Foscarnet
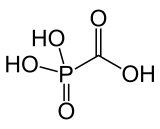
Foscarnet (phosphonomethanoic acid) is an antiviral medication which is primarily used to treat viral infections involving the Herpesviridae family. It is classified as a pyrophosphate analog DNA polymerase inhibitor. Foscarnet is the conjugate base of a chemical compound with the formula HO2CPO3H2.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Foscavir |
| Other names | phosphonomethanoic acid, dihydroxyphosphinecarboxylic acid oxide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601144 |
| Pregnancy category | |
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | 14–17% |
| Elimination half-life | 3.3–6.8 hours |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | CH3O5P |
| Molar mass | 126.005 g/mol 300.1 g/mol (foscarnet trisodium hexahydrate) g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Foscarnet was approved for medical use in 1991.[1]
Medical use
This phosphonic acid derivative (marketed by Clinigen as foscarnet sodium under the trade name Foscavir) is an antiviral medication used to treat herpes viruses, including drug-resistant cytomegalovirus (CMV) and herpes simplex viruses types 1 and 2 (HSV-1 and HSV-2). It is particularly used to treat CMV retinitis. Foscarnet can be used to treat highly treatment-experienced patients with HIV as part of salvage therapy.[2][3][4]
Mechanism of action
Foscarnet is a structural mimic of the anion pyrophosphate[5] that selectively inhibits the pyrophosphate binding site on viral DNA polymerases at concentrations that do not affect human DNA polymerases.
In individuals treated with the DNA polymerase inhibitors acyclovir or ganciclovir, HSV or CMV particles can develop mutant protein kinases (thymidine kinase or UL97 protein kinase, respectively) that make them resistant to these antiviral drugs. However, unlike acyclovir and ganciclovir, foscarnet is not activated by viral protein kinases, making it useful in acyclovir- or ganciclovir-resistant HSV and CMV infections.
However, acyclovir- or ganciclovir-resistant mutants with alterations in viral DNA polymerase may also be resistant to foscarnet.[6][7]
Administration
Intravenous infusion or intravitreous injection.
Side effects
- Nephrotoxicity—Increase in serum creatinine levels occurs on average in 45% of patients receiving foscarnet. Other nephrotoxic drugs should be avoided. Nephrotoxicity is usually reversible and can be reduced by dosage adjustment and adequate hydration.
- Electrolyte disturbances—Changes in calcium, magnesium (Harisson 16th ed page2244) potassium and phosphate levels occurs commonly and regular monitoring of electrolytes is necessary to avoid clinical toxicity.
- Genital ulceration—Occurs more commonly in men and usually occurs during induction use of foscarnet. It is most likely a contact dermatitis due to high concentrations of foscarnet in urine. It usually resolves rapidly following discontinuation of the drug.
- CNS—Paraesthesias, irritability and hallucinations.
References
- Long, Sarah S.; Pickering, Larry K.; Prober, Charles G. (2012). Principles and Practice of Pediatric Infectious Disease. Elsevier Health Sciences. p. 1502. ISBN 978-1437727029.
- Canestri A, Ghosn J, Wirden M, et al. (2006). "Foscarnet salvage therapy for patients with late-stage HIV disease and multiple drug resistance". Antivir. Ther. (Lond.). 11 (5): 561–6. PMID 16964823.
- Mathiesen S, Dam E, Roge B, et al. (2007). "Long-term foscarnet therapy remodels thymidine analogue mutations and alters resistance to zidovudine and lamivudine in HIV-1". Antivir. Ther. (Lond.). 12 (3): 335–43. PMID 17591023.
- Meyer PR, Rutvisuttinunt W, Matsuura SE, So AG, Scott WA (2007). "Stable complexes formed by HIV-1 reverse transcriptase at distinct positions on the primer-template controlled by binding deoxynucleoside triphosphates or foscarnet". J. Mol. Biol. 369 (1): 41–54. doi:10.1016/j.jmb.2007.03.006. PMC 1986715. PMID 17400246.
- Meyer PR, Rutvisuttinunt W, Matsuura SE, So AG, Scott WA (May 2007). "Stable complexes formed by HIV-1 reverse transcriptase at distinct positions on the primer-template controlled by binding deoxynucleoside triphosphates or foscarnet". J. Mol. Biol. 369 (1): 41–54. doi:10.1016/j.jmb.2007.03.006. PMC 1986715. PMID 17400246.
- Bonnafous P, Naesens L, Petrella S, et al. (2007). "Different mutations in the HHV-6 DNA polymerase gene accounting for resistance to foscarnet". Antivir. Ther. (Lond.). 12 (6): 877–88. PMID 17926642.
- Tchesnokov EP, Gilbert C, Boivin G, Götte M (February 2006). "Role of helix P of the human cytomegalovirus DNA polymerase in resistance and hypersusceptibility to the antiviral drug foscarnet". J. Virol. 80 (3): 1440–50. doi:10.1128/JVI.80.3.1440-1450.2006. PMC 1346920. PMID 16415021.
Sources
- Harrison Textbook of Medicine 16th ed, page 2244