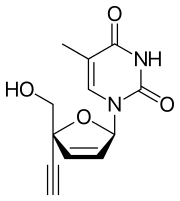
Censavudine
Censavudine (INN),[1] is an investigational new drug being developed by Bristol Myers-Squibb for the treatment of HIV infection. It was originally developed at Yale University.[2]
 | |
 | |
| Clinical data | |
|---|---|
| Other names | 4'-ethynylstavudine, festinavir |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.225.812 |
| Chemical and physical data | |
| Formula | C12H12N2O4 |
| Molar mass | 248.235 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Renaming
Until 2013, censavudine has been known as festinavir, but the name was changed to avoid confusion with HIV protease inhibitors which all bear class suffix "–navir" (e.g. tipranavir, lopinavir, saquinavir etc.).
References
- "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed International Nonproprietary Names: List 110" (PDF). World Health Organization. pp. 409–410.
- Alcorn, Keith (21 December 2010). "Bristol-Myers Squibb buys festinavir, new NRTI active against MDR HIV". aidsmap.com. aidsmap. Retrieved 24 June 2011.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.