight against the infection. Those who have high risk of sepsis during a leptospirosis infection are found to have the HLA-DQ6 genotype, possibly due to superantigen activation, which damages bodily organs.[19]
Humoral immunity is the main immune response against the Leptospira cells. Agglutinating antibodies such as Immunoglobulin M and Immunoglobulin G antibodies are produced against the bacteria. Such antibodies are mainly directed against the LPS.[27] Leptospira’s LPS only activates toll-like receptor 2 (TLR2) in monocytes in humans. The Lipid A molecule of the bacteria is not recognized by human TLR4 receptors. Therefore, the lack of Leptospira recognition by TLR4 receptors probably contributes to leptospirosis disease process in humans.[27]
Although there are various mechanisms in the human body to fight against the bacteria, the Leptospira is well adapted to such inflammatory condition created by the human body.[14] In the bloodstream, Leptospira can activate host plasminogen to become plasmin that breaks down extracellular matrix, degrades fibrin clots and complemental proteins (C3b and C5) to avoid opsonisation. The bacteria can also recruit complement regulators such as Factor H, C4b-binding protein, factor H-like binding protein, and vitronectin to prevent the activation of membrane attack complex on its surface. Leptospira also secretes proteases to degrade complement proteins such as C3. Leptospira can bind to thrombin that decreases the fibrin formation. Reduced fibrin formation increases the risk of bleeding.[14] Specifically, the L. interrogans serovar Pomona has the SphA gene, which codes for Sphingomyelinase C that break down red blood cells. L. interrogans serovar Lai has the SphH gene, which codes for pore-forming protein and the sph2 gene where both of them damage the membranes of red blood cells.[27]
Leptospira spreads rapidly to all organs through the bloodstream. The bacteria are subsequently cleared from the human body except in the kidneys where they persist.[14] Leptospira mainly affects the liver. Congested liver sinusoids and perisinusoidal spaces have been reported. Leptospira tend to invade spaces between hepatocytes. Hepatocyte apoptosis has also been reported. The damaged hepatocytes and hepatocyte intercellular junctions cause leakage of bile into blood, resulting in elevated levels of bilirubin in those with jaundice. Meanwhile, in the lungs, petechiae or frank bleeding can be found at alveolar septum and spaces between alveoli.[19] Leptospira can cause mild to severe kidney failure. This is possibly due to reduced expression of Sodium–hydrogen antiporter 3 at the proximal renal tubule, causing reduced water absorption and increased urinary excretion. In humans, TLR2 detection of Leptospira causes inflammation of the kidney in the first two weeks of infection which results in interstitial nephritis. The kidney failure can recover completely or ended up as atrophy and fibrosis.[19] Rarely, inflammation of the heart muscles, coronary arteries, and aorta are found.[23]
Diagnosis
Lab tests
For those who are infected, a complete blood count may show high white cell count and low platelet count. When low hemoglobin count is present together with low white cell count and thromobocytopenia, bone marrow suppression should be considered.[19] Erythrocyte sedimentation rate and C-reactive protein may also be elevated.[21]
The kidneys are commonly involved in leptospirosis. Blood urea and creatinine levels will be elevated although the urine output is normal or high. Leptospirosis causes increased potassium excretion in the urine, which leads to low potassium level in the blood[19] and low sodium level.[21] Thus, those infected who have poor oral intake and high urine output will often have severe dehydration which increases the risk of death.[19] Urine analysis may reveal the presence of protein, presence of white blood cells, and microscopic haematuria.[21] The bacteria then move into kidneys after 10 days. Therefore, a urine culture will remain positive for leptospirosis from 10 days until 30 days of infection.[21]
For those with liver involvement, mild elevations of transaminases and direct bilirubin can be observed in liver function tests. The Leptospira Icterohaemorrhagiae serogroup is most commonly associated with jaundice and elevated bilirubin levels. In those with glucose-6-phosphate dehydrogenase deficiency, leptospirosis can contribute to acute hemolytic anaemia and conjugated hyperbilirubinemia.[19] Abnormal serum amylase and lipase levels (associated with pancreatitis) can be seen in those who are admitted into a hospital due to leptospirosis. Impaired kidney function with creatinine clearance less than 50 ml/min is associated with elevated pancreatic enzymes.[19]
For those with severe headache that shows signs of meningitis, a lumbar puncture can be attempted. If infected, cerebrospinal fluid (CSF) examination shows lymphocytic predominance with a cell count of 500/mm3, protein between 50 and 100 mg/ml, and normal glucose levels. These findings are consistent with aseptic meningitis.[19]
Serological tests
Rapid detection of Leptospira can be done by detecting the IgM antibodies using ELISA. Typically, isolates of L. biflexa antigen are used to detect the IgM antibodies. This test can quickly determine the diagnosis and help in early treatment of leptospirosis. However, the test specificity depends upon the type of antigen used, the presence of antibodies from previous infections, and the presence of other diseases such as Epstein-Barr virus infection, viral hepatitis, and cytomegalovirus infection can cause false positive results.[19] Other rapid screening test have been developed such as dipsticks, latex and slide agglutination tests.[21]
The Microscopic Agglutination Test (MAT) is the reference test for the diagnosis of leptospirosis.[19] MAT is a test where human or animal serum is mixed with various types of Leptospira antigens serovars. The mixture is then examined under a microscope to look for agglutination. The MAT is then read by dark field microscopy. The highest dilution where 50% agglutination occurs is the end result.[19] MAT titres of 1:100 to 1:800 are diagnostic of leptospirosis.[21] A fourfold or greater rise in titre of two sera taken at symptoms onset and at 3 to 10 days of disease onset confirms the diagnosis. During the acute phase of the disease, MAT is not specific in detecting a serotype of Leptospira because of cross-reactivity between the serovars.[19] In the convalescent phase, MAT is more specific in detecting the serovar types.[19] MAT requires a panel of live antigens and requires laborious work.[23]
Imaging
In those who have lung involvement, a chest X-ray may demonstrate diffuse alveolar opacities.[19] Both lungs are usually involved, with infection more commonly at the bases and periphery of the lungs, which can be present as early as 24 hours of the infection. Other patterns such as interstitial lung disease, pleural effusion, and ground-glass opacity of the lungs are uncommon.[43]
Diagnostic criteria
In 1982, World Health Organisation (WHO) proposed the Faine's criteria for the diagnosis of leptospirosis. It consists of three parts:A (clinical findings), B (epidemiological factors), and C (lab findings and bacteriological data). Since the original Faine's criteria only included culture and MAT in part C which is difficult and complex to perform; the modified Faine criteria was proposed in 2004 to include ELISA and slide agglutination tests which are easier to perform. In 2012, Modified Faine’s Criteria (with Amendment) was proposed to include shortness of breath and coughing up blood in the diagnosis. In 2013, India recommended modified Faine's criteria in the diagnosis of leptospirosis.[44]
| Modified Faine’s criteria (with amendment) 2012 | |||||||
| Part A: Clinical data | Score | Part B: Epidemiological factors | Score | Part C: Bacteriological and laboratory Findings | Score | ||
|---|---|---|---|---|---|---|---|
| Headache | 2 | Rainfall | 5 | Isolation of leptospira in culture | Diagnosis certain | ||
| Fever | 2 | Contact with contaminated environment | 4 | Polymerase chain reaction | 25 | ||
| Fever >39 °C | 2 | Animal contact | 1 | ELISA IgM positive | 15 | ||
| Conjunctival suffusion | 4 | Slide agglutination test positive | 15 | ||||
| Meningism | 4 | Other rapid tests | 15 | ||||
| Myalgia | 4 | MAT – single positive in high titer | 15 | ||||
| Conjuctival suffusion + Meningism + Myalgia | 10 | MAT – Rising titer / seroconversion (paired sera) | 25 | ||||
| Jaundice | 1 | ||||||
| Albuminuria / Nitrogen retention | 2 | ||||||
| Haemoptysis/ dyspnoea | 2 | ||||||
Presumptive diagnosis of leptospirosis is made when:[44]
- Part A or Part A & Part B score : 26 or more
- Part A, B, C (Total) : 25 or more
- A score between 20 and 25 suggests leptospirosis as a possible diagnosis.
Others
Leptospiral DNA can be amplified by using polymerase chain reaction (PCR) from serum, urine, aqueous humour, CSF, and autopsy specimens.[19] PCR can detect the presence of leptospiral DNA in blood even after the initiation of antibiotics.[21] It can also detect the presence of the bacteria without the appearance of antibodies during the first few days of the infection.[23] During the first eight days of infection, the bacteria can be detected by quantitative PCR and can reach as high is 106 bacteria per ml of blood.[19] The inability of human TLR4 to recognise the Leptospires allows the bacterial titres to be significantly higher when compared to Enterobacteriaceae infections.[19] Although PCR can detect more cases when compared to culture, it cannot detect the specific serotype of Leptospira, thus affecting its value in epidemiological studies.[19] MAT subsequently replaces PCR in detecting Leptospira infection.[19]
Molecular typing such as pulsed-field gel electrophoresis, multilocus sequence typing, multiple loci VNTR analysis can identify Leptospira to species level by analyzing 16S ribosomal RNA gene of isolates.[23]
Leptospira is a slow-growing bacteria. Blood samples containing Leptospira can be cultured in both liquid and solid media with nutrients such as long-chain fatty acids, vitamin B1 and B12, and ammonium salts.[27] The most common medium used is Ellinghausen-McCullough-Johnson-Harris medium (EMJH), which contains oleic acid, bovine serum albumin, and polysorbate,[27] incubated at 28 to 30 °C.[45] Other samples that can be cultured are: CSF and peritoneal washings during the first week of infection, and urine samples from the second week of infection. However, since survival of Leptospira is limited in urine, a phosphate buffered saline is used to enhance the bacteria growth in urine culture. Since contamination is prevalent in urine cultures, antibiotics such as fluorouracil are used to inhibit the growth of other bacteria in culture. The cultures are examined weekly under a dark field microscope until 13 weeks of incubation.[19]
Prevention

The incidence of leptospirosis cases can be reduced by improving housing, infrastructure, and sanitation standards. Rodent abatement efforts and flood mitigation projects can also help to prevent leptospirosis.[19] Proper use of personal protective equipment (PPE) by people who have a high risk of occupational exposure can prevent leptospirosis infections in most cases.[19] Effective rat control and avoidance of urine contaminated water sources are essential preventive measures.[6]
Human vaccines are available only in a few countries, such as Cuba, China[6] and France. Both human and animal vaccines only cover a few strains of the bacteria.[46] Side effects such as nausea, injection site redness and swelling have been reported after the vaccine was injected. Since the immunity induced by one serovar of Leptospirosis is only protective against that specific serovar of leptospirosis, trivalent vaccines have been developed.[19] However, leptospiral vaccines do not confer long lasting immunity to humans or animals.[14] Dog vaccines are effective for at least one year.[46]
Doxycycline has been provided once a week as a prophylaxis to minimize infections during outbreaks in endemic regions.[47] Doxycycline was found to be effective in reducing the number of leptospirosis cases in military personnel undergoing exercises in the jungles. In another study, doxycycline administered after exposure to antibiotics was found reduce the number of people with symptomatic disease after a heavy rainfall in endemic areas.[19] However, there is no strong evidence that chemoprophylaxis is effective in containing outbreaks of leptospirosis,[48] and use of antibiotics increases antibiotic resistance. Pre-exposure prophylaxis may be beneficial for individuals traveling to high-risk areas for a short stay.[49]
Treatment
Most leptospiral cases resolve spontaneously. Early initiation of antibiotics may prevent the progression to severe disease. Therefore, in resource limited settings, antibiotics can be started once leptospirosis is suspected in history taking and examination.[19]
For mild leptospirosis, antibiotic recommendations such as doxycycline, azithromycin, ampillicin and amoxicillin were solely based on in vitro testing.[21] In 2001, the World Health Organization published a report which recommended oral doxycycline (2 mg/kg up to 100 mg 12 hourly) for 5 to 7 days for those with mild leptospirosis. Tetracycline, ampicillin, and amoxicillin can also be used in such cases.[50] However, in areas where rickettsia and leptospirosis are both endemic, either azithromycin or doxycycline are the drugs of choice.[21]
Based on a 1988 study, intravenous (IV) benzylpenicillin (also known as penicillin G) is recommended for the treatment of severe leptospirosis.[21] Intravenous benzylpenicillin (30 mg/kg up to 1.2g 6 hourly) is used for 5 to 7 days. Amoxicillin, ampicillin, and erythromycin may also be used for severe cases.[50] Ceftriaxone (1g IV every 24 hours for 7 days) and penicillin G (1.5 million units IV every six hours for seven days) have been shown to have similar efficacy in treating severe leptospirosis.[19][21][51] In another study on severe leptospirosis conducted in 2004, both cefotaxime (1g IV every six hours for seven days) and doxycycline (200 mg initially followed by 100 mg IV every 12 hours for seven days) showed similar efficacy when compared to penicillin G (1.5 million units IV every six hours for seven days).[21][52] Therefore, there is no evidence on risk of death reduction when comparing the usage of intravenous benzylpenicillin with ceftriaxone or cefotaxime.[21] Another study conducted in 2007 also showed no difference in efficacy between doxycycline (200 mg initially followed by 100 mg orally every 12 hours for seven days) or azithromycin (2 g on day 1 followed by 1 g daily for two more days) for suspected leptospirosis. There was no difference in the resolution of fever and azithromycin is better tolerated than doxycycline.[53][54][55]
Those who can be treated as outpatients should receive doxycycline or azithromycin. Doxycycline can shorten the duration of leptospirosis by two days, improve symptoms, and prevent the shedding of organisms in urine. Azithromycin and amoxicillin are given to pregnant women and children.[19] Rarely, a Jarisch–Herxheimer reaction can develop in the first few hours after antibiotic administration.[21] However, according to a meta-analysis done in 2012, the benefit of antibiotics in the treatment of leptospirosis was unclear although the use of antibiotics may reduce the duration of illness by two to four days.[21][54] Another meta-analysis done in 2013 reached a similar conclusion.[21][55]
For those with severe leptospirosis, such as potassium wasting with high kidney output dysfunction, intravenous hydration and potassium supplements should be given to prevent dehydration and hypokalemia. When acute kidney failure occurs, early initiation of haemodialysis or peritoneal dialysis can help to improve survival. For those with respiratory failure, tracheal intubation with low tidal volume improves survival rates.[19]
The use of corticosteroids has been proposed for the treatment of leptospirosis because Leptospira infection can induce the release of chemical signals which promote inflammation of blood vessels in the lungs. However, there is insufficient evidence to determine whether the use of corticosteroids is beneficial.[21][56]
Prognosis
The overall risk of death for leptospirosis is 5% to 10%.[27] For those with jaundice, the case fatality can increase to 5% to 15%.[25] For those infected presented with confusion and neurological signs, they have a high risk of death.[19] Other factors that increase the risk of death include: reduced urine output, age more than 36 years, and respiratory failure.[19] With proper care, most of those infected will recover completely. Those with acute kidney failure may suffer persistent mild kidney impairment post recovery.[19] In those with severe lung involvement, the risk of death is 50 to 70%.[21]
Thirty percent of affected people may suffer chronic leptospirosis syndrome for up to two years which is characterized by weakness, muscle pain, and headaches.[19] Eye problems can occur in 10% of those who recovered from leptospirosis.[25] These complications can range from mild anterior uveitis to severe panuveitis (which involves all the three vascular layers of the eye) post recovery. In up to 80% of those infected, leptospira DNA can be found in the aqueous humour of the eye.[19] The eye problems usually have good prognosis when being treated or they are self-limiting.[25]
Epidemiology
It is estimated that seven to ten million people are infected by leptospirosis annually.[12] One million cases of severe leptospirosis occur annually, with 58,900 deaths.[13] Leptospirosis is found in both urban and rural areas in tropical, subtropical, and temperate regions. The risk of death is 5 to 10%.[27]
Annual rates of infection vary from 0.1 to 1 per 100,000 in temperate climates to 10 to 100 per 100,000 in tropical climates.[47][57] The disease is persistently observed in South and Southeast Asia, Oceania, Caribbean,[27] Latin America and Africa.[25] Antarctica is the only place not affected by leptospirosis.[25] In the United States, there were 100 to 150 leptospirosis cases annually.[58] In 1994, leptospirosis ceased to be a notifiable disease in the United States except in 36 territories where leptospirosis are prevalent such as Hawaii, Texas, California, and Puerto Rico.[59] About 50% of the cases occurred in Puerto Rico. In January 2013, leptosprirosis was reinstated as a nationally notifiable disease in the United States.[58]
The number of new cases of leptospirosis is difficult to estimate since many cases are not reported. There are many reasons for this, but the biggest issue is differentiating the disease from other conditions with a similar presentation.[57] Laboratory testing is lacking in many areas.[57] The global incidence of leptospirosis has been underestimated because most affected countries lack notification or notification is not mandatory.[19] In the context of global epidemiology, the socioeconomic status of many of the world's population is closely tied to malnutrition; subsequent lack of micronutrients may lead to increased risk of infection and death due to leptospirosis infection.[60] Micronutrients such as iron, calcium, and magnesium represent important areas of future research.[60]
The number of cases increases during the rainy season in the tropics and during the late summer or early fall in Western countries. This happens because leptospires survive best in fresh water, damp alkaline soil, vegetation, and mud with temperatures higher than 22 °C.[61] This also leads to increased risk of exposure to populations during flood conditions, and leptospire concentrations to peak in isolated pools during drought. There is no evidence of leptospirosis having any effect on sexual and age-related differences.[61] However, a major risk factor for development of the disease is occupational exposure, a disproportionate number of working-aged males are affected.[61] There have been reported outbreaks where more than 40% of people are younger than 15.[61] Outbreaks that occurred after the 1940s in the United States have happened mostly in the late summer seasons, which happens to be the driest part of the year. The people at the highest risk for leptospirosis are young people whose age ranges from 5–16 years old, and can also range to young adults.[62]
History
The disease was first described by Adolf Weil in 1886 when he reported an "acute infectious disease with enlargement of spleen, jaundice, and nephritis."[16] Even before Weil's description, the disease was known as "rice field jaundice" in ancient Chinese text, "autumn fever", "seven-day fever",[63] and "nanukayami fever"[64] in Japan; in Europe and Australia, the disease was associated with certain occupations with names such as "cane-cutter's disease", "swine-herd's disease", and "Sclammfieber" (mud fever).[63] It has historically been known as "black jaundice",[65] or "dairy farm fever" in New Zealand.[66] Leptospirosis was postulated as the cause of an epidemic among Native Americans along the coast of what is now New England during 1616-19. The disease was most likely brought to the New World by Europeans.[67]
Leptospira was first observed in 1907 from a post mortem kidney tissue slice by Stimson using silver deposition staining technique. He called the organism Spirocheta interrogans because the bacteria resembles a question mark.[63][68] In 1908, a Japanese research group led by Inada and Ito first identified this bacterium as the causative agent of leptospirosis[69] and in 1916 noted its presence in rats.[70] Leptospirosis were frequently contracted by coal mine workers in Japan. The organism was named Spirocheta icterohaemorrhagiae. The Japanese group also experimented with the first leptospiral immunisation studies in guinea pigs. They demonstrated that by injecting the infected guinea pigs with sera from convalescent humans or goats, passive immunity can be provided to the guinea pigs. In 1917, the Japanese group discovered rats as the carriers of leptospirosis.[63] Unaware of the Japanese group's work, two German groups independently and almost simultaneously published their first demonstration of transmitting leptospiral infection in guinea pigs in October 1915. They named the organism Spirochaeta nodosa and Spirochaeta Icterogenes respectively.[63]
Leptospirosis was subsequently recognised as a disease of all mammalian species. In 1933, Dutch workers reported the isolation of Leptospira canicola which specifically infects dogs. In 1940, the strain that specifically infects cattle was first reported in Russia.[63] In 1942, soldiers at Fort Bragg, North Carolina were recorded to have an infectious disease which caused rash over their shinbones. This disease was later known to be caused by leptospirosis.[19] By the 1950s, the number of serovars that infects various mammals had expanded significantly. In the 1980s, leptospirosis was recognised as a veterinary disease of major economic importance.[63]
In 1982, there were about 200 serovars of Leptospira available for classification. The International Committee on Systematic Bacteriology's subcommittee on Taxonomy of Leptospira proposed classifying these serovars into two big groups: L. interrogans containing pathogenic serovars and L. biflexa containing saprohytic serovars.[63] In 1979, the leptospiral family of Leptospiraceae was proposed. In the same year, Leptospira illini was reclassified as the new genus Leptonema.[63] In 2002, "Lepthangamushi syndrome" was coined to describe a series of overlapping symptoms of leptospirosis with Hantavirus hemorrhagic fever with renal syndrome, and scrub typhus caused by Orientia tsutsugamushi.[71][72] In 2005, Leptospira parva was classified as Turneriella.[63] With DNA-DNA hybridisation technology, L. interrogans was divided into seven species. More Leptospira species have been discovered since then.[63] The WHO established the Leptospirosis Burden Epidemiology Reference Group (LERG) to review the latest disease epidemiological data of leptospirosis, formulate a disease transmission model, and identify gaps in knowledge and research. The first meeting was convened in 2009. In 2011, LERG estimated that the global annual incidence of leptospirosis is 5 to 14 cases per 100,000 population.[19]
Other animals
Animals are also presented with similar clinical features when compared to humans. Incubation (time of exposure to first symptoms) in animals is anywhere from 2 to 20 days.[73] Clinical signs can appear in 5 to 15 days in dogs. Incubation period can be prolonged in cats. Leptospirosis can cause abortions after 2 to 12 weeks in cattle, and 1 to 4 weeks of infection in pigs. The illness tends to be milder in reservoir hosts. The most commonly affected organs are the kidneys, liver, and reproductive system but other organs can be affected.[25] In dogs, the acute clinical signs include fever, loss of appetite, shivering, muscle pain, weakness, and urinary symptoms. Vomiting, diarrhea, and abdominal pain may also present. Petechiae and ecchymoses may be seen on mucous membranes. Bleeding from the lungs may also be seen in dogs. In chronic presentations, the affected dog may have no symptoms. In animals that have died of leptospirosis, their kidneys may be swollen with grey and white spots, mottling, or scarring. Their liver may be enlarged with areas of cell death. Petechiae and ecchymoses may be found in various organs.[25][73][74] Inflammation of the blood vessels may occur, causing swelling and potentially disseminated intravascular coagulation. Inflammation of the heart, pericardial sac around the heart, meningeal layers covering the brain and spinal cord, and uveitis are also possible.[9] Risk of death or disability in animals varies depending upon the species and age of the animals. In adult pigs and cattle, reproductive signs are the most common signs of leptospirosis. Up to 40% of the cows may have a spontaneous abortion. Younger animals usually develop more severe disease. 80% of dogs can survive with treatment but the survival rate is reduced if the lungs are involved.[25] At least five important serovars exist in the United States and Canada, all of which cause disease in dogs: Icterohaemorrhagiae, Canicola, Pomona, Grippotyphosa, and Bratislava.[9][73][74]
ELISA and Microscopic Agglutination Tests are most commonly used to diagnose leptospirosis in animals. The bacteria can be detected in blood, urine, and milk or liver, kidney, or other tissue samples by using immunofluorescence or immunohistochemical or polymerase chain reaction techniques. The organisms stain poorly with gram stain, therefore, silver staining or immunogold silver staining are used. Dark field microscopy can be used to detect Leptospira but it is neither sensitive nor specific in detecting the organism.[25] A positive culture for leptospirosis is definitive but the availability is limited and culture results can take 13 to 26 weeks for a result, limiting its utility.[25] Paired acute and convalescent samples are preferred for serological diagnosis of leptospirosis in animals. However, a positive serological sample from an aborted fetus is diagnostic of leptospirosis.[25]
Various antibiotics such as doxycycline, penicillins, dihydrostreptomycin, and streptomycin have been used to treat leptospirosis in animals. Fluid therapy, blood transfusion, and respiratory support may be required in severe disease.[25] For horses, the primary treatment is anti-inflammatory drugs.[25]
Leptospirosis vaccines are available for animals such as pigs, dogs, cattle, sheep, and goats. Vaccines for cattle usually contain Leptospira serovar Hardjo and Pomona, for dogs, the vaccines usually contain serovar Icterohaemorrhagiae and Canicola.[25] Isolation of infected animals and prophylactic antibiotics are also effective in preventing leptospirosis transmission between animals. Environmental control and sanitation also reduce transmission rates.[25]
References
- Berger, Stephen (2018). Leptospirosis: Global Status. GIDEON Informatics Inc. p. 7. ISBN 9781498820318.
- Mosby's Medical Dictionary (9 ed.). Elsevier Health Sciences. 2013. p. 697. ISBN 9780323112581. Archived from the original on 8 September 2017.
- McKay, James E. (2001). Comprehensive health care for dogs. Minnetonka, MN.: Creative Pub. International. p. 97. ISBN 9781559717830.
- James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 978-0-7216-2921-6.:290
- Slack, A (July 2010). "Leptospirosis". Australian Family Physician. 39 (7): 495–8. PMID 20628664.
- McBride, AJ; Athanazio, DA; Reis, MG; Ko, AI (October 2005). "Leptospirosis". Current Opinion in Infectious Diseases. 18 (5): 376–86. doi:10.1097/01.qco.0000178824.05715.2c. PMID 16148523.
- Picardeau M, Bertherat E, Jancloes M, Skouloudis AN, Durski K, Hartskeerl RA (January 2014). "Rapid tests for diagnosis of leptospirosis: current tools and emerging technologies". Diagnostic Microbiology and Infectious Disease. 78 (1): 1–8. doi:10.1016/j.diagmicrobio.2013.09.012. PMID 24207075.
- Wasiński B, Dutkiewicz J (2013). "Leptospirosis—current risk factors connected with human activity and the environment". Annals of Agricultural and Environmental Medicine. 20 (2): 239–44. PMID 23772568. Archived from the original on 14 September 2014.
- Langston CE, Heuter KJ (July 2003). "Leptospirosis. A re-emerging zoonotic disease". Veterinary Clinics of North America: Small Animal Practice. 33 (4): 791–807. doi:10.1016/S0195-5616(03)00026-3. PMID 12910744.
- Picardeau M (January 2013). "Diagnosis and epidemiology of leptospirosis". Médecine et Maladies Infectieuses. 43 (1): 1–9. doi:10.1016/j.medmal.2012.11.005. PMID 23337900.
- Farrar, Jeremy; Hotez, Peter; Junghanss, Thomas; Kang, Gagandeep; Lalloo, David; White, Nicholas J. (2013). Manson's Tropical Diseases E-Book. Elsevier Health Sciences. p. 438. ISBN 9780702053061. Archived from the original on 8 September 2017.
- "Leptospirosis". NHS. 11 July 2012. Archived from the original on 14 March 2014. Retrieved 14 March 2014.
- Costa F, Hagan JE, Calcagno J, Kane M, Torgerson P, Martinez-Silveira MS, Stein C, Abela-Ridder B, Ko AI (2015). "Global Morbidity and Mortality of Leptospirosis: A Systematic Review". PLOS Neglected Tropical Diseases. 9 (9): e0003898. doi:10.1371/journal.pntd.0003898. PMC 4574773. PMID 26379143.
- Picardeau M (May 2017). "Virulence of the zoonotic agent of leptospirosis: still terra incognita?". Nature Reviews. Microbiology. 15 (5): 297–307. doi:10.1038/nrmicro.2017.5. PMID 28260786.
- "Leptospirosis (Infection)". Centers for Disease Control and Prevention. Archived from the original on 11 October 2014. Retrieved 8 November 2014.
- Weil, A. (1886). "Über eine eigenthümliche, mit Milztumor, Icterus und Nephritis einhergehende, acute Infektionskrankheit" [On a strange, acute infectious disease, accompanied by swelling of the spleen, icterus, and nephritis]. Deutsches Archiv für Klinische Medizin (in German). 39: 209–232.
- "Leptospirosis" (PDF). The Center for Food Security and Public Health. October 2013. Archived (PDF) from the original on 24 November 2014. Retrieved 8 November 2014.
- Faine, Solly; Adler, Ben; Bolin, Carole (1999). "Clinical Leptospirosis in Animals". Leptospira and Leptospirosis (Revised 2nd ed.). Melbourne, Australia: MediSci. p. 113. ISBN 978-0-9586326-0-7.
- David, A Haake; Paul, N Levett (25 May 2015). Leptospirosis in Humans. Current Topics in Microbiology and Immunology. 387. pp. 65–97. doi:10.1007/978-3-662-45059-8_5. ISBN 978-3-662-45058-1. PMC 4442676. PMID 25388133.
- Levett PN (April 2001). "Leptospirosis". Clinical Microbiology Reviews. 14 (2): 296–326. doi:10.1128/CMR.14.2.296-326.2001. PMC 88975. PMID 11292640.
- Alison, B Lane; Michael, M Dore (25 November 2016). "Leptospirosis: A clinical review of evidence based diagnosis, treatment and prevention". World Journal of Clinical Infectious Diseases. 6 (4): 61–66. doi:10.5495/wjcid.v6.i4.61.
- Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Diaz MM, Lovett MA, Levett PN, Gilman RH, Willig MR, Gotuzzo E, Vinetz JM (December 2003). "Leptospirosis: a zoonotic disease of global importance". The Lancet Infectious Diseases. 3 (12): 757–71. doi:10.1016/s1473-3099(03)00830-2. PMID 14652202.
- Bennett, John E; Raphael, Dolin; Martin, J Blaser; Bart, J Currie (2015). "223". Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases (Eighth ed.). Elsevier. pp. 2541–2549. ISBN 978-1-4557-4801-3.
- Farr RW (July 1995). "Leptospirosis". Clinical Infectious Diseases. 21 (1): 1–6, quiz 7–8. doi:10.1093/clinids/21.1.1. PMID 7578715.
- Spickler, AR; Leedom, Larson KR (October 2013). "Leptospirosis (Fact sheet)" (PDF). The Center for food security and public health. Retrieved 15 March 2019. Cite journal requires
|journal=(help) - Ko AI, Goarant C, Picardeau M (October 2009). "Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen". Nature Reviews Microbiology. 7 (10): 736–47. doi:10.1038/nrmicro2208. PMC 3384523. PMID 19756012.
- Evangelista, KV; Coburn, J (September 2010). "Leptospira as an emerging pathogen: a review of its biology, pathogenesis and host immune responses". Future Microbiology. 5 (9): 1413–1425. doi:10.2217/fmb.10.102. PMC 3037011. PMID 20860485.
- Cameron CE (2015). "Leptospiral structure, physiology, and metabolism". Current Topics in Microbiology and Immunology. 387: 21–41. doi:10.1007/978-3-662-45059-8_3. ISBN 978-3-662-45058-1. PMID 25388131.
- Ahmad, SN; Shah, S; Ahmad, FMH (2005). "Laboratory Diagnosis of Leptospirosis". Journal of Postgraduate Medicine. 51 (3): 195–200. PMID 16333192.
- Cerqueira GM, Picardeau M (September 2009). "A century of Leptospira strain typing". Infection, Genetics and Evolution. 9 (5): 760–8. doi:10.1016/j.meegid.2009.06.009. PMID 19540362.
- Kiktenko VS; Balashov, NG; Rodina, VN (1976). "Leptospirosis infection through insemination of animals". Journal of Hygiene, Epidemiology, Microbiology, and Immunology. 21 (2): 207–213. PMID 987112.
- Barragan V, Olivas S, Keim P, Pearson T (October 2017). "Critical Knowledge Gaps in Our Understanding of Environmental Cycling and Transmission of Leptospira spp". Applied and Environmental Microbiology. 83 (19). doi:10.1128/AEM.01190-17. PMC 5601346. PMID 28754706.
- Pappachan MJ, Sheela M, Aravindan KP. Relation of rainfall pattern and epidemic leptospirosis in the Indian state of Kerala. J Epidemiol Community Health. 2004;58:1054.
- Guerra MA (February 2009). "Leptospirosis". Journal of the American Veterinary Medical Association. 234 (4): 472–8, 430. doi:10.2460/javma.234.4.472. PMID 19222355.
- Allan KJ, Biggs HM, Halliday JE, Kazwala RR, Maro VP, Cleaveland S, Crump JA (2015). "Epidemiology of Leptospirosis in Africa: A Systematic Review of a Neglected Zoonosis and a Paradigm for 'One Health' in Africa". PLOS Neglected Tropical Diseases. 9 (9): e0003899. doi:10.1371/journal.pntd.0003899. PMC 4569256. PMID 26368568.
- Shaw RD (June 1992). "Kayaking as a risk factor for leptospirosis". Missouri Medicine. 89 (6): 354–7. PMID 1620089.
- "Weil's Disease at Work". injury-compensation-zone.co.uk. Injury Compensation. Archived from the original on 3 December 2013.
- A.T.W, Song; L, Abas; L.C., Andrade; W, Andraus (15 December 2015). "A first report of leptospirosis after liver transplantation". Transplant Infectious Disease. 18 (1): 137–140. doi:10.1111/tid.12490. PMID 26671230.
- Puliyath, G; Singh, S (October 2012). "Leptospirosis in pregnancy". European Journal of Clinical Microbiology & Infectious Diseases. 31 (10): 2491–2496. doi:10.1007/s10096-012-1625-7. PMID 22549729.
- Carles, G; Montoya, E; Joly, F; Peneau, C (1995). "Leptospirosis and pregnancy. Eleven cases in French Guyana". Journal de Gynécologie Obstétrique et Biologie de la Reproduction. 24 (4): 418–421. PMID 7650320.
- Suan-Li, Liana Koe; Kim, Teng Tan; Thiam, Chye Tan (February 2014). "Leptospirosis in pregnancy with pathological fetal cardiotocography changes". Singapore Medical Journal. 55 (2): e20–e24. doi:10.11622/smedj.2013194. PMC 4291937. PMID 24712035.
- Shaked, Y; Shpilherg, O (1993). "Leptospirosis in pregnancy and its effect on the fetus: case report and review". Clinical Infectious Diseases. 17 (2): 241–243. doi:10.1093/clinids/17.2.241. PMID 8399874.
- Carlos, Roberto Ribeiro; Eduardo, P. Bethlem (June 2002). "Pulmonary complications of leptospirosis". Clinics in Chest Medicine. 23 (2): 469–478. doi:10.1016/S0272-5231(01)00010-7. PMID 12092040.
- Kumar, SS (2013). "7". Indian Guidelines for the Diagnosis and Management of Human Leptospirosis (PDF). India. pp. 23–29. Archived from the original (PDF) on 25 September 2016. Retrieved 16 November 2019.
- Rule PL, Alexander AD (1986). "Gellan gum as a substitute for agar in leptospiral media". Journal of Clinical Microbiology. 23 (3): 500–504. PMC 268682. PMID 3754265.
- Goldstein RE (November 2010). "Canine leptospirosis". Veterinary Clinics of North America: Small Animal Practice. 40 (6): 1091–101. doi:10.1016/j.cvsm.2010.07.008. PMID 20933138.
- Pavli A, Maltezou HC (2008). "Travel-acquired leptospirosis". Journal of Travel Medicine. 15 (6): 447–53. doi:10.1111/j.1708-8305.2008.00257.x. PMID 19090801.
- "Leptospirosis: an emerging public health problem". Weekly Epidemiological Record. 86 (6): 45–50. February 2011. PMID 21302385.
- Brett-Major DM, Lipnick RJ (2009). "Antibiotic prophylaxis for leptospirosis". Cochrane Database of Systematic Reviews (3): CD007342. doi:10.1002/14651858.CD007342.pub2. PMID 19588424.
- WHO recommended strategies for the prevention and control of communicable diseases. World Health Organization - Department of Communicable Disease Control, Prevention and Eradication. 2001. p. 104. Archived from the original on 5 May 2019.
- Panaphut, T; Domrongkitchaiporn, S; Vibhagool, A (June 2003). "Ceftriaxone compared with sodium penicillin g for treatment of severe leptospirosis". Clinical Infectious Diseases. 36 (12): 1507–1513. doi:10.1086/375226. PMID 12802748.
- Suputtamongkol, Y; Niwattayakul, K; Suttinont, C (November 2004). "An open, randomized, controlled trial of penicillin, doxycycline, and cefotaxime for patients with severe leptospirosis". Clinical Infectious Diseases. 39 (10): 1417–1424. doi:10.1086/425001. PMID 15546074.
- Phimda, K; Hoontrakul, S; Suttinont, C (September 2007). "Doxycycline versus azithromycin for treatment of leptospirosis and scrub typhus". Antimicrobial Agents and Chemotherapy. 51 (9): 3259–3263. doi:10.1128/AAC.00508-07. PMC 2043199. PMID 17638700.
- Brett-Major, DM; Coldren, R (15 February 2012). "Antibiotics for leptospirosis". The Cochrane Database of Systematic Reviews (2): CD008264. doi:10.1002/14651858.CD008264.pub2. PMID 22336839.
- Jaykaran, Charan; Deepak, Saxena; Summaiya, Mulla (May 2013). "Antibiotics for the Treatment of Leptospirosis: Systematic Review and Meta-Analysis of Controlled Trials". International Journal of Preventive Medicine. 4 (5): 501–510. PMC 3733179. PMID 23930159.
- Rodrigo, C; Lakshitha de Silva, N; Goonaratne, R (December 2014). "High dose corticosteroids in severe leptospirosis: a systematic review". Transactions of the Royal Society of Tropical Medicine and Hygiene. 108 (12): 743–750. doi:10.1093/trstmh/tru148. PMID 25266477.
- "WHO | Leptospirosis Burden Epidemiology Reference Group (LERG)". www.who.int. Retrieved 30 November 2017.
- "Healthcare Workers - Technical Information for Leptospirosis". Centers for Disease Control and Prevention (CDC). 9 November 2017. Archived from the original on 11 January 2019. Retrieved 28 April 2019.
- Marta A, Guerra (September 2013). "Leptospirosis: Public health perspectives". Biologicals. 41 (5): 295–297. doi:10.1016/j.biologicals.2013.06.010. PMC 4629849. PMID 23850378.
- Herman, Heather S.; Mehta, Saurabh; Cárdenas, Washington B.; Stewart-Ibarra, Anna M.; Finkelstein, Julia L. (7 July 2016). "Micronutrients and Leptospirosis: A Review of the Current Evidence". PLOS Neglected Tropical Diseases. 10 (7): e0004652. doi:10.1371/journal.pntd.0004652. ISSN 1935-2735. PMC 4936698. PMID 27387046.
- "Leptospirosis: Practice Essentials, Background, Pathophysiology". 17 November 2017. Cite journal requires
|journal=(help) - Galton, Mildred M. (February 1959). "The epidemiology of leptospirosis in the United States". Public Health Reports. 74 (2): 141–148. doi:10.2307/4590398. ISSN 0094-6214. JSTOR 4590398. PMC 1929197. PMID 13623988.
- Ben, Adler (2015). "Leptospira and Leptospirosis - History of Leptospirosis and Leptospira". Current Topics in Microbiology and Immunology. 387: 1–9. doi:10.1007/978-3-662-45059-8_1. PMID 25388129.
- Dorland's illustrated medical dictionary (32nd ed.). Philadelphia: Elsevier/Saunders. 2012. p. 1231. ISBN 9781455709854. Archived from the original on 8 September 2017.
- David Clapham (2004). Small Water Supplies: A Practical Guide. Routledge. p. 125. ISBN 9781134457496. Archived from the original on 8 September 2017.
- Christmas, BW; Tennent, RB; Lindsay, PG (1974). "Dairy farm fever in New Zealand: a local outbreak of human leptospirosis". New Zealand Medical Journal. 79 (514): 901–904. PMID 4527727.
- Marr JS, Cathey JT (2010). "New hypothesis for cause of epidemic among native Americans, New England, 1616-1619". Emerging Infect. Dis. 16 (2): 281–286. doi:10.3201/eid1602.090276. PMC 2957993. PMID 20113559.
- Stimson, A.M. (1907). "Note on an organism found in yellow-fever tissue". Public Health Reports. 22 (18): 541. doi:10.2307/4559008. JSTOR 4559008.
- Inada R, Ito Y (1908). "A report of the discovery of the causal organism (a new species of spirocheta) of Weil's disease". Tokyo Ijishinshi. 1915: 351–60.
- Inanda R, Ido Y, Hoke R, Kaneko R, Ito H (1916). "The etiology, mode of infection and specific therapy of Weil's disease". The Journal of Experimental Medicine. 23 (3): 377–402. doi:10.1084/jem.23.3.377. PMC 2125418. PMID 19867994.
- Alberto, EPM; Alfonso, JRM; Gabriela, B (22 July 2016). "ChikDenMaZika Syndrome: the challenge of diagnosing arboviral infections in the midst of concurrent epidemics". Annals of Clinical Microbiology and Antimicrobials. 15: 42. doi:10.1186/s12941-016-0157-x. PMC 4957883.
- "284184004: Lepthangamushi syndrome (disorder)". Archived from the original on 18 November 2019. Retrieved 18 November 2019.
- Klopfleisch R, Kohn B, Plog S, Weingart C, Nöckler K, Mayer-Scholl A, Gruber AD (2011). "An Emerging Pulmonary Haemorrhagic Syndrome in Dogs: Similar to the Human Leptospiral Pulmonary Haemorrhagic Syndrome?". Veterinary Medicine International. 33: 928541. doi:10.4061/2010/928541. PMC 3025382. PMID 21274452.
- Kohn B, Steinicke K, Arndt G, Gruber AD, Guerra B, Jansen A, Kaser-Hotz B, Klopfleisch R, Lotz F, Luge E, Nöckler K (2010). "Pulmonary abnormalities in dogs with leptospirosis". Journal of Veterinary Internal Medicine. 24 (6): 1277–1282. doi:10.1111/j.1939-1676.2010.0585.x. PMID 20738768.
External links
| Classification | |
|---|---|
| External resources |
- "Leptospirosis". U.S. Disease Control and Prevention Center. 21 November 2018.
- "Leptospira". NCBI Taxonomy Browser. 171.