Hyperglycemia
Hyperglycemia (also spelled hyperglycaemia or hyperglycæmia), is a condition in which an excessive amount of glucose circulates in the blood plasma. This is generally a blood sugar level higher than 11.1 mmol/l (200 mg/dl), but symptoms may not start to become noticeable until even higher values such as 15–20 mmol/l (~250–300 mg/dl). A subject with a consistent range between ~5.6 and ~7 mmol/l (100–126 mg/dl) (American Diabetes Association guidelines) is considered slightly hyperglycemic, while above 7 mmol/l (126 mg/dl) is generally held to have diabetes. For diabetics, glucose levels that are considered to be too hyperglycemic can vary from person to person, mainly due to the person's renal threshold of glucose and overall glucose tolerance. On average however, chronic levels above 10–12 mmol/L (180–216 mg/dl) can produce noticeable organ damage over time.
| Hyperglycemia | |
|---|---|
| Other names | High blood sugar |
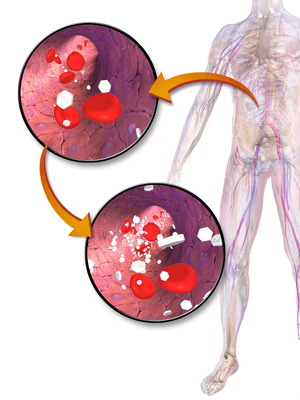 | |
| White hexagons in the image represent glucose molecules, which are increased in the lower image. | |
| Specialty | Endocrinology |
Signs and symptoms
The degree of hyperglycemia can change over time depending on the metabolic cause, for example, impaired glucose tolerance or fasting glucose, and it can depend on treatment.[1] Temporary hyperglycemia is often benign and asymptomatic. Blood glucose levels can rise well above normal and cause pathological and functional changes for significant periods without producing any permanent effects or symptoms. [1] During this asymptomatic period, an abnormality in carbohydrate metabolism can occur which can be tested by measuring plasma glucose. [1] However, chronic hyperglycemia at above normal levels can produce a very wide variety of serious complications over a period of years, including kidney damage, neurological damage, cardiovascular damage, damage to the retina or damage to feet and legs. Diabetic neuropathy may be a result of long-term hyperglycemia. Impairment of growth and susceptibility to certain infection can occur as a result of chronic hyperglycemia.[1]
Acute hyperglycemia involving glucose levels that are extremely high is a medical emergency and can rapidly produce serious complications (such as fluid loss through osmotic diuresis). It is most often seen in persons who have uncontrolled insulin-dependent diabetes.
The following symptoms may be associated with acute or chronic hyperglycemia, with the first three composing the classic hyperglycemic triad:
- Polyphagia – frequent hunger, especially pronounced hunger
- Polydipsia – frequent thirst, especially excessive thirst
- Polyuria – increased volume of urination (not an increased frequency, although it is a common consequence)
- Blurred vision
- Fatigue
- Restlessness
- Weight loss
- Poor wound healing (cuts, scrapes, etc.)
- Dry mouth
- Dry or itchy skin
- Tingling in feet or heels
- Erectile dysfunction
- Recurrent infections, external ear infections (swimmer's ear)
- Cardiac arrhythmia
- Stupor
- Coma
- Seizures
Frequent hunger without other symptoms can also indicate that blood sugar levels are too low. This may occur when people who have diabetes take too much oral hypoglycemic medication or insulin for the amount of food they eat. The resulting drop in blood sugar level to below the normal range prompts a hunger response.
Polydipsia and polyuria occur when blood glucose levels rise high enough to result in excretion of excess glucose via the kidneys, which leads to the presence of glucose in the urine. This produces an osmotic diuresis.
Signs and symptoms of diabetic ketoacidosis may include:
- Ketoacidosis
- Kussmaul hyperventilation (deep, rapid breathing)
- Confusion or a decreased level of consciousness
- Dehydration due to glycosuria and osmotic diuresis
- Increased thirst
- 'Fruity' smelling breath odor
- Nausea and vomiting
- Impairment of cognitive function, along with increased sadness and anxiety[2][3]
- Weight loss
Hyperglycemia causes a decrease in cognitive performance, specifically in processing speed, executive function, and performance.[4] Decreased cognitive performance may cause forgetfulness and concentration loss.[4]
Complications
In untreated hyperglycemia, a condition called ketoacidosis may develop because decreased insulin levels increase the activity of hormone sensitive lipase.[5] The degradation of triacylglycerides by hormone-sensitive lipase produces free fatty acids that are eventually converted to acetyl-coA by beta-oxidation.
Ketoacidosis is a life-threatening condition which requires immediate treatment. Symptoms include: shortness of breath, breath that smells fruity (such as pear drops), nausea and vomiting, and very dry mouth. Chronic hyperglycemia (high blood sugar) injures the heart in patients without a history of heart disease or diabetes and is strongly associated with heart attacks and death in subjects with no coronary heart disease or history of heart failure.[6]
Also, life-threatening consequences of hyperglycemia is nonketotic hyperosmolar syndrome.[1]
Perioperative hyperglycemia has been associated with immunosuppression, increased infections, osmotic diuresis, delayed wound healing, delayed gastric emptying, sympatho-adrenergic stimulation, and increased mortality. In addition, it reduces skin graft success, exacerbates brain, spinal cord, and renal damage by ischemia, worsens neurologic outcomes in traumatic head injuries, and is associated with postoperative cognitive dysfunction following CABG.[7]
Causes
Diabetes mellitus
Chronic hyperglycemia that persists even in fasting states is most commonly caused by diabetes mellitus. In fact, chronic hyperglycemia is the defining characteristic of the disease. Intermittent hyperglycemia may be present in prediabetic states. Acute episodes of hyperglycemia without an obvious cause may indicate developing diabetes or a predisposition to the disorder.
In diabetes mellitus, hyperglycemia is usually caused by low insulin levels (Diabetes mellitus type 1) and/or by resistance to insulin at the cellular level (Diabetes mellitus type 2), depending on the type and state of the disease. Low insulin levels and/or insulin resistance prevent the body from converting glucose into glycogen (a starch-like source of energy stored mostly in the liver), which in turn makes it difficult or impossible to remove excess glucose from the blood. With normal glucose levels, the total amount of glucose in the blood at any given moment is only enough to provide energy to the body for 20–30 minutes, and so glucose levels must be precisely maintained by the body's internal control mechanisms. When the mechanisms fail in a way that allows glucose to rise to abnormal levels, hyperglycemia is the result.
Ketoacidosis may be the first symptom of immune-mediated diabetes, particularly in children and adolescents. [1] Also, patients with immune-mediated diabetes, can change from modest fasting hyperglycemia to severe hyperglycemia and even ketoacidosis as a result of stress or an infection. [1]
Insulin resistance
Obesity has been contributing to increased insulin resistance due to the population's daily caloric intake rising. Insulin resistance increases hyperglycemia because the body becomes over saturated by glucose. Insulin resistance desensitizes insulin receptors, preventing insulin from lowering blood sugar levels. [8]
Drugs
Certain medications increase the risk of hyperglycemia, including corticosteroids, octreotide, beta blockers, epinephrine, thiazide diuretics, statins, niacin, pentamidine, protease inhibitors, L-asparaginase,[9] and antipsychotics.[10] The acute administration of stimulants such as amphetamines typically produces hyperglycemia; chronic use, however, produces hypoglycemia. Some of the newer psychiatric medications, such as Zyprexa (Olanzapine) and Cymbalta (Duloxetine), can also cause significant hyperglycemia.
Thiazides are used to treat type 2 diabetes but it also causes severe hyperglycemia. [1]
Critical illness
The following conditions can also cause hyperglycemia in the absence of diabetes. 1) Dysfunction of the thyroid, adrenal, and pituitary glands 2) Numerous diseases of the pancreas 3) Severe increases in blood glucose may be seen in sepsis and certain infections 4) Intracranial diseases (frequently overlooked) can also cause hyperglycemia. Encephalitis, brain tumors (especially those located near the pituitary gland), brain bleeds, and meningitis are prime examples. 5) Mild to high blood sugar levels are often seen in convulsions and terminal stages of many diseases. Prolonged, major surgeries can temporarily increase glucose levels.[citation needed] Certain forms of severe stress and physical trauma can increase levels for a brief time as well yet rarely exceeds 6.6 mmol/l (120 mg/dl).
Stress
A high proportion of patients suffering an acute stress such as stroke or myocardial infarction may develop hyperglycemia, even in the absence of a diagnosis of diabetes. (Or perhaps stroke or myocardial infarction was caused by hyperglycemia and undiagnosed diabetes.) Human and animal studies suggest that this is not benign, and that stress-induced hyperglycemia is associated with a high risk of mortality after both stroke and myocardial infarction.[11]
Somatostatinomas and aldosteronoma-induced hypokalemia can cause hyperglycemia but usually disappears after the removal of the tumour. [1]
Mechanism
Hormones such as the growth hormone, glucagon, cortisol and catecholamines, can cause hyperglycemia when they are present in the body in excess amounts. [1] Also, increased proinflammatory cytokines that interrupt carbohydrate metabolism, leading to excessive glucose production and reduced uptake in tissues, can cause hyperglycemia. [12]
Environmental Factors
Hyperglycemia is lower in higher income groups since there is access to better education and resources. Low-middle income groups are more likely to develop hyperglycemia due education and access to food options. [13] Living in warmer climates can reduce hyperglycemia due to increased physical activity while people are less active in colder climates. [14]
Population
Hyperglycemia one of the main symptoms of diabetes and it has substantially affected the population making it an epidemic due to the population's increased calorie consumption.[15] Healthcare providers are trying to work more closely with people allowing them more freedom with interventions that suit their lifestyle.[16] As physical inactivity and calorie consumption increases it makes individuals more susceptible to developing hyperglycemia. [17] Hyperglycemia is caused by type 1 diabetes and non-whites have a higher susceptibility for it. [18]
Diagnosis
Monitoring
It is critical for patients who monitor glucose levels at home to be aware of which units of measurement their testing kit uses. Glucose levels are measured in either:
- Millimoles per liter (mmol/l) is the SI standard unit used in most countries around the world.
- Milligrams per deciliter (mg/dl) is used in some countries such as the United States, Japan, France, Egypt and Colombia.
Scientific journals are moving towards using mmol/l; some journals now use mmol/l as the primary unit but quote mg/dl in parentheses.[19]
Glucose levels vary before and after meals, and at various times of day; the definition of "normal" varies among medical professionals. In general, the normal range for most people (fasting adults) is about 4 to 6 mmol/l or 80 to 110 mg/dl. (where 4 mmol/l or 80 mg/dl is "optimal".) A subject with a consistent range above 7 mmol/l or 126 mg/dl is generally held to have hyperglycemia, whereas a consistent range below 4 mmol/l or 70 mg/dl is considered hypoglycemic. In fasting adults, blood plasma glucose should not exceed 7 mmol/l or 126 mg/dL. Sustained higher levels of blood sugar cause damage to the blood vessels and to the organs they supply, leading to the complications of diabetes.[20]
Chronic hyperglycemia can be measured via the HbA1c test. The definition of acute hyperglycemia varies by study, with mmol/l levels from 8 to 15 (mg/dl levels from 144 to 270).[21]
Defects in insulin secretion, insulin action, or both, results in hyperglycemia.[1]
Chronic hyperglycemia can be measured by Clinical urine tests which can detect sugar in the urine or microalbuminuria which could be a symptom of diabetes[22]

Treatment
Treatment of hyperglycemia requires elimination of the underlying cause, such as diabetes. Acute hyperglycemia can be treated by direct administration of insulin in most cases. Severe hyperglycemia can be treated with oral hypoglycemic therapy and lifestyle modification.[23]

In diabetes mellitus (by far the most common cause of chronic hyperglycemia), treatment aims at maintaining blood glucose at a level as close to normal as possible, in order to avoid serious long-term complications. This is done by a combination of proper diet, regular exercise, and insulin or other medication such as metformin, etc.
Those with hyperglycaemia can be treated using sulphonylureas or metformin or both. These drugs help by improving glycaemic control.[24] Dipeptidyl peptidase 4 inhibitor alone or in combination with basal insulin can be used as a treatment for hyperglycemia with patients still in hospital. [12]
Increasing aerobic exercise to at least 30 minutes will make better use of glucose accumulated in the body since glucose is being used for energy by the muscle.[25]
Calorie restriction would be one of the main lifestyle changes because it reduces over eating which contributes to hyperglycemia.[26]
Diets higher in healthy saturated fats and whole wheat carbohydrates such as the Mediterranean diet can help reduce carbohydrate intake to better control hyperglycemia. [27] Diets such as intermittent fasting and ketogenic diet help reduce calorie consumption which could significantly reduce hyperglycemia.
Carbohydrates are the main cause for hyperglycemia, whole wheat items should be substituted for non whole wheat items. Fruits are apart of a complete nutritious diet, but should be limited due to its high sugar content.[28]
Etymology
The origin of the term is Greek: prefix ὑπέρ- hyper- "over-", γλυκός glycos "sweet wine, must", αἷμα haima "blood", -ία, -εια -ia suffix for abstract nouns of feminine gender.
References
- American Diabetes Association (2014). "Diagnosis and Classification of Diabetes Mellitus". Diabetes Care. 37: S81–S90. doi:10.2337/dc14-s081. PMC 3632174. PMID 24357215.
- Pais I, Hallschmid M, Jauch-Chara K, et al. (2007). "Mood and cognitive functions during acute euglycaemia and mild hyperglycaemia in type 2 diabetic patients". Exp. Clin. Endocrinol. Diabetes. 115 (1): 42–46. doi:10.1055/s-2007-957348. PMID 17286234.
- Sommerfield AJ, Deary IJ, Frier BM (2004). "Acute hyperglycemia alters mood state and impairs cognitive performance in people with type 2 diabetes". Diabetes Care. 27 (10): 2335–40. doi:10.2337/diacare.27.10.2335. PMID 15451897.
- Geijselaers, Stefan L.C.; Sep, Simone J.S.; Claessens, Danny; Schram, Miranda T.; Van Boxtel, Martin P.J.; Henry, Ronald M.A.; Verhey, Frans R.J.; Kroon, Abraham A.; Dagnelie, Pieter C.; Schalkwijk, Casper G.; Van Der Kallen, Carla J.H.; Biessels, Geert Jan; Stehouwer, Coen D.A. (2017). "The Role of Hyperglycemia, Insulin Resistance, and Blood Pressure in Diabetes-Associated Differences in Cognitive Performance—The Maastricht Study". Diabetes Care. 40 (11): 1537–1547. doi:10.2337/dc17-0330. PMID 28842522.
- Kraemer, Fredric B.; Shen, Wen-Jun (2002). "Hormone-sensitive lipase". Journal of Lipid Research. 43 (10): 1585–1594. doi:10.1194/jlr.R200009-JLR200. ISSN 0022-2275. PMID 12364542.
- "Chronic hyperglycemia may lead to cardiac damage". Journal of the American College of Cardiology. 2012-02-03. Retrieved 3 February 2012.
- Miller, Miller’s Anesthesia, 7th edition, pp. 1716, 2674, 2809.
- "Adipose Tissue Insulin Resistance in Youth on the Spectrum From Normal Weight to Obese and From Normal Glucose Tolerance to Impaired Glucose Tolerance to Type 2 Diabetes". Diabetes care. 42(2): 265–272. 2019.
- Cetin M, Yetgin S, Kara A, et al. (1994). "Hyperglycemia, ketoacidosis and other complications of L-asparaginase in children with acute lymphoblastic leukemia". J Med. 25 (3–4): 219–29. PMID 7996065.
- Luna B, Feinglos MN (2001). "Drug-induced hyperglycemia". JAMA. 286 (16): 1945–48. doi:10.1001/jama.286.16.1945. PMID 11667913.
- Capes SE, Hunt D, Malmberg K, Pathak P, Gerstein HC (2001). "Stress hyperglycemia and prognosis of stroke in nondiabetic and diabetic patients: a systematic overview". Stroke. 32 (10): 2426–32. doi:10.1161/hs1001.096194. PMID 11588337.
- Umpierrez, Guillermo E.; Pasquel, Francisco J. (2017). "Management of Inpatient Hyperglycemia and Diabetes in Older Adults". Diabetes Care. 40 (4): 509–517. doi:10.2337/dc16-0989. PMC 5864102. PMID 28325798.
- Ma, Ronald CW; Popkin, Barry M (2017). "Intergenerational diabetes and obesity—A cycle to break?". PLOS ONE. 14(10).
- Ishii, MD1, Hajime; Suzuki, MD1, Hodaka; Baba, MD1, Tsuneharu; Nakamura, BS2, Keiko; Watanabe, MD1, Tsuyoshi (2001). "Seasonal Variation of Glycemic Control in Type 2 Diabetic Patients". American Diabetes Association.
- American Diabetes Association (2019). "Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2019" (PDF). Diabetes care. 42(supplement 1): S13–S28.
- Inzucchi, MD, Silvio E; Bergenstal, MD, Richard M; Buse, MD, PHD, John B; Diamant, MD, PHD, Michaela; Ferrannini, MD, Ele; Nauck, MD, Michael; Peters, MD, Anne L; Tsapas, MD, PHD, Apostolos; Wender, MD, Richard; Matthews, MD, DPHIL, David R (2012). "Management of Hyperglycemia in Type 2 Diabetes: A Patient-Centered Approach". Diabetes care. 35(6): 1364–1370.CS1 maint: multiple names: authors list (link)
- Lee IM, Shiroma EJ, Lobelo F, Puska P, Blair SN, Katzmarzyk PT (2012). "Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy". Lancet. 380 (9838): 219–229.
- "Diabetes in Normal-Weight Individuals: High Susceptibility in Nonwhite Populations". Diabetes Care. 42(12): 2164–2166. 2019.
- "diabetes FAQ: general (part 1 of 5)Section - What are mg/dl and mmol/l? How to convert? Glucose? Cholesterol?". www.faqs.org.
- Total Health Life (2005). "High Blood Sugar". Total Health Institute. Retrieved May 4, 2011.
- Giugliano D, Marfella R, Coppola L, et al. (1997). "Vascular effects of acute hyperglycemia in humans are reversed by L-arginine. Evidence for reduced availability of nitric oxide during hyperglycemia". Circulation. 95 (7): 1783–90. doi:10.1161/01.CIR.95.7.1783. PMID 9107164.
- Florvall, Gösta; Basu, PHD, Samar; Helmersson, PHD, Johanna; Larsson, MD, PHD, Anders (2006). "Hemocue Urine Albumin Point-Of-Care Test Shows Strong Agreement With the Results Obtained With a Large Nephelometer". Diabetes care. 29(2): 422–423.CS1 maint: multiple names: authors list (link)
- Ron Walls; John J. Ratey; Robert I. Simon (2009). Rosen's Emergency Medicine: Expert Consult Premium Edition – Enhanced Online Features and Print (Rosen's Emergency Medicine: Concepts & Clinical Practice (2v.)). St. Louis: Mosby. ISBN 978-0-323-05472-0.
- Pearson, Ewan R.; Starkey, Bryan J.; Powell, Roy J.; Gribble, Fiona M.; Clark, Penny M.; Hattersley, Andrew T. (2003). "Genetic cause of hyperglycaemia and response to treatment in diabetes". The Lancet. 362 (9392): 1275–1281. doi:10.1016/s0140-6736(03)14571-0. PMID 14575972.
- Aronson, Ronnie; Brown, Ruth E; Li, Aihua; Riddell, Michael C. "Optimal Insulin Correction Factor in Post–High-Intensity Exercise Hyperglycemia in Adults With Type 1 Diabetes: The FIT Study". American Diabetes Association. 42(1): 10–16.
- "High Blood sugar". Total health institute. 2005.
- Mattei, Josiemer; Bigornia, Sherman J; Sotos-Prieto, Mercedes; Scott, Tammy; Gao, Xiang; Tucker, Katherine L (2019). "The Mediterranean Diet and 2-Year Change in Cognitive Function by Status of Type 2 Diabetes and Glycemic Control". Diabetes care. 42(8): 1372–1379.
- "Dietary Guidelines 2015-2020". US Department of Health. 2015.
External links
| Classification | |
|---|---|
| External resources |