Cys-loop receptor
The Cys-loop ligand-gated ion channel superfamily is composed of nicotinic acetylcholine, GABAA, GABAA-ρ, glycine, 5-HT3, and zinc-activated (ZAC) receptors that are composed of five protein subunits that form a pentameric arrangement around a central pore. There are usually 2 alpha subunits and 3 other beta, gamma, or delta subunits (some consist of 5 alpha subunits). The name "Cys-loop" is because this type of receptors possess a characteristic loop formed by 13 highly conserved amino acids between two cysteine (Cys) residues which form a disulfide bond, near the N-terminal extracellular domain.
Cys-loop receptors are known only in eukaryotes, however they are part of a larger family of pentameric ligand-gated ion channels, which outside the Cys-loop clade are missing the pair of bridging cysteine residues.[1] The larger superfamily includes bacterial (e.g. GLIC) as well as non-Cys-loop eukaryotic receptors, and is referred to as "pentameric ligand-gated ion channels", or "Pro-loop receptors".[2]
All subunits consist of a conserved extracellular large N-terminal domain, three highly conserved transmembrane domains, a cytoplasmic loop of variable size and amino acid sequence, and a fourth transmembrane domain with a relatively short and variable extracellular C terminal. The neurotransmitters bind at the interface between subunits in the extracellular domain.
Each subunit contains 4-membrane-spanning alpha helices (M1, M2, M3, M4). The pore is formed primarily by the M2 helices.[3]
The M3-M4 linker is the intracellular domain that binds the cytoskeleton.
Binding
Much of what we understand about cys-loop receptors comes from inferences made while studying various members of this family. For instance, by studying the structures of acetylcholine binding proteins (AChBP) it has been determined that the binding sites are made up of six loops with the first three forming the principal face and then next three forming the complementary face. The last loop on the principal face wraps over the ligand in the active receptor. This site is also shown repeatedly to be abundant in aromatic residues.[4]
It has been shown in recent literature[4] that the Trp residue on loop B is crucial for both agonist and antagonist binding. The neurotransmitter is taken into the binding site where it interacts (through hydrogen and cation-π bonding) with the amino acid resides in the aromatic box, located on the principal face of the binding site. Another essential interaction is between the agonist and a tyrosine on loop C,[5] which undergoes a conformational change as a result of the interaction and rotates down to cap the molecule in the binding site.
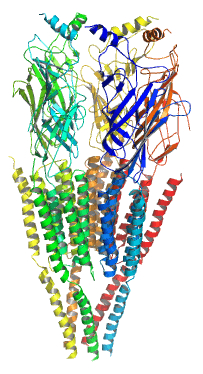
Channel gating
Through research done on nicotinic acetylcholine receptors it has been determined that the channels are activated through allosteric interactions between the binding and gating domains. Once the agonist binds it brings about conformational changes (including moving a beta sheet of the amino-terminal domain, and outward movement from loops 2, F and cys-loop which are tied to the M2-M3 linker and pull the channel open). Electron microscopy (at 9 Å) shows that the opening is caused by rotation at the M2 domain, but other studies on crystal structures of these receptors has shown that the opening could be a result from a M2 tilt which leads to pore dilation and a quaternary turn of the entire pentameric receptor.[6]
References
- Tasneem A, Iyer L, Jakobsson E, Aravind L (2004). "Identification of the prokaryotic ligand-gated ion channels and their implications for the mechanisms and origins of animal Cys-loop ion channels". Genome Biology. 6 (1): R4. doi:10.1186/gb-2004-6-1-r4. PMC 549065. PMID 15642096.
- Jaiteh M, Taly A, Hénin J (2016). "Evolution of Pentameric Ligand-Gated Ion Channels: Pro-Loop Receptors". PLoS One. 11 (3): e0151934. doi:10.1371/journal.pone.0151934. PMC 4795631. PMID 26986966.
- Sine S; Engel A (2006). "Recent advances in Cys-loop receptor structure and function". Nature. 440 (7083): 448–55. doi:10.1038/nature04708. PMID 16554804.
- Van Arnam, EB; Dougherty, DA (August 14, 2014). "Functional probes of drug-receptor interactions implicated by structural studies: cys-loop receptors provide a fertile testing ground". Journal of Medicinal Chemistry. 57 (15): 6289–6300. doi:10.1021/jm500023m. PMC 4136689. PMID 24568098.
- Bourne, Y; et al. (October 19, 2005). "Structures of Aplysia AChBP complexes with nicotinic agonists and antagonists reveal distinctive binding interfaces and conformations". The EMBO Journal. 24 (20): 3635–3646. doi:10.1038/sj.emboj.7600828. PMC 1276711. PMID 16193063.
- Huang, Y; Zhang, JL; Wu, W; Chang, YC (June 2009). "Allosteric activation mechanism of the cys-loop receptors". Acta Pharmacologica Sinica. 30 (6): 663–672. doi:10.1038/aps.2009.51. PMC 4002373. PMID 19444220.
- Yakel, J (February 15, 2010). "Advances and hold-ups in the study of structure, function and regulation of cys-loop ligand-gated ion channels and receptors". The Journal of Physiology. 588 (588 (Pt. 4)): 555–556. doi:10.1113/jphysiol.2009.185488. PMC 2828129. PMID 20173078.
- Pless, SA; Lynagh, T (April 24, 2014). "Principles of agonist recognition in Cys-loop receptors". Frontiers in Physiology. 5: 160. doi:10.3389/fphys.2014.00160. PMC 4006026. PMID 24795655.
- Albuquerque, EX; et al. (2009). "Mammalian Nicotinic Acetylcholine Receptors: From Structure to Function". Physiol Rev. 89 (1): 73–120. doi:10.1152/physrev.00015.2008. PMC 2713585. PMID 19126755.