Atovaquone
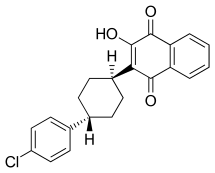
Atovaquone (alternative spelling: atavaquone) is a chemical compound that belongs to the class of naphthoquinones. Atovaquone is a hydroxy-1,4-naphthoquinone, an analog of ubiquinone, with antipneumocystic activity. It is manufactured in the US in the liquid form, or oral suspension, under the brand name Mepron.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Mepron |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693003 |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 2.2–3.2 days |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.158.738 |
| Chemical and physical data | |
| Formula | C22H19ClO3 |
| Molar mass | 366.837 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 216 to 219 °C (421 to 426 °F) |
SMILES
| |
InChI
| |
| | |
Uses
Atovaquone is a medication used to treat or prevent:
- For pneumocystis pneumonia (PCP),[2][3] it is used in mild cases, although it is not approved for treatment of severe cases.
- For toxoplasmosis,[4] the medication has antiparasitic and therapeutic effects.
- For malaria, it is one of the two components (along with proguanil) in the drug Malarone. Malarone has fewer side effects and is more expensive than mefloquine.[5] Resistance has been observed.[6]
- For babesia, it is often used in conjunction with oral azithromycin.[7]
Trimethoprim/sulfamethoxazole (TMP-SMX, Bactrim) is generally considered first-line therapy for PCP or toxoplasmosis. However, atovaquone may be used in patients who cannot tolerate, or are allergic to, sulfonamide medications such as TMP-SMX. In addition, atovaquone has the advantage of not causing myelosuppression, which is an important issue in patients who have undergone bone marrow transplantation.
Atovaquone is given prophylactically to kidney transplant patients to prevent PCP in cases where Bactrim is contraindicated for the patient.
Malaria
Atovaquone, as a combination preparation with proguanil, has been commercially available from GlaxoSmithKline since 2000 as Malarone for the treatment and prevention of malaria.
References
- GlaxoSmithKline (June 2015). "Mepron". Drugs.com. Retrieved June 22, 2016.
- Hughes W, Leoung G, Kramer F, et al. (May 1993). "Comparison of atovaquone (566C80) with trimethoprim-sulfamethoxazole to treat Pneumocystis carinii pneumonia in patients with AIDS". N. Engl. J. Med. 328 (21): 1521–7. doi:10.1056/NEJM199305273282103. PMID 8479489.
- Dohn MN, Weinberg WG, Torres RA, et al. (August 1994). "Oral atovaquone compared with intravenous pentamidine for Pneumocystis carinii pneumonia in patients with AIDS. Atovaquone Study Group". Ann. Intern. Med. 121 (3): 174–80. doi:10.7326/0003-4819-121-3-199408010-00003. PMID 7880228.
- Djurković-Djaković O, Milenković V, Nikolić A, Bobić B, Grujić J (December 2002). "Efficacy of atovaquone combined with clindamycin against murine infection with a cystogenic (Me49) strain of Toxoplasma gondii". J. Antimicrob. Chemother. 50 (6): 981–7. doi:10.1093/jac/dkf251. PMID 12461021.
- Malarone: New Malaria Medication With Fewer Side-effects Archived May 14, 2006, at the Wayback Machine
- Färnert A, Lindberg J, Gil P, et al. (March 2003). "Evidence of Plasmodium falciparum malaria resistant to atovaquone and proguanil hydrochloride: case reports". BMJ. 326 (7390): 628–9. doi:10.1136/bmj.326.7390.628. PMC 151974. PMID 12649236.
- Krause PJ, Lepore T, Sikand VK, et al. (November 2000). "Atovaquone and azithromycin for the treatment of babesiosis". N. Engl. J. Med. 343 (20): 1454–8. doi:10.1056/NEJM200011163432004. PMID 11078770.
External links
| Wikimedia Commons has media related to Atovaquone. |