Fluspirilene
Fluspirilene (Redeptin, Imap, R6218) is a diphenylbutylpiperidine typical antipsychotic drug, used for the treatment of schizophrenia.[1] It is administered intramuscularly.[2] It was discovered at Janssen Pharmaceutica in 1963.[3] A 2007 systematic review investigated the efficacy of fluspirilene decanoate for people with schizophrenia:
| Summary | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Participant numbers in each comparison were small so power to identify clear difference is limited. Randomized controlled trial data identified no clear differences between the long-acting injection of fluspirilene and oral medication for outcomes that include adverse effects.[4] | ||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | IM |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.015.835 |
| Chemical and physical data | |
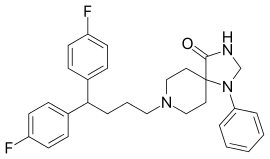
| Formula | C29H31F2N3O |
| Molar mass | 475.573 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
See also
References
- van Epen JH (1970). "Experience with fluspirilene (R 6218), a long-acting neuroleptic". Psychiatr Neurol Neurochir. 73 (4): 277–284. PMID 5478771.
- Janssen PA, Niemegeers CJ, Schellekens KH, Lenaerts FM, Verbruggen FJ, van Nueten JM, Marsboom RH, Hérin VV, Schaper WK (1970). "The pharmacology of fluspirilene (R 6218), a potent, long-acting and injectable neuroleptic drug". Arzneimittelforschung. 20 (11): 1689–1698. PMID 4992598.
- C. Janssen, NV Res. Lab., BE 633914 (1963)
- Abhijnhan, A; Adams, C; David, A (2007). "Depot fluspirilene for schizophrenia". Cochrane Database of Systematic Reviews. 1: CD001718.pub2. doi:10.1002/14651858.CD001718.pub2.
| Typical |
|
|---|---|
| Disputed | |
| Atypical |
|
| Others | |
| |
| D1-like |
| ||||
|---|---|---|---|---|---|
| D2-like |
| ||||
| |||||
Histamine receptor modulators | |
|---|---|
| H1 |
|
| H2 |
|
| H3 |
|
| H4 |
|
See also: Receptor/signaling modulators • Monoamine metabolism modulators • Monoamine reuptake inhibitors | |
Serotonin receptor modulators | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.