Halobacterium salinarum
Halobacterium salinarum is an extremely halophilic marine obligate aerobic archaeon. Despite its name, this is not a bacterium, but rather a member of the domain Archaea.[1] It is found in salted fish, hides, hypersaline lakes, and salterns. As these salterns reach the minimum salinity limits for extreme halophiles, their waters become purple or reddish color due to the high densities of halophilic Archaea.[1] H. salinarum has also been found in high-salt food such as salt pork, marine fish, and sausages. The ability of H. salinarum to survive at such high salt concentrations has led to its classification as an extremophile.
| Halobacterium salinarum | |
|---|---|
| Scientific classification | |
| Domain: | |
| Kingdom: | |
| Phylum: | |
| Class: | |
| Order: | |
| Family: | |
| Genus: | |
| Species: | H. salinarum |
| Binomial name | |
| Halobacterium salinarum corrig. (Harrison and Kennedy 1922) Elazari-Volcani 1957 | |
| Synonyms | |
|
Pseudomonas salinaria Harrison and Kennedy 1922 | |
Cell morphology and metabolism
Halobacteria are single-celled, rod-shaped microorganisms that are among the most ancient forms of life and appeared on Earth billions of years ago. The membrane consists of a single lipid monolayer surrounded by an S-layer.[2] The S-layer is made of a cell-surface glycoprotein, which accounts for approximately 50% of the cell surface proteins.[3] These proteins form a lattice in the membrane. Sulfate residues are abundant on the glycan chains of the glycoprotein, giving it a negative charge. The negative charge is believed to stabilize the lattice in high-salt conditions.[4]
Amino acids are the main source of chemical energy for H. salinarum, particularly arginine and aspartate, though they are able to metabolize other amino acids, as well.[2] H. salinarum have been reported to not be able to grow on sugars, and therefore need to encode enzymes capable of performing gluconeogenesis to create sugars. Although "H. salinarum" is unable to catabolize glucose, the transcription factor TrmB has been proven to regulate the gluconeogenic production of sugars found on the S-layer glycoprotein.
Adaptation to extreme conditions
High salt
To survive in extremely salty environments, this archaeon—as with other halophilic Archaeal species—utilizes compatible solutes (in particular potassium chloride) to reduce osmotic stress.[5] Potassium levels are not at equilibrium with the environment, so H. salinarum expresses multiple active transporters which pump potassium into the cell.[2] At extremely high salt concentrations protein precipitation will occur. To prevent the salting out of proteins, H. salinarum encodes mainly acidic proteins. The average isoelectric point of H. salinarum proteins is 5.03.[6] These highly acidic proteins are overwhelmingly negative in charge and are able to remain in solution even at high salt concentrations.[1]
Low oxygen and phototrophy
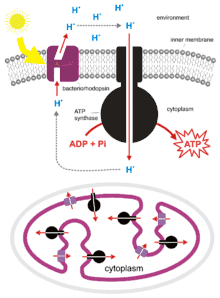
H. salinarum can grow to such densities in salt ponds that oxygen is quickly depleted. Though it is an obligate aerobe, it is able to survive in low-oxygen conditions by utilizing light-energy. H. salinarum express the membrane protein bacteriorhodopsin[9] which acts as a light-driven proton pump. It consists of two parts, the 7-transmembrane protein, bacterioopsin, and the light-sensitive cofactor, retinal. Upon absorption of a photon, retinal changes conformation, causing a conformational change in the bacterioopsin protein which drives proton transport.[10] The proton gradient which is formed can then be used to generate chemical energy by ATP synthase.
To obtain more oxygen H. salinarum produce gas vesicles, which allow them to float to the surface where oxygen levels are higher and more light is available.[11] These vesicles are complex structures made of proteins encoded by at least 14 genes.[12] Gas vesicles were first discovered in H. salinarum in 1967.[13]
UV protection
There is little protection from the Sun in salt ponds, so H. salinarum are often exposed to high amounts of UV radiation. To compensate, they have evolved a sophisticated DNA repair mechanism. The genome encodes DNA repair enzymes homologous to those in both bacteria and eukaryotes.[1] This allows H. salinarum to repair damage to DNA faster and more efficiently than other organisms and allows them to be much more UV tolerant.
H. salinarum is responsible for the bright pink or red appearance of the Dead Sea and other bodies of salt water. This red color is due primarily to the presence of bacterioruberin, a 50 carbon carotenoid pigment present within the membrane of H. salinarum. The primary role of bacterioruberin in the cell is to protect against DNA damage incurred by UV light.[14] This protection is not, however, due to the ability of bacterioruberin to absorb UV light. Bacterioruberin protects the DNA by acting as an antioxidant, rather than directly blocking UV light.[15] It is able to protect the cell from reactive oxygen species produced from exposure to UV by acting as a target. The bacterioruberin radical produced is less reactive than the initial radical, and will likely react with another radical, resulting in termination of the radical chain reaction.[16]
Protection against ionizing radiation and desiccation
H. salinarum is polyploid[17] and highly resistant to ionizing radiation and desiccation, conditions that induce DNA double-strand breaks.[18] Although chromosomes are initially shattered into many fragments, complete chromosomes are regenerated by making use of over-lapping fragments. Regeneration occurs by a process involving DNA single-stranded binding protein, and is likely a form of homologous recombinational repair.[19]
Genome
Whole genome sequences are available for two strains of H. salinarum, NRC-1[2] and R1.[20] The Halobacterium sp. NRC-1 genome consists of 2,571,010 base pairs on one large chromosome and two mini-chromosomes. The genome encodes 2,360 predicted proteins.[2] The large chromosome is very G-C rich (68%).[21] High GC-content of the genome increases stability in extreme environments. Whole proteome comparisons show the definite archaeal nature of this halophile with additional similarities to the Gram-positive Bacillus subtilis and other bacteria.
As a model organism
H. salinarum is as easy to culture as E. coli and serves as an excellent model system. Methods for gene replacement and systematic knockout have been developed,[22] so H. salinarum is an ideal candidate for the study of archaeal genetics and functional genomics.
For hydrogen production
Hydrogen production using H. salinarum coupled to a hydrogenase donor like E. coli are reported in literature.[23]
Oldest DNA ever recovered
A sample of a close genetic relative of H. salinarum encapsulated inments estimated at 121 million years old. Oddly, the material had been also recovered earlier, but it proved to be so similar to the modern descendants that scientists had believed the earlier samples were contaminated.
Scientists have previously recovered similar genetic material from the Michigan Basin, the same region where the latest discovery was made. But that DNA, discovered in a salt-cured buffalo hide in the 1930s, was so similar to that of modern microbes that many scientists believed the samples had been contaminated.[24] The curing salt had been derived from a mine in Saskatchewan, the site of the most recent sample described by Jong Soo Park of Dalhousie University in Halifax, Nova Scotia, Canada.[25]
Russell Vreeland of Ancient Biomaterials Institute of West Chester University in Pennsylvania, USA, performed an analysis of all known halophilic bacteria, which yielded the finding that Park's bacteria contained six segments of DNA never seen before in the halophiles. Vreeland also tracked down the buffalo skin and determined that the salt came from the same mine as Park's sample. He has also discovered an even older halophile estimated at 250 million years old in New Mexico.[26] However, his findings date the crystal surrounding the bacteria, and DNA analysis suggests the bacteria themselves are likely to be less ancient.[27]
References
- Dassarma, Shiladitya (2007). "Extreme Microbes". American Scientist. 95 (3): 224. doi:10.1511/2007.65.224.
- Ng, W. V.; Kennedy, S. P.; Mahairas, G. G.; Berquist, B; Pan, M; Shukla, H. D.; Lasky, S. R.; Baliga, N. S.; Thorsson, V; Sbrogna, J; Swartzell, S; Weir, D; Hall, J; Dahl, T. A.; Welti, R; Goo, Y. A.; Leithauser, B; Keller, K; Cruz, R; Danson, M. J.; Hough, D. W.; Maddocks, D. G.; Jablonski, P. E.; Krebs, M. P.; Angevine, C. M.; Dale, H; Isenbarger, T. A.; Peck, R. F.; Pohlschroder, M; et al. (2000). "Genome sequence of Halobacterium species NRC-1". Proceedings of the National Academy of Sciences. 97 (22): 12176–81. Bibcode:2000PNAS...9712176N. doi:10.1073/pnas.190337797. PMC 17314. PMID 11016950.
- Mescher, M. F.; Strominger, J. L. (1976). "Purification and characterization of a prokaryotic glucoprotein from the cell envelope of Halobacterium salinarium". The Journal of Biological Chemistry. 251 (7): 2005–14. PMID 1270419.
- Sára, M; Sleytr, U. B. (2000). "S-Layer proteins". Journal of Bacteriology. 182 (4): 859–68. doi:10.1128/jb.182.4.859-868.2000. PMC 94357. PMID 10648507.
- Pérez-Fillol, M; Rodríguez-Valera, F (1986). "Potassium ion accumulation in cells of different halobacteria". Microbiología. 2 (2): 73–80. PMID 3271061.
- Kozlowski, LP (26 October 2016). "Proteome-pI: proteome isoelectric point database". Nucleic Acids Research. 45 (D1): D1112–D1116. doi:10.1093/nar/gkw978. PMC 5210655. PMID 27789699.
- Nicholls D. G.; Ferguson S. J. (1992). Bioenergetics 2 (2nd ed.). San Diego: Academic Press. ISBN 9780125181242.
- Stryer, Lubert (1995). Biochemistry (fourth ed.). New York – Basingstoke: W. H. Freeman and Company. ISBN 978-0716720096.
- Oesterhelt, D; Stoeckenius, W (1973). "Functions of a new photoreceptor membrane". Proceedings of the National Academy of Sciences of the United States of America. 70 (10): 2853–7. Bibcode:1973PNAS...70.2853O. doi:10.1073/pnas.70.10.2853. PMC 427124. PMID 4517939.
- Andersson, M; Malmerberg, E; Westenhoff, S; Katona, G; Cammarata, M; Wöhri, A. B.; Johansson, L. C.; Ewald, F; Eklund, M; Wulff, M; Davidsson, J; Neutze, R (2009). "Structural dynamics of light-driven proton pumps". Structure. 17 (9): 1265–75. doi:10.1016/j.str.2009.07.007. PMID 19748347.
- Oren, A., Ecology of extremely halophilic microorganisms, Vreeland, R.H., Hochstein, L.I., editors, The Biology of Halophilic Bacteria, CRC Press, Inc., Boca Raton, Florida, 1993, p. 25–54.
- Walsby, A. E. (1994). "Gas vesicles". Microbiological Reviews. 58 (1): 94–144. PMC 372955. PMID 8177173.
- Larsen, H; Omang, S; Steensland, H (1967). "On the gas vacuoles of the halobacteria". Archiv für Mikrobiologie. 59 (1): 197–203. doi:10.1007/bf00406332. PMID 5602456.
- Shahmohammadi, H. R.; Asgarani, E; Terato, H; Saito, T; Ohyama, Y; Gekko, K; Yamamoto, O; Ide, H (1998). "Protective roles of bacterioruberin and intracellular KCl in the resistance of Halobacterium salinarium against DNA-damaging agents". Journal of Radiation Research. 39 (4): 251–62. Bibcode:1998JRadR..39..251S. doi:10.1269/jrr.39.251. PMID 10196780.
- Ide, H., Takeshi, S., Hiroaki, T., Studies on the antioxidation activity of bacterioruberin, Urakami Found Mem, 1998, 6:127–33.
- Saito, T., Miyabe, Y., Ide, H., Yamamoto, O., Hydroxyl radical scavenging ability of bacterioruberin, Radiat Phys Chem, 1997, 50(3):267–9.
- Soppa J (2011). "Ploidy and gene conversion in Archaea". Biochem. Soc. Trans. 39 (1): 150–4. doi:10.1042/BST0390150. PMID 21265763.
- Kottemann M, Kish A, Iloanusi C, Bjork S, DiRuggiero J (2005). "Physiological responses of the halophilic archaeon Halobacterium sp. strain NRC1 to desiccation and gamma irradiation". Extremophiles. 9 (3): 219–27. doi:10.1007/s00792-005-0437-4. PMID 15844015.
- DeVeaux LC, Müller JA, Smith J, Petrisko J, Wells DP, DasSarma S (2007). "Extremely radiation-resistant mutants of a halophilic archaeon with increased single-stranded DNA-binding protein (RPA) gene expression". Radiat. Res. 168 (4): 507–14. Bibcode:2007RadR..168..507D. doi:10.1667/RR0935.1. PMID 17903038.
- Pfeiffer, F; Schuster, S. C.; Broicher, A; Falb, M; Palm, P; Rodewald, K; Ruepp, A; Soppa, J; Tittor, J; Oesterhelt, D (2008). "Evolution in the laboratory: The genome of Halobacterium salinarum strain R1 compared to that of strain NRC-1". Genomics. 91 (4): 335–46. doi:10.1016/j.ygeno.2008.01.001. PMID 18313895.
- Joshi, J. G.; Guild, W. R.; Handler, P (1963). "The presence of two species of DNA in some halobacteria". Journal of Molecular Biology. 6: 34–8. doi:10.1016/s0022-2836(63)80079-0. PMID 13964964.
- Peck, R. F.; Dassarma, S; Krebs, M. P. (2000). "Homologous gene knockout in the archaeon Halobacterium salinarum with ura3 as a counterselectable marker". Molecular Microbiology. 35 (3): 667–76. doi:10.1046/j.1365-2958.2000.01739.x. PMID 10672188.
- Brijesh Rajanandam, K S; Siva Kiran, R R (2011). "Optimization of hydrogen production by Halobacterium salinarium coupled with E coli using milk plasma as fermentative substrate". Journal of Biochemical Technology. 3 (2): 242–244. ISSN 0974-2328.
- Reilly, Michael; The Discovery Channel. "World's oldest known DNA discovered". Retrieved 3 September 2010.
- Park, J. S.; Vreeland, R. H.; Cho, B. C.; Lowenstein, T. K.; Timofeeff, M. N. & Rosenweig, W. D. (December 2009). "Haloarchaeal diversity in 23, 121 and 419 MYA salts". Geobiology. 7 (5): 515–23. doi:10.1111/j.1472-4669.2009.00218.x. PMID 19849725.
- Vreeland, H; Rosenzweig, W D; Lowenstein, T; Satterfield, C; Ventosa, A (December 2006). "Fatty acid and DNA analyses of Permian bacteria isolated from ancient salt crystals reveal differences with their modern relatives". Extremophiles. 10 (1): 71–8. doi:10.1007/s00792-005-0474-z. PMID 16133658.
- Graur, Dan; Pupko, Tal (2001-02-15). "The Permian Bacterium that Isn't". Molecular Biology and Evolution. Oxford Journals. 18 (6): 1143–1146. doi:10.1093/oxfordjournals.molbev.a003887. PMID 11371604. Retrieved 2010-11-16.
Further reading
- Horia, Todor; Dulmage, Keely; Gillum, Nicholas; Bain, James; Muehlbauer, Michael; Schmid, Amy (2014). "A transcription factor links growth rate and metabolism in the hypersaline adapted archaeon Halobacterium salinarum". Molecular Microbiology. 93 (6): 1172–1182. doi:10.1111/mmi.12726. PMID 25060603.
- Kahaki, Fatemeh Abarghooi; Babaeipour, Valiollah; Memari, Hamid Rajabi; Mofid, Mohammad Reza (2014). "High Overexpression and Purification of Optimized Bacterio-Opsin from Halobacterium Salinarum R1 in E-coli". Applied Biochemistry and Biotechnology. 174 (4): 1558–1571. doi:10.1007/s12010-014-1137-2. PMID 25123363.