Cytotoxic T cell
A cytotoxic T cell (also known as TC, cytotoxic T lymphocyte, CTL, T-killer cell, cytolytic T cell, CD8+ T-cell or killer T cell) is a T lymphocyte (a type of white blood cell) that kills cancer cells, cells that are infected (particularly with viruses), or cells that are damaged in other ways.
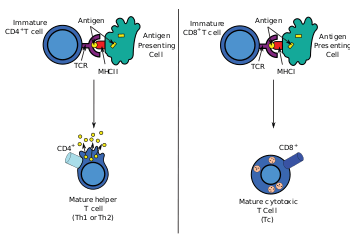
Most cytotoxic T cells express T-cell receptors (TCRs) that can recognize a specific antigen. An antigen is a molecule capable of stimulating an immune response, and is often produced by cancer cells or viruses. Antigens inside a cell are bound to class I MHC molecules, and brought to the surface of the cell by the class I MHC molecule, where they can be recognized by the T cell. If the TCR is specific for that antigen, it binds to the complex of the class I MHC molecule and the antigen, and the T cell destroys the cell.
In order for the TCR to bind to the class I MHC molecule, the former must be accompanied by a glycoprotein called CD8, which binds to the constant portion of the class I MHC molecule. Therefore, these T cells are called CD8+ T cells.
The affinity between CD8 and the MHC molecule keeps the TC cell and the target cell bound closely together during antigen-specific activation. CD8+ T cells are recognized as TC cells once they become activated and are generally classified as having a pre-defined cytotoxic role within the immune system. However, CD8+ T cells also have the ability to make some cytokines.
Development
The immune system must recognize millions of potential antigens. There are fewer than 30,000 genes in the human body, so it is impossible to have one gene for every antigen. Instead, the DNA in millions of white blood cells in the bone marrow is shuffled to create cells with unique receptors, each of which can bind to a different antigen. Some receptors bind to tissues in the human body itself, so to prevent the body from attacking itself, those self-reactive white blood cells are destroyed during further development in the thymus, in which iodine is necessary for its development and activity.[1]
TCRs have two parts, usually an alpha and a beta chain. (Some TCRs have a gamma and a delta chain.) Hematopoietic stem cells in the bone marrow migrate into the thymus, where they undergo V(D)J recombination of their beta-chain TCR DNA to form a developmental form of the TCR protein, known as pre-TCR. If that rearrangement is successful, the cells then rearrange their alpha-chain TCR DNA to create a functional alpha-beta TCR complex. This highly-variable genetic rearrangement product in the TCR genes helps create millions of different T cells with different TCRs, helping the body's immune system respond to virtually any protein of an invader. The vast majority of T cells express alpha-beta TCRs (αβ T cells), but some T cells in epithelial tissues (like the gut) express gamma-delta TCRs (gamma delta T cells), which recognize non-protein antigens.
T cells with functionally stable TCRs express both the CD4 and CD8 co-receptors and are therefore termed "double-positive" (DP) T cells (CD4+CD8+). The double-positive T cells are exposed to a wide variety of self-antigens in the thymus and undergo two selection criteria:
- positive selection, in which those double-positive T cells that bind to foreign antigen in the presence of self MHC. They will differentiate into either CD4+ or CD8+ depending on which MHC is associated with the antigen presented (MHC1 for CD8, MHC2 for CD4). In this case, the cells would have been presented antigen in the context of MHC1. Positive selection means selecting those TCRs capable of recognizing self MHC molecules.
- negative selection, in which those double-positive T cells that bind too strongly to MHC-presented self antigens undergo apoptosis because they could otherwise become autoreactive, leading to autoimmunity.
Only those T cells that bind to the MHC-self-antigen complexes weakly are positively selected. Those cells that survive positive and negative selection differentiate into single-positive T cells (either CD4+ or CD8+), depending on whether their TCR recognizes an MHC class I-presented antigen (CD8) or an MHC class II-presented antigen (CD4). It is the CD8+ T-cells that will mature and go on to become cytotoxic T cells following their activation with a class I-restricted antigen.
Activation
With the exception of some cell types, such as non-nucleated cells (including erythrocytes), Class I MHC is expressed by all host cells. When these cells are infected with a virus (or another intracellular pathogen), the cells degrade foreign proteins via antigen processing. These result in peptide fragments, some of which are presented by MHC Class I to the T cell antigen receptor (TCR) on CD8+ T cells.
The activation of cytotoxic T cells is dependent on several simultaneous interactions between molecules expressed on the surface of the T cell and molecules on the surface of the antigen-presenting cell (APC). For instance, consider the two signal model for TC cell activation.
| Signal | T cell | APC | Description |
| First Signal | TCR | peptide-bound MHC class I molecule | There is a second interaction between the CD8 coreceptor and the class I MHC molecule to stabilize this signal. |
| Second Signal | CD28 molecule on the T cell | either CD80 or CD86 (also called B7-1 and B7-2) | CD80 and CD86 are known as costimulators for T cell activation. This second signal can be assisted (or replaced) by stimulating the TC cell with cytokines released from T helper cells. |
A simple activation of naive CD8+ T cells requires the interaction with professional antigen-presenting cells, mainly with matured dendritic cells. To generate longlasting memory T cells and to allow repetitive stimulation of cytotoxic T cells, dendritic cells have to interact with both, activated CD4+ helper T cells and CD8+ T cells.[2]</ref>[3] During this process, the CD4+ helper T cells "license" the dendritic cells to give a potent activating signal to the naive CD8+ T cells.
Furthermore, maturation of CD8+ T cells is mediated by CD40 signalling.[4] Once the naïve CD8+ T cell is bound to the infected cell, the infected cell is triggered to release CD40.[4] This CD40 release, with the aid of helper T cells, will trigger differentiation of the naïve CD8+ T cells to mature CD8+ T cells.[4]
While in most cases activation is dependent on TCR recognition of antigen, alternative pathways for activation have been described. For example, cytotoxic T cells have been shown to become activated when targeted by other CD8 T cells leading to tolerization of the latter.[5]
Once activated, the TC cell undergoes clonal expansion with the help of the cytokine Interleukin-2 (IL-2), which is a growth and differentiation factor for T cells. This increases the number of cells specific for the target antigen that can then travel throughout the body in search of antigen-positive somatic cells.
Effector functions
When exposed to infected/dysfunctional somatic cells, TC cells release the cytotoxins perforin, granzymes, and granulysin. Through the action of perforin, granzymes enter the cytoplasm of the target cell and their serine protease function triggers the caspase cascade, which is a series of cysteine proteases that eventually lead to apoptosis (programmed cell death).
A second way to induce apoptosis is via cell-surface interaction between the TC and the infected cell. When a TC is activated it starts to express the surface protein FAS ligand (FasL)(Apo1L)(CD95L), which can bind to Fas (Apo1)(CD95) molecules expressed on the target cell. However, this Fas-Fas ligand interaction is thought to be more important to the disposal of unwanted T lymphocytes during their development or to the lytic activity of certain TH cells than it is to the cytolytic activity of TC effector cells. Engagement of Fas with FasL allows for recruitment of the death-induced signaling complex (DISC).[6] The Fas-associated death domain (FADD) translocates with the DISC, allowing recruitment of procaspases 8 and 10.[6] These caspases then activate the effector caspases 3, 6, and 7, leading to cleavage of death substrates such as lamin A, lamin B1, lamin B2, PARP (poly ADP ribose polymerase), and DNA-PKcs (DNA-activated protein kinase). The final result is apoptosis of the cell that expressed Fas.
The transcription factor Eomesodermin is suggested to play a key role in CD8+ T cell function, acting as a regulatory gene in the adaptive immune response.[7] Studies investigating the effect of loss-of-function Eomesodermin found that a decrease in expression of this transcription factor resulted in decreased amount of perforin produced by CD8+ T cells.[7]
Role in disease pathogenesis
During hepatitis B virus (HBV) infection cytotoxic T cells kill infected cells and produce antiviral cytokines capable of purging HBV from viable hepatocytes. They also play an important pathogenic role, contributing to nearly all of the liver injury associated with HBV infection.[8] Platelets have been shown to facilitate the accumulation of virus-specific cytotoxic T cells into the infected liver.[9]
Cytotoxic T cells have been implicated in the progression of arthritis: depletion of knee joint cartilage macromolecules such as glycosaminoglycans by cytotoxic T cells and macrophages has been observed in a rat model of the disease.[10]
CD8+ T cells have been found to play a role in HIV infection. HIV over time has developed many strategies to evade the host cell immune system. For example, HIV has adopted very high mutation rates to allow them to escape recognition by CD8+ T cells.[11] They are also able to down-regulate expression of surface MHC Class I proteins of cells that they infect, in order to further evade destruction by CD8+ T cells.[11] If CD8+ T cells cannot find, recognize and bind to infected cells, the virus will not be destroyed and will continue to grow.
Furthermore, it has been recently discovered that CD8+ T cells play a critical role in Type 1 diabetes.[12] It was previously thought that this autoimmune disease was exclusively controlled by CD4+ cells - but recent studies in a diabetic mouse model showed that CD8+ T cells also engaged in the destruction of insulin-producing pancreatic cells.[12]
See also
References
- Venturi S, Venturi M (September 2009). "Iodine, thymus, and immunity". Nutrition. 25 (9): 977–9. doi:10.1016/j.nut.2009.06.002. PMID 19647627.
- Hivroz C, Chemin K, Tourret M, Bohineust A (2012). "Crosstalk between T lymphocytes and dendritic cells". Critical Reviews in Immunology. 32 (2): 139–55. doi:10.1615/CritRevImmunol.v32.i2.30. PMID 23216612.
- Hoyer S, Prommersberger S, Pfeiffer IA, Schuler-Thurner B, Schuler G, Dörrie J, Schaft N (December 2014). "Concurrent interaction of DCs with CD4(+) and CD8(+) T cells improves secondary CTL expansion: It takes three to tango". European Journal of Immunology. 44 (12): 3543–59. doi:10.1002/eji.201444477. PMID 25211552.
- Bennett SR, Carbone FR, Karamalis F, Flavell RA, Miller JF, Heath WR (June 1998). "Help for cytotoxic-T-cell responses is mediated by CD40 signalling". Nature. 393 (6684): 478–80. Bibcode:1998Natur.393..478B. doi:10.1038/30996. PMID 9624004.
- Milstein O, Hagin D, Lask A, Reich-Zeliger S, Shezen E, Ophir E, Eidelstein Y, Afik R, Antebi YE, Dustin ML, Reisner Y (January 2011). "CTLs respond with activation and granule secretion when serving as targets for T-cell recognition". Blood. 117 (3): 1042–52. doi:10.1182/blood-2010-05-283770. PMC 3035066. PMID 21045195.
- Bakshi RK, Cox MA, Zajac AJ (2014). "Cytotoxic T Lymphocytes". Encyclopedia of Medical Immunology. pp. 332–342. doi:10.1007/978-0-387-84828-0_36. ISBN 978-0-387-84827-3.
- Pearce EL, Mullen AC, Martins GA, Krawczyk CM, Hutchins AS, Zediak VP, Banica M, DiCioccio CB, Gross DA, Mao CA, Shen H, Cereb N, Yang SY, Lindsten T, Rossant J, Hunter CA, Reiner SL (November 2003). "Control of effector CD8+ T cell function by the transcription factor Eomesodermin". Science. 302 (5647): 1041–3. doi:10.1126/science.1090148. PMID 14605368.
- Iannacone M, Sitia G, Guidotti LG (2006). "Pathogenetic and antiviral immune responses against hepatitis B virus". Future Virology. 1 (2): 189–96. doi:10.2217/17460794.1.2.189.
- Iannacone M, Sitia G, Isogawa M, Marchese P, Castro MG, Lowenstein PR, Chisari FV, Ruggeri ZM, Guidotti LG (November 2005). "Platelets mediate cytotoxic T lymphocyte-induced liver damage". Nature Medicine. 11 (11): 1167–9. doi:10.1038/nm1317. PMC 2908083. PMID 16258538.
- Subramanian S, Ramalingam K (2005). "Electron microscopic evidence on the participation Cytotoxic T Lymphocytes and Macrophages in Mtb adjuvant induced connective tissue inflammation and arthritogenesis in Rattus norvegicus". Asian Journal of Microbiology, Biotechnology and Environmental Sciences. 7 (2): 227–233. ISSN 0972-3005.
- Gulzar N, Copeland KF (January 2004). "CD8+ T-cells: function and response to HIV infection". Current HIV Research. 2 (1): 23–37. doi:10.2174/1570162043485077. PMID 15053338.
- Tsai S, Shameli A, Santamaria P (2008). "CD8+ T cells in type 1 diabetes". Advances in Immunology. 100: 79–124. doi:10.1016/S0065-2776(08)00804-3. ISBN 9780123743268. PMID 19111164.
External links

- T-cell Group - Cardiff University