Abrocitinib
Abrocitinib (code name PF-04965842) is a Janus kinase inhibitor drug which is currently under investigation for the treatment of atopic dermatitis. It was developed by Pfizer.
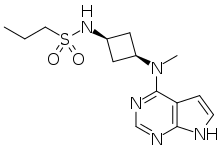 | |
| Clinical data | |
|---|---|
| Other names | PF-04965842 |
| Routes of administration | Oral |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C14H21N5O2S |
| Molar mass | 323.42 g/mol g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Mechanism of action
It is a selective inhibitor of the enzyme janus kinase 1 (JAK1).[1]
Timeline
- April 2016: initiation of Phase 2b trial
- December 2017: initiation of JADE Mono-1 Phase 3 trial[2]
- May 2018: Results of Phase 2b trial posted
References
- "Efficacy and Safety of Oral Janus Kinase 1 Inhibitor Abrocitinib for Patients With Atopic Dermatitis: A Phase 2 Randomized Clinical Trial". JAMA Dermatology. October 2, 2019. doi:10.1001/jamadermatol.2019.2855. Retrieved November 27, 2019.
- "Study to Evaluate Efficacy and Safety of PF-04965842 in Subjects Aged 12 Years And Older With Moderate to Severe Atopic Dermatitis (JADE Mono-1)". ClinicalTrials.gov. Retrieved November 26, 2019.
This article is issued from
Wikipedia.
The text is licensed under Creative
Commons - Attribution - Sharealike.
Additional terms may apply for the media files.