Leishmaniasis
Leishmaniasis is a disease caused by parasites of the Leishmania type.[2] It is spread by the bite of certain types of sandflies.[2] The disease can present in three main ways: cutaneous, mucocutaneous, or visceral.[2] The cutaneous form presents with skin ulcers, while the mucocutaneous form presents with ulcers of the skin, mouth, and nose, and the visceral form starts with skin ulcers and then later presents with fever, low red blood cells, and enlarged spleen and liver.[2][3]
| Leishmaniasis | |
|---|---|
| Other names | Leishmaniosis |
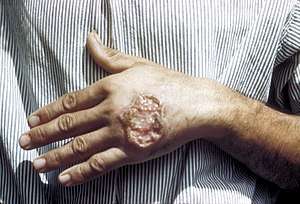 | |
| Cutaneous leishmaniasis in the hand of a Central American adult | |
| Pronunciation |
|
| Specialty | Infectious disease |
| Symptoms | Skin ulcers, fever, low red blood cells, enlarged liver[2][3] |
| Causes | Leishmania parasites spread by sandflies[2] |
| Prevention | Bug nets, insecticide[2] |
| Frequency | 4–12 million[4][5] |
| Deaths | 24,200 (2015)[6] |
Infections in humans are caused by more than 20 species of Leishmania.[2] Risk factors include poverty, malnutrition, deforestation, and urbanization.[2] All three types can be diagnosed by seeing the parasites under the microscope.[2] Additionally, visceral disease can be diagnosed by blood tests.[3]
Leishmaniasis can be partly prevented by sleeping under nets treated with insecticide.[2] Other measures include spraying insecticides to kill sandflies and treating people with the disease early to prevent further spread.[2] The treatment needed is determined by where the disease is acquired, the species of Leishmania, and the type of infection.[2] Some possible medications used for visceral disease include liposomal amphotericin B,[7] a combination of pentavalent antimonials and paromomycin,[7] and miltefosine.[8] For cutaneous disease, paromomycin, fluconazole, or pentamidine may be effective.[9]
About 4 to 12 million people are currently infected[4][5] in some 98 countries.[3] About 2 million new cases[3] and between 20 and 50 thousand deaths occur each year.[2][10] About 200 million people in Asia, Africa, South and Central America, and southern Europe live in areas where the disease is common.[3][11] The World Health Organization has obtained discounts on some medications to treat the disease.[3] It is classified as a neglected tropical disease.[12] The disease may occur in a number of other animals, including dogs and rodents.[2]
Signs and symptoms
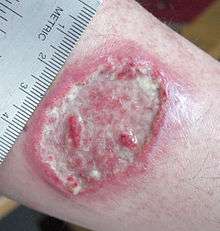
The symptoms of leishmaniasis are skin sores which erupt weeks to months after the person is bitten by infected sand flies.
Leishmaniasis may be divided into the following types:[13]
- Cutaneous leishmaniasis is the most common form, which causes an open sore at the bite sites, which heals in a few months to a year and half, leaving an unpleasant-looking scar.[2][3] Diffuse cutaneous leishmaniasis produces widespread skin lesions which resemble leprosy, and may not heal on its own.[3]
- Mucocutaneous leishmaniasis causes both skin and mucosal ulcers with damage primarily of the nose and mouth.[2][3]
- Visceral leishmaniasis or kala-azar ('black fever') is the most serious form, and is potentially fatal if untreated.[2] Other consequences, which can occur a few months to years after infection, include fever, damage to the spleen and liver, and anemia.[2]
Leishmaniasis is considered one of the classic causes of a markedly enlarged (and therefore palpable) spleen; the organ, which is not normally felt during examination of the abdomen, may even become larger than the liver in severe cases.
Cause
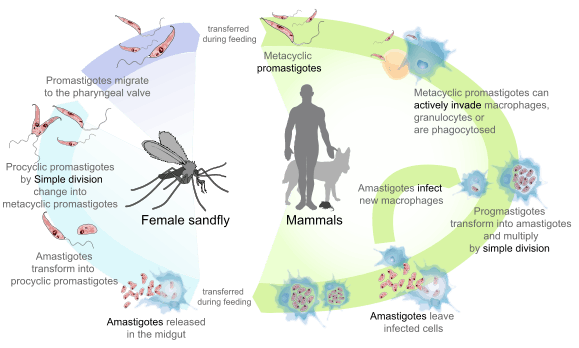
Leishmaniasis is transmitted by the bite of infected female phlebotomine sandflies[2] which can transmit the protozoa Leishmania.[2] (1) The sandflies inject the infective stage, metacyclic promastigotes, during blood meals. (2) Metacyclic promastigotes that reach the puncture wound are phagocytized by macrophages, and (3) transform into amastigotes. (4) Amastigotes multiply in infected cells and affect different tissues, depending in part on the host, and in part on which Leishmania species is involved. These differing tissue specificities cause the differing clinical manifestations of the various forms of leishmaniasis. (5,6) Sandflies become infected during blood meals on infected hosts when they ingest macrophages infected with amastigotes. (7) In the sandfly's midgut, the parasites differentiate into promastigotes, (8) which multiply, differentiate into metacyclic promastigotes, and migrate to the proboscis.
The genomes of three Leishmania species (L. major, L. infantum, and L. braziliensis) have been sequenced, and this has provided much information about the biology of the parasite. For example, in Leishmania, protein-coding genes are understood to be organized as large polycistronic units in a head-to-head or tail-to-tail manner; RNA polymerase II transcribes long polycistronic messages in the absence of defined RNA pol II promoters, and Leishmania has unique features with respect to the regulation of gene expression in response to changes in the environment. The new knowledge from these studies may help identify new targets for urgently needed drugs and aid the development of vaccines.[14]
Vector
Although most of the literature mentions only one genus transmitting Leishmania to humans (Lutzomyia) in the New World, a 2003 study by Galati suggested a new classification for New World sand flies, elevating several subgenera to the genus level. Elsewhere in the world, the genus Phlebotomus is considered the vector of leishmaniasis.[14]
Organisms
Visceral disease is usually caused by Leishmania donovani, L. infantum, or L. chagasi,[3] but occasionally these species may cause other forms of disease.[3] The cutaneous form of the disease is caused by more than 15 species of Leishmania.[3]
Risk factors
Risk factors include poverty, malnutrition, deforestation, lack of sanitation, suppressed immune system and urbanization.[2]
Diagnosis
Leishmaniasis is diagnosed in the hematology laboratory by direct visualization of the amastigotes (Leishman–Donovan bodies). Buffy-coat preparations of peripheral blood or aspirates from marrow, spleen, lymph nodes, or skin lesions should be spread on a slide to make a thin smear and stained with Leishman stain or Giemsa stain (pH 7.2) for 20 minutes. Amastigotes are seen within blood and spleen monocytes or, less commonly, in circulating neutrophils and in aspirated tissue macrophages. They are small, round bodies 2–4 μm in diameter with indistinct cytoplasm, a nucleus, and a small, rod-shaped kinetoplast. Occasionally, amastigotes may be seen lying free between cells.[15] However, the retrieval of tissue samples is often painful for the patient and identification of the infected cells can be difficult. So, other indirect immunological methods of diagnosis are developed, including enzyme-linked immunosorbent assay, antigen-coated dipsticks, and direct agglutination test. Although these tests are readily available, they are not the standard diagnostic tests due to their insufficient sensitivity and specificity.
Several different polymerase chain reaction (PCR) tests are available for the detection of Leishmania DNA.[3] With this assay, a specific and sensitive diagnostic procedure is finally possible. The most sensitive PCR tests use minicircle kinetoplast DNA found in the parasite. Kinetoplast DNA contains sequences for mitochondrial proteins in its maxicircles(~25-50 per parasite), and guide RNA in its minicircles(~10'000 per parasite) of the kinetoplast. With this specific method, one can still detect Leishmania even with a very low parasite load. When needing to diagnose a specific species of Leishmania, as opposed to only detection, other PCR methods have been superior.[16]
Most forms of the disease are transmitted only from nonhuman animals, but some can be spread between humans. Infections in humans are caused by about 21 of 30 species that infect mammals;[17] the different species look the same, but they can be differentiated by isoenzyme analysis, DNA sequence analysis, or monoclonal antibodies.
Prevention
- Leishmaniasis can be partly prevented by using nets treated with insecticide while sleeping.[2] To provide good protection against sandflies, fine mesh sizes of 0.6 mm or less are required, but a mosquito net with 1.2mm mesh will provide a limited reduction in the number of sandfly bites.[18] Finer mesh sizes have the downside of higher cost and reduced air circulation which can cause overheating. Many Phlebotomine sandfly attacks occur at sunset rather than at night, so it may also be useful to put nets over doors and windows or to use insect repellents.
- Use of insecticide-impregnated dog collars and treatment or culling of infected dogs.
- Spraying houses and animal shelters with insecticides.[18]
Treatment
The treatment is determined by where the disease is acquired, the species of Leishmania, and the type of infection.[2] For visceral leishmaniasis in India, South America, and the Mediterranean, liposomal amphotericin B is the recommended treatment and is often used as a single dose.[3][7] Rates of cure with a single dose of amphotericin have been reported as 95%.[3] In India, almost all infections are resistant to pentavalent antimonials.[3] In Africa, a combination of pentavalent antimonials and paromomycin is recommended.[7] These, however, can have significant side effects.[3] Miltefosine, an oral medication, is effective against both visceral and cutaneous leishmaniasis.[8] Side effects are generally mild, though it can cause birth defects if taken within 3 months of getting pregnant.[3][8] It does not appear to work for L. major or L. braziliensis.[9]
The evidence around the treatment of cutaneous leishmaniasis is poor.[3] A number of topical treatments may be used for cutaneous leishmaniasis. Which treatments are effective depends on the strain, with topical paromomycin effective for L. major, L. tropica, L. mexicana, L. panamensis, and L. braziliensis.[9] Pentamidine is effective for L. guyanensis.[9] Oral fluconazole or itraconazole appears effective in L. major and L. tropica.[3][9] There is limited evidence to support the use of heat therapy in cutaneous leishmaniasis as of 2015.[19]
Epidemiology
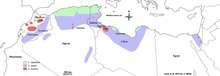
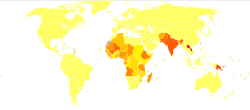
Out of 200 countries and territories reporting to WHO, 97 countries and territories are endemic for leishmaniasis.[21] The settings in which leishmaniasis is found range from rainforests in Central and South America to deserts in western Asia and the Middle East. It affects as many as 12 million people worldwide, with 1.5–2.0 million new cases each year.[22] The visceral form of leishmaniasis has an estimated incidence of 500,000 new cases.[23] In 2014, more than 90% of new cases reported to WHO occurred in six countries: Brazil, Ethiopia, India, Somalia, South Sudan and Sudan.[24] As of 2010, it caused about 52,000 deaths, down from 87,000 in 1990.[10] Different types of the disease occur in different regions of the world.[2] Cutaneous disease is most common in Afghanistan, Algeria, Brazil, Colombia, and Iran, while mucocutaneous disease is most common in Bolivia, Brazil, and Peru, and visceral disease is most common in Bangladesh, Brazil, Ethiopia, India, and Sudan.[2]
Leishmaniasis is found through much of the Americas from northern Argentina to South Texas, though not in Uruguay or Chile, and has recently been shown to be spreading to North Texas.[25] Leishmaniasis is also known as papalomoyo, papa lo moyo, úlcera de los chicleros, and chiclera in Latin America.[26] During 2004, an estimated 3,400 troops from the Colombian army, operating in the jungles near the south of the country (in particular around the Meta and Guaviare departments), were infected with leishmaniasis. Allegedly, a contributing factor was that many of the affected soldiers did not use the officially provided insect repellent because of its disturbing odor. Nearly 13,000 cases of the disease were recorded in all of Colombia throughout 2004, and about 360 new instances of the disease among soldiers had been reported in February 2005.[27]
The disease is found across much of Asia, and in the Middle East. Within Afghanistan, leishmaniasis occurs commonly in Kabul, partly due to bad sanitation and waste left uncollected in streets, allowing parasite-spreading sand flies an environment they find favorable.[28][29] In Kabul, the number of people infected was estimated to be at least 200,000, and in three other towns (Herat, Kandahar, and Mazar-i-Sharif) about 70,000 more occurred, according to WHO figures from 2002.[30][31] Kabul is estimated as the largest center of cutaneous leishmaniasis in the world, with around 67,500 cases as of 2004.[32] Africa, in particular the East and North,[20] is also home to cases of leishmaniasis.
Leishmaniasis is mostly a disease of the developing world, and is rarely known in the developed world outside a small number of cases, mostly in instances where troops are stationed away from their home countries. Leishmaniasis has been reported by U.S. troops stationed in Saudi Arabia and Iraq since the Gulf War of 1990, including visceral leishmaniasis.[33][34][35] In September 2005, the disease was contracted by at least four Dutch marines who were stationed in Mazar-i-Sharif, Afghanistan, and subsequently repatriated for treatment.[36][37]
History
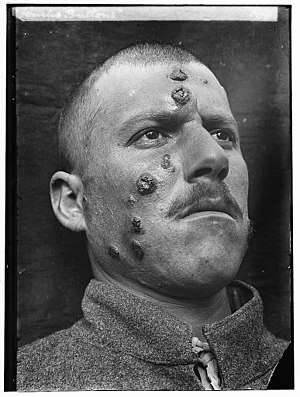
Descriptions of conspicuous lesions similar to cutaneous leishmaniasis appear on tablets from King Ashurbanipal from the seventh century BCE, some of which may have derived from even earlier texts from 1500 to 2500 BCE. Persian physicians, including Avicenna in the 10th century CE, gave detailed descriptions of what was called balkh sore.[38] In 1756, Alexander Russell, after examining a Turkish patient, gave one of the most detailed clinical descriptions of the disease. Physicians in the Indian subcontinent would describe it as kala-azar (pronounced kālā āzār, the Urdu, Hindi, and Hindustani phrase for "black fever", kālā meaning black and āzār meaning fever or disease). In the Americas, evidence of the cutaneous form of the disease in Ecuador and Peru appears in pre-Inca pottery depicting skin lesions and deformed faces dating back to the first century CE. Some 15th- and 16th-century texts from the Inca period and from Spanish colonials mention "valley sickness", "Andean sickness", or "white leprosy", which are likely to be the cutaneous form.[39]
It remains unclear who first discovered the organism. David Douglas Cunningham, Surgeon Major of the British Indian army, may have seen it in 1885 without being able to relate it to the disease.[40][41] Peter Borovsky, a Russian military surgeon working in Tashkent, conducted research into the etiology of "oriental sore", locally known as sart sore, and in 1898 published the first accurate description of the causative agent, correctly described the parasite's relation to host tissues and correctly referred it to the protozoa. However, because his results were published in Russian in a journal with low circulation, his results were not internationally acknowledged during his lifetime.[42] In 1901, Leishman identified certain organisms in smears taken from the spleen of a patient who had died from "dum-dum fever" (Dum Dum is an area close to Calcutta) and proposed them to be trypanosomes, found for the first time in India.[43] A few months later, Captain Charles Donovan (1863–1951) confirmed the finding of what became known as Leishman-Donovan bodies in smears taken from people in Madras in southern India.[44] But it was Ronald Ross who proposed that Leishman-Donovan bodies were the intracellular stages of a new parasite, which he named Leishmania donovani.[45] The link with the disease kala-azar was first suggested by Charles Donovan, and was conclusively demonstrated by Charles Bentley's discovery of L. donovani in patients with kala-azar.[46] The disease became a major problem for Allied troops fighting in Sicily during the Second World War; research by Leonard Goodwin then showed pentostam was an effective treatment.[47]
Society and culture
The Institute for OneWorld Health has reintroduced the drug paromomycin for treatment of leishmaniasis, results with which led to its approval as an orphan drug. The Drugs for Neglected Diseases Initiative is also actively facilitating the search for novel therapeutics. A treatment with paromomycin will cost about $10. The drug had originally been identified in the 1960s, but had been abandoned because it would not be profitable, as the disease mostly affects poor people.[48] The Indian government approved paromomycin for sale in August 2006.[49]
By 2012 the World Health Organization had successfully negotiated with the manufacturers to achieve a reduced cost for liposomal amphotericin B, to $18 a vial, but a number of vials are needed for treatment and it must be kept at a stable, cool temperature.[3]
Research

As of 2017, no vaccine for humans was available.[50][51]
People who recover from cutaneous leishmaniasis are protected against future infections, and based on this some traditional societies have for centuries intentionally infected vulnerable people in inconspicuous locations; bedouins for example have sandflies bite their children's buttocks, and people in the middle east have transferred fluid from lesions on infected people to non-infected people using thorns.[52][53] This process is called "leishmanization".[54]
In the early 1900s scientists learned how to culture the parasite, and work in the 1940s led by Saul Adler led to the practice of leishmanization being widespread in Israel and Russia until the 1980s, when large-scale clinical trials showed that the practice led to long-term skin lesions, exacerbation of psoriasis, and immunosuppression in some people.[52][53] During the Iran–Iraq War over 2 million people in Iran were vaccinated this way.[55][56] As of 2006 such vaccines were still licensed and used in Uzbekistan.[55]
Clinical trials with killed parasites had conflicting results in the 1940s, and work on such vaccines did not resume until the 1970s, when there were promising small clinical trials, and which continued with extensive clinical trials in Ecuador, Brazil, in Iran, through the 1990s.[52]
Preclinical work with genetically-modified live attenuated parasite vaccines was conducted in the 1990s and 2000s, as did work with synthetic peptides, recombinant proteins, glycoproteins and glycolipids from leishmania species, and naked DNA.[52][54] As of 2016, none of these second-generation vaccine candidates had reached the market, and few had been tested in clinical trials.[50]
In 2008, a vaccine for dogs was launched in Brazil, which is known as LeishTech, a recombinant protein based vaccine.[57] In 2011 CaniLeish, a vaccine made with antigens from L. infantum, was licensed in Europe.[54][57] Efforts are on ongoing to develop further vaccines for dogs with one second generation vaccine up for approval in Brazil as of 2017.[51]
See also
- CVBD Canine vector-borne diseases
- Tropical disease
References
- "Leishmaniasis definition and meaning | Collins English Dictionary". Archived from the original on 24 December 2013. Retrieved 23 December 2013.
- "Leishmaniasis Fact sheet N°375". World Health Organization. January 2014. Archived from the original on 21 February 2014. Retrieved 17 February 2014.
- Barrett, MP; Croft, SL (2012). "Management of trypanosomiasis and leishmaniasis". British Medical Bulletin. 104: 175–96. doi:10.1093/bmb/lds031. PMC 3530408. PMID 23137768.
- "Leishmaniasis Magnitude of the problem". World Health Organization. Archived from the original on 26 October 2013. Retrieved 17 February 2014.
- GBD 2015 Disease and Injury Incidence and Prevalence, Collaborators. (8 October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- GBD 2015 Mortality and Causes of Death, Collaborators. (8 October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/s0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- Sundar, S; Chakravarty, J (January 2013). "Leishmaniasis: an update of current pharmacotherapy". Expert Opinion on Pharmacotherapy. 14 (1): 53–63. doi:10.1517/14656566.2013.755515. PMID 23256501.
- Dorlo, TP; Balasegaram, M; Beijnen, JH; de Vries, PJ (November 2012). "Miltefosine: a review of its pharmacology and therapeutic efficacy in the treatment of leishmaniasis". The Journal of Antimicrobial Chemotherapy. 67 (11): 2576–97. doi:10.1093/jac/dks275. PMID 22833634.
- Minodier, P; Parola, P (May 2007). "Cutaneous leishmaniasis treatment". Travel Medicine and Infectious Disease. 5 (3): 150–8. doi:10.1016/j.tmaid.2006.09.004. PMID 17448941.
- Lozano, R (15 December 2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet. 380 (9859): 2095–128. doi:10.1016/S0140-6736(12)61728-0. hdl:10536/DRO/DU:30050819. PMID 23245604.
- Ejazi, SA; Ali, N (January 2013). "Developments in diagnosis and treatment of visceral leishmaniasis during the last decade and future prospects". Expert Review of Anti-infective Therapy. 11 (1): 79–98. doi:10.1586/eri.12.148. PMID 23428104.
- "Neglected Tropical Diseases". cdc.gov. 6 June 2011. Archived from the original on 4 December 2014. Retrieved 28 November 2014.
- James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. pp. 422–428. ISBN 978-0-7216-2921-6.
- Myler P; Fasel Nhirf; f). (2008). Leishmania: After The Genome. Caister Academic Press. ISBN 978-1-904455-28-8. Archived from the original on 23 April 2008.
- Dacie, John V.; Bain, Barbara J.; Imelda Bates (2006). Dacie and Lewis practical hematology. Philadelphia: Churchill Livingstone/Elsevier. ISBN 978-0-443-06660-3.
- Bensoussan, Esther; Nasereddin, Abedelmajeed; Jonas, Flory; Schnur, Lionel F; Jaffe, Charles L. (2006). "Comparison of PCR Assays for Diagnosis of Cutaneous Leishmaniasis". Journal of Clinical Microbiology. 44 (4): 1435–1439. doi:10.1128/JCM.44.4.1435-1439.2006. PMC 1448629. PMID 16597873.
- "Parasites - Leishmaniasis". Centers for Disease Control and Prevention. January 2013.
-
- Alexander; Maroli (2003), "Control of phlebotomine sandflies", Medical and Veterinary Entomology, 17 (1): 1–18, doi:10.1046/j.1365-2915.2003.00420.x, PMID 12680919
- Von Stebut, E (March 2015). "Leishmaniasis". Journal der Deutschen Dermatologischen Gesellschaft. 13 (3): 191–200, quiz 201. doi:10.1111/ddg.12595. PMID 25721626.
- Aoun, K.; Bouratbine, A. (2014). "Cutaneous Leishmaniasis in North Africa: a review". Parasite. 21: 14. doi:10.1051/parasite/2014014. PMC 3952656. PMID 24626301.

- "Leishmaniasis: Situation and trends". WHO Global Health Observatory. Retrieved 30 May 2018.
- Leishmaniasis: Magnitude of the problem Archived 26 October 2013 at the Wayback Machine. World Health Organization.
- "Hope for tropical disease vaccine". BBC News. 23 April 2006. Archived from the original on 27 April 2006.
- "Epidemiological situation: Epidemiology". World Health Organization. Retrieved 30 May 2018.
- "Dallas News: Rare, non-fatal skin disease found in N. Texans". Archived from the original on 27 December 2009. Retrieved 2 June 2015.
- "Papalomoyo" (PDF). Archived from the original (PDF) on 23 July 2011. Retrieved 16 August 2010.
- "Informes - Informe de Fronteras Febrero 2005". Servicio Jesuita a Refugiados. Archived from the original on 10 November 2005.
- Al Jazeera English – CENTRAL/S. ASIA – Kabul: A city in intensive care Archived June 18, 2007, at the Wayback Machine
- Robert Birsel (7 May 2007). "Disfiguring skin disease plagues Afghanistan". e-Ariana. Reuters. Archived from the original on 10 December 2015. Retrieved 8 December 2015.
- Robert Birsel (28 June 2002). "Disfiguring epidemic hits 270,000 Afghans". e-Ariana. Reuters. Archived from the original on 10 December 2015. Retrieved 8 December 2015.
- "WHO Seeking Funds to Prevent Leishmaniasis Outbreak in Afghanistan". voanews. October 2009.
- World Health Organization action in Afghanistan aims to control debilitating leishmaniasis. Archived 26 October 2010 at the Wayback Machine
- Kennedy, Kelly (30 March 2010). "VCS Advocacy in the News: VA May Designate 9 Infectious Diseases as Related to Gulf War". Veterans for Common Sense. Archived from the original on 13 February 2011. Retrieved 10 February 2011.
- Business: Company's mesh will help troops beat 'Baghdad boils' Archived 16 March 2005 at the Wayback Machine
- "Archived copy" (PDF). Archived from the original (PDF) on 12 October 2007. Retrieved 17 September 2007.CS1 maint: archived copy as title (link)
- Saurabh Bhatia, Divakar Goli (2016). Leishmaniasis: Biology, Control and New Approaches for Its Treatment. CRC Press. ISBN 9781315341897.
- Pieter-Paul van Thiel, Tjalling Leenstra, Henry J. de Vries, Allard van der Sluis, Tom van Gool, Alex C. Krull, Michèle van Vugt, Peter J. de Vries, Jimmy E. Zeegelaar, Aldert Bart, Wendy F. van der Meide, Henk D. F. H. Schallig, William R. Faber, and Piet A. Kager (December 2010). "Cutaneous Leishmaniasis (Leishmania major Infection) in Dutch Troops Deployed in Northern Afghanistan: Epidemiology, Clinical Aspects, and Treatment". Am J Trop Med Hyg. 83 (6): 1295–300. doi:10.4269/ajtmh.2010.10-0143. PMC 2990047. PMID 21118937.CS1 maint: multiple names: authors list (link)
- Cox, Francis E G (1996). The Wellcome Trust illustrated history of tropical diseases. Clinical Microbiology Reviews. 15. London: The Wellcome Trust. pp. 206–217. doi:10.1128/CMR.15.4.595-612.2002. ISBN 978-1-869835-86-6. OCLC 35161690. PMC 126866. PMID 12364371.
- "WHO: Leishmaniasis background information – a brief history of the disease". Archived from the original on 15 March 2014.
- Cunningham, DD (1885). On the presence of peculiar parasitic organisms in the tissue of a specimen of Delhi boil. Scientific memoirs officers Medical Sanitary Departments Government India. Calcutta: Printed by the superintendent of government printing, India. pp. 21–31. OCLC 11826455.
- Cox FE (2002). "History of Human Parasitology". Clin. Microbiol. Rev. 15 (4): 595–612. doi:10.1128/CMR.15.4.595-612.2002. PMC 126866. PMID 12364371.
- Hoare C.A. (1938). "Early discoveries regarding the parasite of oriental sore". Transactions of the Royal Society of Tropical Medicine and Hygiene. 32 (1): 67–92. doi:10.1016/S0035-9203(38)90097-5.
- Leishman, W. B. (1903). "On the possibility of the occurrence of trypanomiasis in India". The British Medical Journal. 1 (2213): 1252–1254. doi:10.1136/bmj.1.2213.1252. PMC 2514706.
- Donovan, C. (1903). "Memoranda: On the possibility of the occurrence of trypanomiasis in India". The British Medical Journal.
- R. Ross (1903). "Further notes on Leishman's bodies". The British Medical Journal. 2 (2239): ii: 1401. doi:10.1136/bmj.2.2239.1401. PMC 2514909. PMID 20761210.
- C. A. Bentley (24 December 1903). "Telegram to R. Ross". Ross Archives: 47/157.
- "Leonard Goodwin – Telegraph". The Daily Telegraph. 14 January 2009. Archived from the original on 20 April 2009. Retrieved 18 January 2009.
- A Small Charity Takes the Reins in Fighting a Neglected Disease Archived 20 December 2016 at the Wayback Machine, New York Times, July 31, 2006.
- "Drug Program – Clinical Trial of Paromomycin". Institute for OneWorld Health. Archived from the original on 6 June 2010. Retrieved 10 February 2011.
- Srivastava, S; Shankar, P; Mishra, J; Singh, S (12 May 2016). "Possibilities and challenges for developing a successful vaccine for leishmaniasis". Parasites & Vectors. 9 (1): 277. doi:10.1186/s13071-016-1553-y. PMC 4866332. PMID 27175732.
- Ghorbani, M; Farhoudi, R (2018). "Leishmaniasis in humans: drug or vaccine therapy?". Drug Design, Development and Therapy. 12: 25–40. doi:10.2147/DDDT.S146521. PMC 5743117. PMID 29317800.
- Handman, E (April 2001). "Leishmaniasis: current status of vaccine development". Clinical Microbiology Reviews. 14 (2): 229–43. doi:10.1128/CMR.14.2.229-243.2001. PMC 88972. PMID 11292637.
- Palatnik-de-Sousa, CB (25 March 2008). "Vaccines for leishmaniasis in the fore coming 25 years". Vaccine. 26 (14): 1709–24. doi:10.1016/j.vaccine.2008.01.023. PMID 18295939.
- Dunning, N. (17 February 2009). "Leishmania vaccines: from leishmanization to the era of DNA technology". Bioscience Horizons. 2 (1): 73–82. doi:10.1093/biohorizons/hzp004.
- Khamesipour, A; Rafati, S; Davoudi, N; Maboudi, F; Modabber, F (March 2006). "Leishmaniasis vaccine candidates for development: a global overview" (PDF). The Indian Journal of Medical Research. 123 (3): 423–38. PMID 16778321. Archived (PDF) from the original on 8 September 2017.
- Azizi MH, MH; et al. (2016). "History of Leishmaniasis in Iran from 19th Century Onward" (PDF). Arch Iran Med. 19 (2): 153–162. Archived (PDF) from the original on 16 January 2017.
- Bowman, Dwight D. (2014). Georgis' Parasitology for Veterinarians (10th ed.). Elsevier Health Sciences. p. 440. ISBN 9781455739882. Archived from the original on 8 September 2017.
External links
| Classification | |
|---|---|
| External resources |
| Wikimedia Commons has media related to Leishmaniasis. |