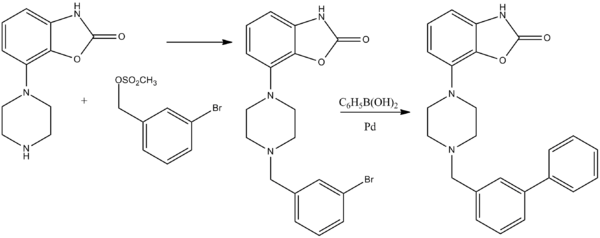
Bifeprunox
Bifeprunox (INN) (code name DU-127,090) is an atypical antipsychotic which, similarly to aripiprazole, combines minimal D2 receptor agonism with serotonin receptor agonism.[1] It was under development for the treatment of schizophrenia but has since been abandoned.[2]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H23N3O2 |
| Molar mass | 385.458 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Bifeprunox has a novel mechanism of action. Conventional antipsychotics are classed into typical and atypical. The typical antipsychotics, such as chlorpromazine and haloperidol, are potent D2 receptor antagonists. The atypical antipsychotics started with clozapine, these are classified as multireceptor interacting compounds, acting as an agonist towards 5-HT1A and an antagonist towards D2 receptors among other 5-HT and DA receptors. Bifeprunox and other novel atypical antipsychotics will instead of antagonizing D2 receptors, will act as partial agonists, as well as partial agonists towards 5-HT1A receptors.[3]
In a multi-center, placebo-controlled study, 20 mg of bifeprunox was found to be significantly more effective than placebo at reducing symptoms of schizophrenia, with a low incidence of side effects.[4]
An NDA for Bifeprunox was filed with the U.S. Food and Drug Administration in January 2007. The FDA rejected the application in August 2007.[5] In June 2009, Solvay and Lundbeck decided to cease development because "efficacy data did not support pursuing the existing development strategy of stabilisation of non-acute patients with schizophrenia."[2]
See also
References
- Cuisiat S, Bourdiol N, Lacharme V, Newman-Tancredi A, Colpaert F, Vacher B (2007). "Towards a new generation of potential antipsychotic agents combining D2 and 5-HT1A receptor activities". J. Med. Chem. 50 (4): 865–76. doi:10.1021/jm061180b. PMID 17300168.
- Pipeline update - following an interim analysis the studies with bifeprunox for the treatment of schizophrenia is discontinued Archived 2011-07-14 at the Wayback Machine Lundbeck Press Release.
- Bardin L, Auclair A, Kleven MS, et al. (2007). "Pharmacological profiles in rats of novel antipsychotics with combined dopamine D2/serotonin 5-HT1A activity: comparison with typical and atypical conventional antipsychotics". Behav Pharmacol. 18 (2): 103–18. doi:10.1097/FBP.0b013e3280ae6c96. PMID 17351418.
- Casey DE, Sands EE, Heisterberg J, Yang HM (October 2008). "Efficacy and safety of bifeprunox in patients with an acute exacerbation of schizophrenia: results from a randomized, double-blind, placebo-controlled, multicenter, dose-finding study". Psychopharmacology. 200 (3): 317–31. doi:10.1007/s00213-008-1207-7. PMID 18597078.
- Wyeth and Solvay say FDA rejects application for antipsychotic drug bifeprunox. Thomson Financial, August 10, 2007.
- Feenstra, R. W.; de Moes, J.; Hofma, J. J.; Kling, H.; Kuipers, W.; Long, S. K.; Tulp, M. T. M.; van der Heyden, J. A. M.; Kruse, C. G. (2001). "New 1-aryl-4-(biarylmethylene)piperazines as potential atypical antipsychotics sharing dopamine D2-receptor and serotonin 5-HT1A-receptor affinities". Bioorg. Med. Chem. Lett. 11 (17): 2345–2349. doi:10.1016/S0960-894X(01)00425-5.